This extension of the DRCR.net Protocol I trial further demonstrates that prompt treatment with ranibizumab is the superior therapeutic modality for diabetic macular edema (DME) over laser with or without intravitreal triamcinolone.
After 5 years of follow-up, patients initially randomized to ranibizumab with prompt or deferred laser showed better mean VA and greater improvement from baseline VA compared with eyes managed initially with laser or combined triamcinolone and laser, even after these eyes received deferred ranibizumab treatment.
Study Design and Treatment Protocol
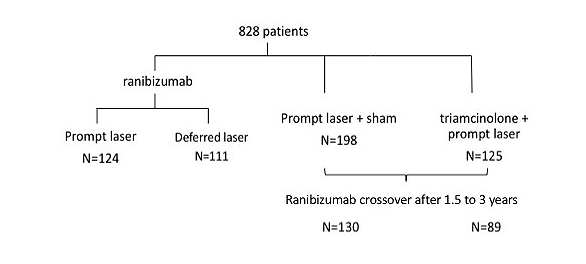
After the one-year outcomes of the study showed superior outcomes in patients randomized to ranibizumab, investigators were given the option to initiate deferred ranibizumab treatment in patients initially randomized to the sham injection + prompt laser or triamcinolone + prompt laser treatment arms, based on their OCT and visual outcomes. Deferred ranibizumab treatment commenced 1.5 to 3 years after enrollment. The decision to initiate differed ranibizumab treatment was at the discretion of the treating physician, after which they followed the same retreatment guidelines in the original ranibizumab group.
During the 5-year period, the median number of ranibizumab injections was 8 in the laser/crossover group, 9 in the triamcinolone + laser/crossover group and 17 in the ranibizumab + deferred laser group. Across the prompt laser groups, eyes received an average of 3–4 laser photocoagulation treatments, principally in the first 3 years before the deferred ranibizumab groups were fully crossed over. Only 15% of eyes or fewer received additional treatments up to year 5, and 56% of ranibizumab + deferred laser eyes never received a single laser treatment, as the presiding physician determined it was not necessary. In addition, very few eyes initially randomized to receive intravitreal triamcinolone required further injections after 3 years.
Outcomes
At year 5, after adjusting for baseline VA, eyes treated with ranibizumab + deferred laser gained an average 4.4 more EDTRS letters than those treated with sham + laser/crossover ranibizumab (P=0.001), and 2.8 more letters than those treated with triamcinolone + laser/crossover ranibizumab (P=0.067). All eyes displayed significant reductions in central subfield thickness, though the laser/crossover group had the thinnest average value (P=0.009). Other differences among the groups were not significant.
Patients in the crossover groups showed rapid improvements following ranibizumab initiation. Within 24 weeks and a median of 5 injections, the laser and triamcinolone + laser/crossover groups gained an average of 4 and 3 letters, respectively. After only 1 injection, both groups showed a substantial reduction in subfield thickness of approximately 60 µm.
Discussion
These findings are consistent with those of RISE/RIDE and add to the evidence that prompt anti-VEGF therapy leads to improved visual outcomes in patients with DME, and that the results may sustain over time. Although all groups achieved significant anatomical improvement by the end of follow-up, those that waited for ranibizumab never reached the visual gains realized by eyes initially treated with ranibizumab.