AUG 26, 2016
By Arif O. Khan, MD, and Elias I. Traboulsi, MD
A Compendium of Inherited Disorders and the Eye, Oxford University Press
Genetics
OMIM Numbers
- Aniridia – 106200
- WAGR syndrome – 194072
- Gillespie syndrome – 206700
Inheritance
- Autosomal dominant
- Sporadic in patients with 11p13 deletions
- Autosomal recessive in Gillespie syndrome
Gene/Gene Map
- Autosomal dominant: 11p13 with PAX6 homeobox developmental gene mutations
- Sporadic: chromosomal deletion at 11p13, location of WT1 gene
- Gillespie syndrome: homozygous, compound heterozygous, or heterozygous mutations in the ITPR1 gene on chromosome 3p26.
- Rare cases with preserved visual function and no mutations in PAX6
Epidemiology
- About 1.8 in 100,000 live births
Clinical Findings
There are three recognized clinical occurrences of aniridia:
- 85% of cases are not associated with overt systemic abnormalities, although deep phenotyping can reveal reduced olfaction and brain MRI abnormalities.
- 13% of cases have the WAGR association of Wilms tumor (nephroblastoma), Aniridia, Genitourinary abnormalities, and mental Retardation (Figure 1). Some of these patients have other malformations, such as hemihypertrophy. Ambiguous genitalia, mental and growth retardation, and multiple minor anomalies (anomalies of the pinna, inguinal and umbilical hernias, microcephaly) are part of the WAGR association. 38 % of WAGR patients develop nephropathy and renal failure in their lifetime.
- 2% of cases have Gillespie syndrome of aniridia, developmental delay, and cerebellar ataxia.
There are congenital ocular findings, including bilateral, total, or partial absence of the iris.
- Aniridia is a misnomer, because careful gonioscopic examination of all patients, even those in whom no iris is visible, results in finding some iris tissue. More often than not, some iris tissue is visible (Figure 2). It is important to distinguish aniridia from the anomalous iris that results from mutations in ACTA2. Patients with the mutations in the latter gene have other congenital malformations that involve the heart, aorta, other blood vessels, and smooth muscles.
- Aniridia is a malformation of the whole eyeball, with abnormalities in most ocular tissues. Patients may have microcornea, keratopathy with corneal pannus (Figure 3), and corneal opacification resulting from absence of limbal stem cells.
- Cataracts are found in 85% of cases, and dislocated lenses in 35%.
- Secondary glaucoma occurs in up to 50% of patients; glaucoma usually develops after the second decade of life as the peripheral iris stump adheres to the angle, occluding it. Goniodysgenesis is noted in some cases with rare cases of infantile glaucoma.
- Foveal and optic nerve hypoplasia are frequently present.
- Nystagmus is observed in the majority of cases, and photophobia is common.
- Bilateral ptosis has been reported.
- Visual acuity is less than 20/60 in almost all patients and less than 20/200 in more than 60%.
- Rare families, with a very mild phenotype, have been reported in whom the incidence of cataracts and glaucoma is less than the classical, more common form of the disease, and in whom visual acuity is better preserved. It is more likely that missense mutations in PAX6 cause mild phenotypes than nonsense mutations or deletions.
- Some PAX6 mutations can result in partial phenotypic features of classic aniridia, such as keratitis, foveal hypoplasia, or cataract, without evident or prominent absence of iris tissue.
- Some patients with PAX6 mutations may present with Peters anomaly or with isolated cataracts or foveal hypoplasia.
- Mutations in other genes can cause overlapping phenotypes (eg, FOXC1, CYP1B1) that can sometimes be differentiated by the absence of foveal hypoplasia.
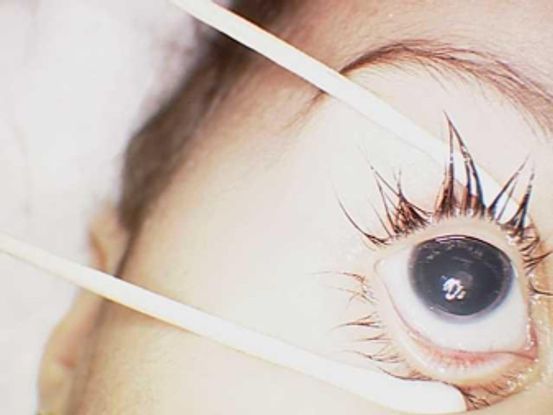
Figure 1. A 4-month old boy was born with a classic aniridia phenotype. Three older siblings were healthy and parents did not have aniridia. Targeted copy number variation at 11p13 confirmed a new heterozygous deletion encompassing both PAX6 and WT1, placing the patient at risk of Wilms tumor.

Figure 2. Iris remnants in a patient with missense mutation in PAX6.

Figure 3. Early superficial keratopathy in a patient with aniridia. As the keratopathy progresses, blood vessels invade the superficial layer of the cornea.
Therapeutic Considerations
- Targeted copy number variation analysis should be obtained in all sporadic cases to rule out a deletion at 11p13.
- All patients with sporadic aniridia in whom mutation analysis of the PAX6 gene has not been done or in whom no intragenic mutation has been demonstrated should have renal sonograms to rule out Wilms tumor. Sonograms are obtained at age 1 year and are continued every 6 months until age 4. Sonograms are not necessary in familial cases, since these are due to intragenic PAX6 mutations with no chance of deletions involving the neighboring Wilms tumor gene.
- Glaucoma and the complications of its surgical treatment are the main causes of acquired vision loss in patients with aniridia. Surgical treatment of the glaucoma may be necessary if it is not controlled by medications. Glaucoma is usually juvenile-onset, although it can be congenital.
- Cataract surgery should be deferred unless the lens opacities are judged to contribute significantly to vision loss.
References
- Elsas FJ, Maumenee IH, Kenyon KR, Yoder F: Familial aniridia with preserved ocular function. Am J Ophthalmol. 1977; 83:718-724.
- Fraumeni JE Jr, Glass AG: Wilms' tumor and congenital aniridia. JAMA. 1968; 206:825-828.
- Gillespie FD: Aniridia, cerebellar ataxia, and oligophrenia in siblings. Arch Ophthalmol. 1965; 73:338-341.
- Glaser T, Walton DS, Maas RL: Genomic structure, evolutionary conservation and aniridia mutations in the human PAX6 gene. Nat Genet. 1992; 2:232-239.
- Glaser T, Ton CC, Mueller R, et al: Absence of PAX6 gene mutations in Gillespie syndrome (partial aniridia cerebellar ataxia and mental retardation). Genomics. 1994;19: 145-148.
- Grant WM, Walton DS: Progressive changes in the angle in congenital aniridia, with development of glaucoma. Am J Ophthalmol. 1974;78:842-847.
- Hanson IM, Fletcher JM, Jordan T, et al: Mutations at the PAX6 locus are found in heterogeneous anterior segment malformations including Peters' anomaly. Nat Genet. 1994; 6:168-173.
- Khan AO, Aldahmesh MA, Al-Abdi L, Mohamed JY, Hashem M, Al-Ghamdi I, Alkuraya FS. Molecular characterization of newborn glaucoma including a distinct aniridic phenotype. Ophthalmic Genet. 2011; 32:138-42.
- Khan AO, Aldahmesh MA, Al-Amri A. Heterozygous FOXC1 mutation (M161K) associated with congenital glaucoma and aniridia in an infant and a milder phenotype in her mother. Ophthalmic Genet. 2008; 29:67-71.
- Milewicz DM, Østergaard JR, Ala-Kokko LM, et al. De novo ACTA2 mutation causes a novel syndrome of multisystemic smooth muscle dysfunction. Am J Med Genet A. 2010; 152A:2437-43.
- Miller RW, Fraumeni JF Jr, Manning MD: Association of Wilms' tumor with aniridia, hemihypertrophy and other congenital malformations. New Engl J Med. 1964; 270:922-927.
- Mirzayans F, Pearce W, MacDonald IM, Walter MA: Mutation of the PAX6 gene in patients with autosomal dominant keratitis. Am J Hum Genet. 1995;57: 539-548.
- Riccardi VM, Hittner HM, Francke UI, et al: The aniridia-Wilms tumor association: the critical role of chromosome band 11p13. Cancer Genet Cytogenet. 1980; 2:131-137.
- Traboulsi EI, Ellison J, Sears J, Maumenee IH, Avallone J, Mohney BG. Aniridia with preserved visual function - a report of four cases with no mutations in PAX6. Am J Ophthalmol. 2008; 145:760-763.
- Yogarajah M, Matarin M, Vollmar C, et al. PAX6, brain structure and function in human adults: advanced MRI in aniridia. Ann Clin Transl Neurol. 2016; 3:314-30.
Traboulsi EI. Compendium of Inherited Disorders and the Eye. New York: Oxford University Press; 2005. Adapted with permission.