AUG 26, 2016
By Arif O. Khan, MD, and Elias I. Traboulsi, MD
A Compendium of Inherited Disorders and the Eye, Oxford University Press
Genetics
Introduction
Microphthalmos, anophthalmos, and coloboma (MAC) represent a spectrum of developmental ocular defects that can occur in isolation or as part of a multisystem syndrome. Nanophthalmos and posterior microphthalmos belong to another spectrum of disease, sometimes termed simple microphthalmos. A complete discussion of the genetics and syndromes related to MAC, nanophthalmos, and posterior microphthalmos is beyond the scope of this entry/article. In addition to selected associated genes, this article reviews selected definitions and clinical aspects of this group of malformations. Table 1 is a practical classification of MAC.
OMIM Numbers
- https://www.ncbi.nlm.nih.gov/omim/?term=microphthalmia
- https://www.ncbi.nlm.nih.gov/omim/?term=anophthalmos
- https://www.ncbi.nlm.nih.gov/omim/?term=coloboma
- https://www.ncbi.nlm.nih.gov/omim/?term=nanophthalmos
- https://www.ncbi.nlm.nih.gov/omim/?term=posterior+microphthalmos
Inheritance
- Sporadic cases are most frequent
- Autosomal dominant, autosomal recessive and X-linked recessive forms exist
Gene/Gene Map
- Most sporadic cases of MAC are not due to detectable gene mutations, although with the advent of next generation sequencing and whole exome sequencing the underlying molecular basis of some are now discovered. Bilateral severe cases not due to environmental cause are more likely to have an identifiable genetic cause. Some of the more common involved genes are: autosomal dominant: SOX2, OTX2, BMP4, PAX6, CHD7, and TFAP2A; autosomal recessive: RAX, FOXE3, STRA6, and SMOC1; and X-linked: BCOR.
- Nanophthalmos and posterior microphthalmos are caused by biallelic mutations in PRSS56 or MFRP.
Epidemiology
- In the United States, the incidence of microphthalmos/clinical anophthalmos is 0.22 per 1000 births; the incidence of coloboma is 0.26 per 1000
Clinical Findings
- In microphthalmos the antero-posterior diameter of the eye in adults is less than 20 mm. In children it is often significantly less than that. Microphthalmos can be unilateral or bilateral, and may or may not be associated with uveal coloboma, hence the general classification into colobomatous and non-colobomatous categories. Complicated microphthalmos refers to the association of a small eye with other ocular abnormalities such as corneal opacification, corectopia, aniridia, cataract, persistent fetal vasculature and/or retinal dysplasia. The diagnosis of microphthalmos can generally be made by clinical inspection. The cornea is usually less than 10 mm in size but may be normal in simple microphthalmos, which is more appropriately termed nanophthalmos or posterior microphthalmos.
- True or primary anophthalmos is extremely rare and results from failure of the optic vesicle to bud out from the cerebral vesicle; the optic nerves and tracts are usually absent. Consecutive or degenerative anophthalmos results from regression or degeneration of the optic vesicle.
- Visual acuity in microphthalmos depends on the extent of retinal involvement and on coexisting ocular pathology and amblyopia. Excellent visual acuity is present in eyes with small iris or choroidal colobomas, while those with macular and optic nerve head involvement may have poor vision. Microphthalmic eyes are usually highly hypermetropic, but are sometimes highly myopic because of staphyloma formation in the area of the coloboma.
- Large colobomas may produce a white pupillary reflex, and small round choroidal colobomas may look like white tumors, leading to diagnostic confusion with retinoblastoma in rare cases. Uncommon ocular complications of microphthalmos/coloboma include angle-closure glaucoma, subretinal neovascularization, and rarely, retinal detachment.
- Colobomas of the uvea are defects in the iris, ciliary body, choroid and/or optic nerve located in the inferior or inferonasal portion of the globe along the line of closure of the embryonic fissure. A cyst may form in the area of defective closure and protrude out from the globe, leading to the microphthalmos with cyst.
- In true anophthalmos, there is complete failure of outgrowth of the primary optic vesicle. If remnants of a malformed eye are present in the orbit the condition is referred to as extreme microphthalmos or clinical anophthalmos.
- In nanophthalmos the axial length of an otherwise normal eye typically varies from 16 to 18mm. The anterior chamber is shallow; if it is grossly normal during childhood the term “posterior microphthalmos” is sometimes used. Hypermetropia is often in the range of 15 to 20D. Visual acuity can be normal early in life with proper refractive correction, but may be decreased if retinal complications occur. The lens is of normal size but is relatively large for the globe. The retinal vessels may be tortuous and engorged. Some patients have poor macular development or a horizontal papillo-macular fold and a "heaped-up" optic nerve head. Uveal effusions may develop and subside spontaneously and lead to chorioretinal pigmentary changes and choroidal thickening. Patients often develop narrow angle glaucoma with time, even though considered to have posterior microphthalmos during childhood. Many cases are autosomal recessive, related to biallelic mutations in PRS556 or MFRP.
- In microphthalmos with cyst the globe is small and there is an inferior uveo-retinal coloboma with a defect in the posterior aspect of the eye through which a cyst lined by neuroectodermally-derived tissue protrudes into the orbit and produces a bluish bulge in the inferior lid. Most cases are isolated, but familial occurrence has been reported in siblings with normal chromosomes and in monozygous twins.
- Congenital cystic eye results from failure of invagination of the optic vesicle. The cyst may also contain lens material. Some cases may represent the oculocerebrocutaneous syndrome of Delleman in which patients have intracranial cysts, agenesis of the corpus callosum and dermal appendages. 15-30% of patients with microphthalmia/coloboma have the CHARGE association (Coloboma of the uvea, congenital Heart disease, Atresia of the choanae, Retardation of growth and mental development, Genital anomalies, and Ear malformations with hearing loss). Four of the six major findings are generally required for diagnosis. Additional abnormalities include seventh cranial nerve palsy in up to 45% of patients and facial clefts. Dominant mutations in CHD7 cause CHARGE and be associated with marked variable expressivity.
Figure 1. Clinical anophthalmos: Although this child appeared to have no globe in his right orbit, MRI imaging revealed a small rudimentary eye. This is the case for most patients with clinical or apparent anophthalmos.
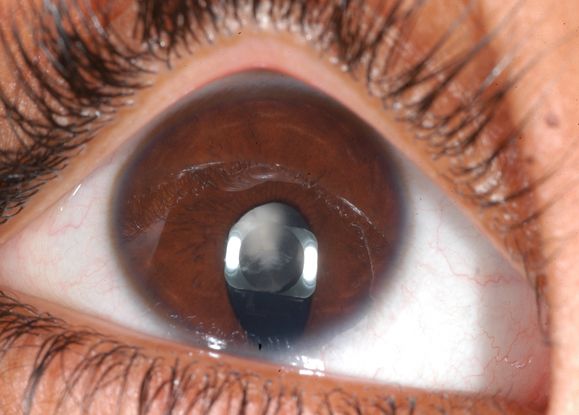
Figure 2. Complex microphthalmos: The right eye of this boy has complex microphthalmos: inferonasal iris coloboma, cataract, and chorioretinal coloboma (not shown). Note the straight edge of the lens inferonasally due to lack of zonular fibers – an abnormality sometimes termed "lens coloboma."
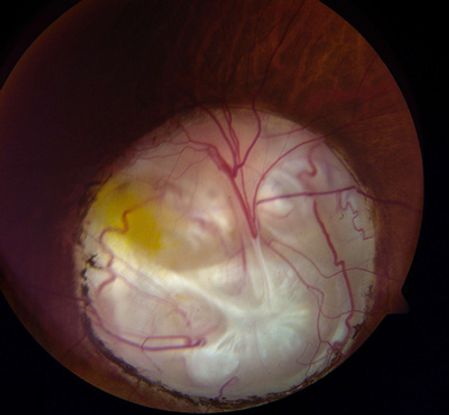
Figure 3. Coloboma of the optic nerve head: The inferonasal chorioretinal coloboma in the right eye of this boy involves the optic disc.
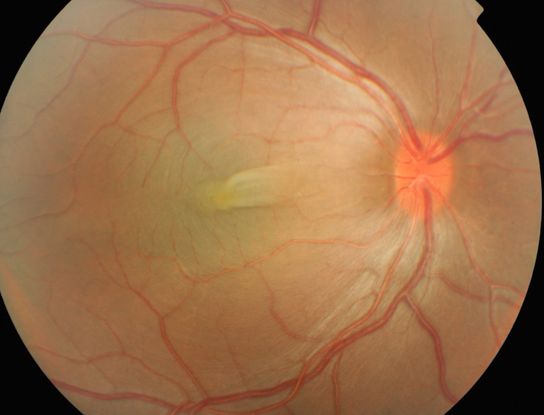
Figure 4. Nanophthalmos/posterior microphthalmos: The right eye of this boy shows the typical fundus appearance of nanopthalmos/posterior microphthalmos – a thickened, boggy retina with slight venous congestion and a horizontal retinal fold across the fovea.
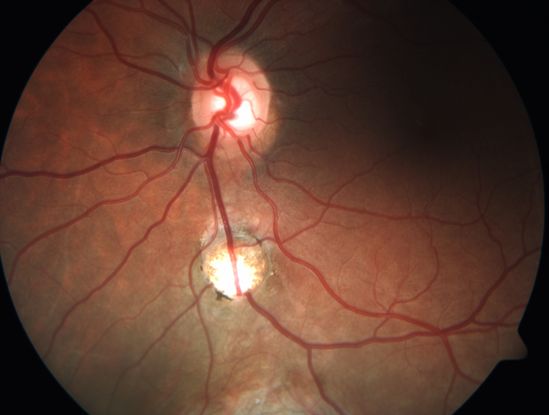
Figure 5. Inferior discrete/localized coloboma along the line of closure of the embryonic fissure
Ocular Findings
- The ocular findings in the individual subtypes are given above in the clinical findings section.
- Hornby et al. studied visual function in 113 children and young adults (196 eyes) with colobomas.11 eyes had microphthalmos with cyst, and 185 eyes had coloboma (associated with microcornea in 155 eyes and with a normal corneal diameter in 30). Microphthalmos was present in 72 of the 185 eyes with coloboma, of which 71 of 72 also had microcornea. The prognosis for vision depended on the phenotype of the better eye. They showed a good correlation of the severity of the colobomatous malformation with visual acuity, reading, and navigational vision. Microphthalmos with cyst had the worst prognosis, coloboma with microcornea and microphthalmos a poor prognosis, coloboma with only microcornea had an intermediate prognosis, and simple coloboma had the best prognosis.
- Nowilaty et al. characterized biometric findings of posterior microphthalmos. In 25 patients from 13 families, they found typical symmetry between right and left eyes and right eye mean cycloplegic refraction to be 15.09 diopters (D) (range 9.88-18.75). Right eye axial length was a mean 16.25 mm (range 14.88-19.88) and had a strong inverse correlation (Pearson coefficient -0.88, P < .0001) with corneal power (mean 48.89 D [range 41.91-52.25]).
Therapeutic Considerations
- Maumenee and Mitchell found that one-third of 82 patients had visual acuity better than 20/40 in the better eye. It is wise not to make any predictions about visual acuity based on the fundus appearance in babies. Surprisingly good vision may be possible in moderately malformed globes while poor vision may be associated with unilateral, seemingly mild cases.
- In microphthlamia with cyst, management depends on the degree of maldevelopment of the microphthalmic eye, the size of the cyst and the cosmetic and functional status of the patient. Fluid from the cyst may be aspirated or the cyst may be excised with or without preservation of the globe.
- Patients with microphthalmia should be worked up for the presence of malformations of the heart, renal or central nervous system and a systemic diagnosis should be obtained in those with multiple malformations. Huynh and co-workers described the prevalence of systemic abnormalities in a series of 99 patients with MAC. A chromosomal microarray study or whole exome sequencing are indicated in those with multiple malformations. Patients with nanophthalmos/posterior microphthalmos are at high risk for development of angle-closure glaucoma with time. In addition, they are at risk for posterior uveal effusions following ocular surgery.
|
Table 1. A Practical Classification of Microphthalmia/Coloboma
|
|
Isolated
|
- Microphthalmia
- Colobomatous
- Isolated uveoretinal coloboma
- Microphthalmia with cyst
- Non-colobomatous
|
|
Microphthalmia with Ocular Anomalies
|
- Microphthalmia with cataract
- Microphthalmia with myopia and corectopia
- Microphthalmia with ectopia lentis
- Microphthalmia with congenital retinal detachment
- Persistent Hyperplastic Primary Vitreous
- Aicardi syndrome
|
|
Microphthalmia with Mental Retardation
|
- Microphthalmia with mental retardation
- Microphthalmia with mental retardation and congenital spastic diplegia (Sjogren-Larsson)
|
|
Microphthalmia with Craniofacial Malformations
|
- Facio-Auriculo-Vertebral sequence
- Hallermann-Streiff syndrome
- Amniotic band syndrome
- Transverse facial cleft
- Microphthalmia with cleft lip/palate
- Microphthalmia with microcephaly
- Microphthalmia with microcephaly and retinal folds
- Microphthalmia with hydrocephalus and congenital retinal non-attachment (Warburg syndrome)
|
|
Microphthalmia with Malformations of the Hands and Feet
|
- Microphthalmia with polydactyly
- Waardenburg's recessive anophthalmia syndrome
|
|
Microphthalmia with Multiple Congenital Anomalies (Syndromes)
|
- CHARGE association
- Duker syndrome
- Lenz microphthalmia syndrome
- Oculo-Dento-Osseous Dysplasia
- Cryptophthalmos syndrome
- Cerebro-Oculo-Facial Syndrome
- Goltz syndrome or focal dermal hypoplasia
- Lowe syndrome
- Meckel-Gruber syndrome
- Basal cell nevus syndrome of Gorlin-Goltz
- Cross syndrome
- Microphthalmia with linear skin defects
|
|
Microphthalmia in Chromosomal Anomalies
|
- T-13 (Patau)
- 4p- (Wolf-Hirschorn)
- 18q-
- 18r
- T-18 (Edward)
- Cat-eye syndrome (marker 22)
- Other chromosomal aberrations
|
|
Microphthalmia and Intrauterine Insults
|
- Maternal drug intake: thalidomide, alcohol, isotretinoin, others
- Maternal vitamin A deficiency
- Maternal fever or radiation exposure
- Maternal uncontrolled phenylketonuria
- Intrauterine infections: CMV, EBV, Varicella, Herpes simplex, Rubella, Toxoplasmosis
|
Resources
http://www.chargesyndrome.org/
References
- Aldahmesh MA, Nowilaty SR, Alzahrani F, et al. Posterior microphthalmos as a genetically heterogeneous condition that can be allelic to nanophthalmos. Arch Ophthalmol. 2011 Jun;129(6):805-807.
- Blindness NStP (1980). Vision Problems in the U.S. New York, National Society to Prevent Blindness.
- Hornby SJ, Adolph S, Gilbert CE, Dandona L, Foster A Visual acuity in children with coloboma: clinical features and a new phenotypic classification system. Ophthalmology. 2000; 107:511-520.
- Huynh N, Blain D, Glaser T, et al. Systemic diagnostic testing in patients with apparently isolated uveal coloboma. Am J Ophthalmol. 2013; 156:1159-1168.
- Fujiki K, Nakajima A, Yasuda N, Tanabe U, Kabaswa K. Genetic analysis of microphthalmos. Ophth Paed Gen. 1982;1:139-149.
- Keppen LD, Brodsky MC, Michael JM, Poindexter AR. Hypogonadotropic hypogonadism in mentally retarded adults with microphthalmia and clinical anophthalmia. Am J Med Genet. 1990;36:285-287.
- Kohn G, El-Shawwa R, El-Rayyes E. Isolated "clinical anophthalmia" in an extensively affected Arab kindred. Clin Genet. 1988;33:321-324.
- Maumenee IH, Mitchell TN. Colobomatous malformations of the eye. Tr Am Ophthalmol Soc. 1990;88:123-132.
- Nair KS, Hmani-Aifa M, Ali Z, et al. Alteration of the serine protease PRSS56 causes angle-closure glaucoma in mice and posterior microphthalmia in humans and mice. Nat Genet. 2011 Jun;43(6):579-584.
- Nowilaty SR, Khan AO, Aldahmesh MA, Tabbara KF, Al-Amri A, Alkuraya FS. Biometric and molecular characterization of clinically diagnosed posterior microphthalmos. Am J Ophthalmol. 2013 Feb;155(2):361-372.
- Sundin OH, Leppert GS, Silva ED, et al. Extreme hyperopia is the result of null mutations in MFRP, which encodes a Frizzled-related protein. Proc Natl Acad Sci USA. 2005 Jul 5;102(27):9553-9558.
- Pagon R. Ocular coloboma. Surv Ophth. 1981;25:223.
- Russell-Eggitt IM, Blake KD, Taylor DS, Wyse RK. The eye in the CHARGE association. Br J Ophthalmol. 1990;:421-426.
- Sundin OH, Leppert GS, Silva ED, et al. Extreme hyperopia is the result of null mutations in MFRP, which encodes a Frizzled-related protein. Proc Natl Acad Sci USA. 2005 Jul 5;102(27):9553-9558.
- Strömland K, Miller M, Cook C. Ocular teratology. Surv Ophthalmol. 1991;35:429-446.
- Traboulsi EI, Lenz W, Gonzales-Ramos M, Siegel J, Macrae W, Maumenee IH. The Lenz microphthalmia syndrome. Am J Ophth. 1988;105:40-45.
- Vingolo EM, Steidl K, Forte R, et al. Autosomal dominant simple microphthalmos. J Med Genet. 1994;31:721-725.
- Warburg M. Diagnostic precision in microphthalmos and coloboma of heterogeneous origin. Ophth Paed Gen. 1981;1:37-42.
- Warburg M. Update of sporadic microphthalmos and coloboma: non-inherited anomalies. Ophth Paed Gen. 1992;13:111-122.
- Warburg M, Friedrich U. Coloboma and microphthalmos in chromosomal aberrations. Chromosomal aberrations and neural crest cell developmental field. Ophth Paed Gen. 1987;8:105-118.
- Weiss AM, Koussef BG, Ross EA, Longbottom J. Complex microphthalmos. Arch Ophthalmol. 1989;107:1619-1624.
- Weiss AH, Kousseff BG, Ross EA, Longbottom J. Simple microphthalmos. Arch Ophthalmol. 1989;107:1625-1630.
- Williamson KA, FitzPatrick DR. The genetic architecture of microphthalmia, anophthalmia and coloboma. Eur J Med Genet. 2014 Aug;57(8):369-380.
Traboulsi EI. Compendium of Inherited Disorders and the Eye. New York: Oxford University Press; 2005. Adapted with permission.