By P.G. Christakis, MD; Y.A. Reginald, MD, FRCSC
Introduction
Acquired inflammatory demyelination of the central nervous system (CNS) is rare in children, with an annual incidence of 0.9 per 100,000.1 The most common presentation of acute demyelination is optic neuritis (23%), acute disseminated encephalomyelitis (22%), and transverse myelitis (22%).1 Demyelinating optic neuritis is inflammation of the optic nerve; it may be part of a clinically isolated syndrome (CIS) or associated with a systemic demyelinating disease. CIS is an acute first episode of presumed CNS demyelination in the absence of encephalopathy (unless due to fever).2,3 Demyelination may be monofocal or multifocal and may include optic neuritis, transverse myelitis, or lesions in the brainstem, cerebellum, or cerebral hemispheres.3 However, the clinical presentation and neuroimaging must exclude demyelinating diseases such as acute disseminated encephalomyelitis, multiple sclerosis, and neuromyelitis optica to be classified as CIS.
Case Presentation
An 8-year-old previously healthy boy presents with 4 days of reduced vision in his right eye, headache, pain on extraocular movements, and malaise. Vision is no light perception (NLP) right eye, 20/20 left eye. The right pupil has a sluggish response to light, with a relative afferent pupillary defect (RAPD). Fundoscopy reveals an elevated right optic nerve with nerve fiber layer edema and peripapillary flame-shaped hemorrhages. There is moderate venous dilation, and dot-blot hemorrhages are seen in all 4 quadrants of the posterior pole. No macular edema is present. The left optic nerve is normal in appearance (Figure 1).

Figure 1. Fundus photographs
A magnetic resonance imaging (MRI) scan is performed that reveals severe thickening of the entire right optic nerve, extending to the optic tract (Figure 2). The remainder of the MRI is normal except for a small focus of enhancement in the left parietal lobe. Visual evoked potentials (VEP) are undetectable in the right eye, and normal on the left. Lumbar puncture shows normal cytology and the cerebrospinal fluid (CSF) is negative for oligoclonal banding. Routine serologic testing is normal. Given the atypical features of optic neuritis, including no light perception (NLP) vision and peripapillary hemorrhages, an inflammatory workup is performed; the result is negative. Anti-NMO IgG is tested and the result is negative.
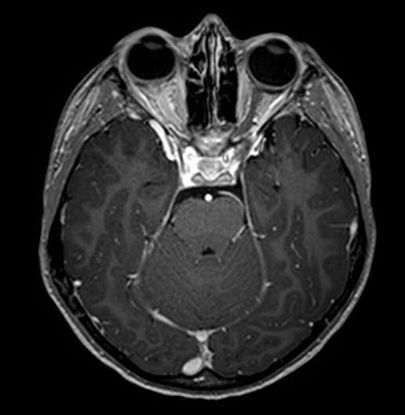
Figure 2. Magnetic resonance imaging (MRI) (T1-FTE sequence)
The patient is diagnosed with optic neuritis as a clinically isolated syndrome and started on high-dose IV methylprednisolone (30 mg/kg/day) for 5 days, followed by oral prednisone (1 mg/kg/day) tapered over 2 weeks. After 2 weeks, his vision has improved to 20/30 in his right eye, 5/6 color plates, and VEP reveals only a mildly reduced P100 peak. Goldmann visual fields show decreased central sensitivity in the right eye, corresponding to his persistent symptom of “graying” of vision (Figure 3).
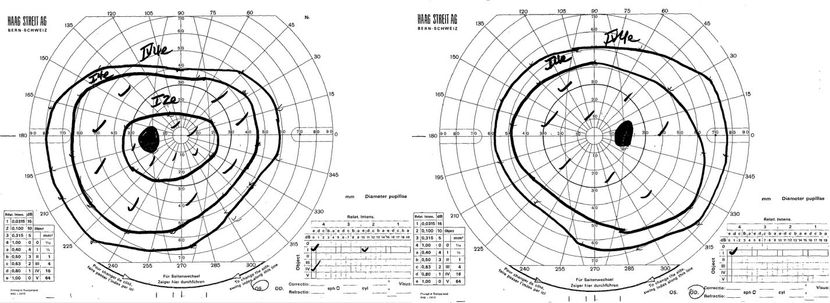
Figure 3. Goldmann visual field testing
After 4 months, his vision has recovered to 20/25 in both eyes, with a trace right RAPD. Fundoscopy reveals normal-appearing optic nerves, and optical coherence tomography (OCT) demonstrates reduced nerve fiber layer thickness in the right eye (79 μm versus 96 μm) (Figure 4).
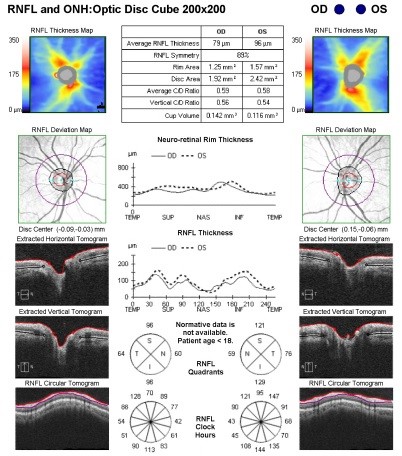
Figure 4. Optical coherence tomography
Eight months later, he presents with a 2-day history of decreased vision in his left eye and mild pain on extraocular movements. Vision is 20/30 right eye, hand motions (HM) left eye with a left RAPD. Fundoscopy reveals an elevated left optic nerve with nerve fiber layer edema and peripapillary flame-shaped hemorrhages. No macular edema is present. An MRI of the brain and spine with contrast is performed, revealing left optic nerve thickening with enhancement along the infraorbital, canalicular, and prechiasmal optic nerve. Antineuromyelitis optica (NMO) IgG testing is repeated and remains negative. The diagnosis of recurrent optic neuritis is made and he is started on high-dose intravenous methylprednisolone (30 mg/kg/day) for 3 days, followed by oral prednisone (1 mg/kg/day) with a taper over 2 weeks. Because he has difficulty sleeping and auditory hallucinations, the prednisone course is tapered early. Vision recovers to 20/25 in both eyes after 4 weeks. No new episodes of demyelination occur over the following 2 years, and serial MRIs are performed and are normal.
Optic Neuritis
Epidemiology and risk factors
Approximately 5% of cases of optic neuritis occur in the pediatric population, with an incidence of 0.2 cases per 100,000 per year.1,4 A meta-analysis of observational studies of children with a clinically isolated syndrome of optic neuritis showed the average age of onset to be 10 years, with a 1.5:1 female-to-male predilection.5 Bilateral optic neuritis was found in 49% of cases, with younger patients (<10 years old) more likely to have bilateral disease (72%) compared to adolescents (30%).5 The most common presenting complaint for pediatric optic neuritis is loss of vision (98%)6,7 and headache or pain on eye movements (37%-56%).6,8,9 Presenting vision ranged from 20/20 to no light perception, with 50%-77% of patients having severe vision loss (<20/200).6,8-10 A relative afferent pupillary defect is found in 100% of patients with unilateral disease and 47% with bilateral disease.6 Disc swelling is present in 40%-74% of cases.6-9,11-13 Fortunately, visual recovery occurs quickly (within 3 months) with 20/40 or better vision in 78%-96% of cases.6,8,9,12,14 Of children presenting with a clinically isolated syndrome, 29% converted to multiple sclerosis (MS) over the duration of the studies (mean follow-up 6.3 years).5 The risk for MS was greatest in children with demyelinating lesions on MRI at presentation (43%) compared to normal MRI (odds ratio of 28 after adjusting for age).5
Whether unilateral or bilateral presentation increases the risk of developing MS is controversial, because age is a confounding variable.5,7,14 Every 1-year increase in age of presentation has been associated with a 32% increased risk of developing MS, even after adjusting for the number of MRI lesions.5 Relapsing optic neuritis is associated with an increased risk of MS,7,10 while a recent flu-like illness may reduce the likelihood that the patient will develop MS.7 Large retrospective case series of pediatric optic neuritis at tertiary care centers diagnosed acute disseminated encephalomyelitis (ADEM) in 7%-24% of cases, MS in 15%-39% of cases, and neuromyelitis optica (NMO) in 0%-7% of cases.6-8,10,11,13,14
There are notable differences in the presentation and prognosis of optic neuritis in the pediatric and adult populations (Table 1). The Optic Neuritis Treatment Trial (ONTT) recruited 457 adults (mean age 32, 77% female) with unilateral optic neuritis (bilateral disease is rare in adults).15 The rate of ocular pain (92%) in the ONTT was higher than in the pediatric population, whereas the rate of disc swelling in the adult population was lower (35%).16 Atypical features for optic neuritis included no light perception vision, severe optic disc edema, peripapillary hemorrhages and retinal exudates, and conferred a lower risk of developing multiple sclerosis.17 Initial MRI revealed multifocal demyelination in 49% of ONTT cases, which correlated strongly with an increased risk of developing MS.18 After 15 years of follow-up, 50% of the ONTT cohort developed MS.18 Of patients with a normal initial MRI, 25% developed MS compared to 72% of patients with demyelination on initial MRI.18 This is higher than the rate of conversion to MS reported in pediatric optic neuritis, even in cohorts followed for 40 years.7
|
Table 1. Comparison of Pediatric and Adult Optic Neuritis
|
|
|
Pediatric5,6,8,9,19
|
Adult16,18
|
|
Age (mean)
|
10 years old
|
32 years old
|
|
Gender
|
Female (59%-72%)
|
Female (77%)
|
|
Laterality
|
Bilateral common (49%)
especially in younger children
|
Unilateral (>95%)
|
|
Presentation
|
Papillitis more common (40%-74%)
|
Papillitis less common (35%)
|
|
Pain less common (37%-56%)
|
Pain more common (92%)
|
|
Investigations
|
Lumbar puncture suggested to rule out infectious etiology
|
No routine investigations
|
|
Abnormal MRI (43%), associated with increased risk of MS
|
Abnormal MRI (49%), associated with increased risk of MS
|
|
Treatment
|
Paucity of evidence.
Data from adult studies applied to pediatric population.
|
High dose IV methylprednisolone then oral prednisone modestly improves time to visual recovery.
|
|
Visual Prognosis
|
Very good
|
Very good
|
|
Progression to MS
|
29% at 6-years
|
50% at 15-years
|
Diagnosis
An initial MRI may be useful in providing prognostic data to adults with a first episode of optic neuritis, or to rule out alternate etiologies when atypical features are present.18 The ONTT found the routine ordering of syphilis testing, antinuclear antibody (ANA) screen, chest radiographs, and lumbar punctures to be low yield and in no cases did these tests change clinical management.16 However, in pediatric cases of acute CNS inflammation, lumbar puncture with cerebrospinal fluid (CSF) analysis is considered essential to rule out an infectious etiology.19 In a demyelinating process, lymphocytosis is nonspecific and cannot discriminate CIS from ADEM or MS.19 However, the presence of oligoclonal bands (OCB) may alert the clinician that the child is at higher risk for developing multiple sclerosis.19 Additionally, MRI may be used to exclude disorders that may mimic acute demyelination, and also to provide prognostic information about the risk for developing MS.5 Follow-up MRI is also advocated in cases of multifocal demyelination to assess whether the lesions resolve (as in ADEM) or for the presence of new lesions (as in MS).19
Treatment
The treatment of optic neuritis is largely based on the ONTT, which demonstrated that the use of high-dose IV methylprednisolone for 3 days followed by a 2-week course of oral prednisone accelerated visual recovery in the first 15 days, but did not improve the overall visual outcome as compared to placebo.15 It advised against the use of oral steroids alone, as this may increase the chance of optic neuritis relapse.18 In the pediatric population, no large scale randomized trials for optic neuritis have been conducted. In fact, survey data has shown that parents would be unwilling to enroll their children in a randomized pediatric optic neuritis treatment trial.20 Consequently, the results of the ONTT have been applied to the pediatric population, and IV methylprednisolone (20-30 mg/kg/day, maximum 1000 mg) for 3-5 days followed by 14-21 days of oral prednisone (1 mg/kg/day) with taper has been used.21 However, even a short course of glucocorticoids in children carries risks including adrenal suppression, risk of infection, hyperglycemia, gastric ulcers, psychiatric disturbance, growth suppression, and osteonecrosis.22 Hence, the decision on whether to treat or observe pediatric optic neuritis should be made on a case-by-case basis, taking into account the child’s risk for developing a chronic demyelinating disease, possible side effects of corticosteroids, and the child’s and parents’ wishes.
Multiple Sclerosis (MS)
Pediatric MS is a chronic demyelinating disease that is defined by clinical and radiographic criteria that demonstrate dissemination of lesions in space (DIS) and time (DIT). Diagnostic criteria include any 1 of the following:2,3
- Two or more non-encephalopathic clinical CNS events of presumed inflammatory etiology separated by >30 days and involving more than 1 area of the CNS.
- One non-encephalopathic episode typical of MS with MRI findings consistent with the 2010 Revised McDonald Criteria for DIS and a follow-up MRI showing at least 1 new lesion consistent with DIT.
- ADEM followed by a nonencephalopathic clinical event >3 months later associated with MRI lesions that fulfill 2010 Revised McDonald Criteria for DIS.
- A first, acute CNS event with MRI findings that show both DIS and DIT (only if child >12 years old).
Only 3% of patients who develop MS experience their first episode prior to age 18, with a female-to-male ratio of approximately 3:1.23 Pediatric MS clinical presentations are similar to those of adults and include: optic neuritis (27%), sensory deficits (20%), motor deficits (15%), brainstem/cerebellar lesions (14%), and spinal cord lesions (13%).23 Younger patients (<12 years old) are more likely to present with an ADEM-like first episode, may have large ill-defined lesions early, and are less likely to have CSF oligoclonal bands than adolescents with MS.24,25 Initial treatment with high-dose intravenous corticosteroids is supported, but given that up to 95% of MS in childhood follows a relapsing-remitting course, there is evidence to suggest that early immunomodulatory therapy improves long-term outcomes.23,26
Neuromyelitis Optica (NMO)
Pediatric NMO is defined as optic neuritis, acute myelitis, and at least 2 of the following 3 supportive criteria:27
- Contiguous spinal cord MRI lesions extending over 3 vertebral segments.
- Brain MRI not meeting criteria for MS.
- Anti-aquaporin-4 (AQP4) IgG seropositive status.
The mean age of onset of pediatric NMO was approximately 14 years old with a female : male ratio of 3:1.28 The most common clinical presentation is optic neuritis (50%), followed by a spinal attack (42%) or opticospinal attack (8%). Similar to adults in which serum aquaporin-4 (AQP4) autoantibodies have 73% sensitivity and 91% specificity for NMO, pediatric studies have shown a sensitivity of 67%.28,29 Brain MRI in confirmed NMO-cases may reveal CNS involvement outside of the optic nerves and spinal cord, although few lesions (<25%) would have features similar to those found in MS.30,19,28 Although NMO has the poorest visual and functional prognosis of the demyelinating pediatric diseases, pediatric onset disease may be milder than the adult variant.31 Although there is a paucity of evidence for treatment of NMO, especially in the pediatric population, immunomodulatory therapies including corticosteroids, azathioprine, and rituximab have been used with some success.32
Acute Demyelinating Encephalomyelitis (ADEM)
ADEM is a multifocal, clinical CNS event of presumed demyelinating etiology with encephalopathy and abnormal MRI in the acute (<3 month) phase. Typical MRI findings include large (>1-2 cm), diffuse, poorly demarcated cerebral white matter lesions, although deep grey matter lesions (for example, thalamic or basal ganglia) may be present.3 ADEM occurs more commonly in young children (<10 years old) and is often associated with a flu-like illness.1,19,33 The occurrence of a second event beyond 3 months is defined as multiphasic ADEM, although this is rare and the majority of these cases are later diagnosed as demyelinating diseases such as multiple sclerosis or neuromyelitis optica.34,35 Although there is a lack of evidence-based management guidelines, treatment with high-dose IV methylprednisolone has been associated with symptomatic recovery and resolution of MRI lesions.34
References
- Banwell B, Kennedy J, Sadovnick D, et al. Incidence of acquired demyelination of the CNS in Canadian children. Neurology. 2009;72(3):232-239.
- Krupp LB, Banwell B, Tenembaum S. Consensus definitions proposed for pediatric multiple sclerosis and related disorders. Neurology. 2007;68(16 Suppl 2):S7-S12.
- Krupp LB, Tardieu M, Amato MP, et al. International Pediatric Multiple Sclerosis Study Group criteria for pediatric multiple sclerosis and immune-mediated central nervous system demyelinating disorders: revisions to the 2007 definitions. Mult Scler. 2013;19(10):1261-1267.
- Rodriguez M, Siva A, Cross SA, O'Brien PC, Kurland LT. Optic neuritis: a population-based study in Olmsted County, Minnesota. Neurology. 1995;45(2):244-250.
- Waldman AT, Stull LB, Galetta SL, Balcer LJ, Liu GT. Pediatric optic neuritis and risk of multiple sclerosis: meta-analysis of observational studies. J AAPOS. 2011;15(5):441-446.
- Wan MJ, Adebona O, Benson LA, Gorman MP, Heidary G. Visual outcomes in pediatric optic neuritis. Am J Ophthalmol. 2014;158(3):503-507 e2.
- Lucchinetti CF, Kiers L, O'Duffy A, et al. Risk factors for developing multiple sclerosis after childhood optic neuritis. Neurology. 1997;49(5):1413-1418.
- Wilejto M, Shroff M, Buncic JR, Kennedy J, Goia C, Banwell B. The clinical features, MRI findings, and outcome of optic neuritis in children. Neurology. 2006;67(2):258-262.
- Lana-Peixoto MA, Andrade GC. The clinical profile of childhood optic neuritis. Arq Neuropsiquiatr. 2001;59(2-B):311-317.
- Absoud M, Cummins C, Desai N, et al. Childhood optic neuritis clinical features and outcome. Arch Dis Child. 2011;96(9):860-862.
- Alper G, Wang L. Demyelinating optic neuritis in children. J Child Neurol. 2009;24(1):45-48.
- Bonhomme GR, Waldman AT, Balcer LJ, et al. Pediatric optic neuritis: brain MRI abnormalities and risk of multiple sclerosis. Neurology. 2009;72(10):881-885.
- Mizota A, Niimura M, Adachi-Usami E. Clinical characteristics of Japanese children with optic neuritis. Pediatr Neurol. 2004;31(1):42-45.
- Brady KM, Brar AS, Lee AG, Coats DK, Paysse EA, Steinkuller PG. Optic neuritis in children: clinical features and visual outcome. J AAPOS. 1999;3(2):98-103.
- Beck RW, Cleary PA, Anderson MM, Jr., et al. A randomized, controlled trial of corticosteroids in the treatment of acute optic neuritis. The Optic Neuritis Study Group. N Engl J Med. 1992;326(9):581-588.
- Optic Neuritis Study Group. The clinical profile of optic neuritis. Experience of the Optic Neuritis Treatment Trial. Arch Ophthalmol. 1991;109(12):1673-1678.
- Beck RW, Trobe JD, Moke PS, et al. High- and low-risk profiles for the development of multiple sclerosis within 10 years after optic neuritis: experience of the optic neuritis treatment trial. Arch Ophthalmol. 2003;121(7):944-949.
- Optic Neuritis Study Group. Multiple sclerosis risk after optic neuritis: final optic neuritis treatment trial follow-up. Arch Neurol. 2008;65(6):727-732.
- Dale RC, Brilot F, Banwell B. Pediatric central nervous system inflammatory demyelination: acute disseminated encephalomyelitis, clinically isolated syndromes, neuromyelitis optica, and multiple sclerosis. Curr Opin Neurol. 2009;22(3):233-240.
- Waldman AT, Shumski MJ, Jerrehian M, Liu GT. Parent and medical professional willingness to enroll children in a hypothetical pediatric optic neuritis treatment trial. Front Neurol. 2011;2:75.
- Pohl D, Waubant E, Banwell B, et al. Treatment of pediatric multiple sclerosis and variants. Neurology. 2007;68(16 Suppl 2):S54-S65.
- Liu D, Ahmet A, Ward L, et al. A practical guide to the monitoring and management of the complications of systemic corticosteroid therapy. Allergy Asthma Clin Immunol. 2013;9(1):30.
- Chitnis T, Glanz B, Jaffin S, Healy B. Demographics of pediatric-onset multiple sclerosis in an MS center population from the Northeastern United States. Mult Scler. 2009;15(5):627-631.
- Chabas D, Ness J, Belman A, et al. Younger children with MS have a distinct CSF inflammatory profile at disease onset. Neurology. 2010;74(5):399-405.
- Polman CH, Reingold SC, Banwell B, et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol. 2011;69(2):292-302.
- Ghezzi A, Banwell B, Boyko A, et al. The management of multiple sclerosis in children: a European view. Mult Scler. 2010;16(10):1258-1267.
- Wingerchuk DM, Lennon VA, Pittock SJ, Lucchinetti CF, Weinshenker BG. Revised diagnostic criteria for neuromyelitis optica. Neurology. 2006;66(10):1485-1489.
- Collongues N, Marignier R, Zééphir H, et al. Long-term follow-up of neuromyelitis optica with a pediatric onset. Neurology. 2010;75(12):1084-1088.
- Lennon VA, Wingerchuk DM, Kryzer TJ, et al. A serum autoantibody marker of neuromyelitis optica: distinction from multiple sclerosis. Lancet. 2004;364(9451):2106-2112.
- Pittock SJ, Lennon VA, Krecke K, Wingerchuk DM, Lucchinetti CF, Weinshenker BG. Brain abnormalities in neuromyelitis optica. Arch Neurol. 2006;63(3):390-396.
- Absoud M, Lim MJ, Appleton R, et al. Paediatric neuromyelitis optica: clinical, MRI of the brain and prognostic features. J Neurol Neurosurg Psychiatry. 2015;86(4):470-472.
- Longoni G, Banwell B, Filippi M, Yeh EA. Rituximab as a first-line preventive treatment in pediatric NMOSDs: Preliminary results in 5 children. Neurol Neuroimmunol Neuroinflamm. 2014;1(4):e46.
- Banwell B, Ghezzi A, Bar-Or A, Mikaeloff Y, Tardieu M. Multiple sclerosis in children: clinical diagnosis, therapeutic strategies, and future directions. Lancet Neurol. 2007;6(10):887-902.
- Tenembaum S, Chamoles N, Fejerman N. Acute disseminated encephalomyelitis: a long-term follow-up study of 84 pediatric patients. Neurology. 2002;59(8):1224-1231.
- Neuteboom RF, Boon M, Catsman Berrevoets CE, et al. Prognostic factors after a first attack of inflammatory CNS demyelination in children. Neurology. 2008;71(13):967-973