Optic disc change is frequently associated with Vogt-Koyanagi-Harada disease (VKH) but not studied in great detail. While optic disc change may hamper the correct diagnosis of VKH, the presence of VKH may also hinder the diagnosis of optic disc disease. This article explores how greater knowledge of optic disc changes seen in patients with VKH may improve the diagnosis and treatment of patients with the disease.
VKH is a multisystem autoimmune disorder principally affecting pigmented tissues in the ocular, auditory, integumentary and central nervous systems. Typically, the eye is the most involved organ. VKH mainly affects darkly pigmented races, such as East and Southeastern Asians, Asian Indians, Middle Easterners, Hispanics and Native Americans. In contrast, it is uncommon in Caucasians and Africans, indicating that pigmentation is not the only etiologic factor in its pathogenesis. For example, HLA-DR4 and Dw53 are strongly associated with VKH in Japanese people. This suggests that genetic factors play a role in the development of VKH.1,2
Patients with VKH usually have bilateral granulomatous panuveitis associated with poliosis, vitiligo, alopecia, dysacousia and signs of meningeal irritation. Based on revised diagnostic criteria, patients are diagnosed with complete VKH, incomplete VKH or probable VKH,3 although the majority are classified as incomplete or probable.
Patients with VKH are typically treated with high-dose systemic corticosteroids, and their prognosis is fair. Although optic disc swelling and hyperemia are common findings, visual field loss from optic disc involvement occurs more rarely, and its visual prognosis is not necessarily good. Considering the generally fair prognosis of VKH, optic disc involvement should be taken more seriously.
Clinical features of VKH
VKH can be categorized into four phases: prodromal, uveitic, convalescent and recurrent. The first phase, prodromal, usually lasts for only a few days. Headache, nausea, fever, meningismus and photophobia are typical signs, with optic disc swelling seen rarely. During the second or uveitic phase, visual disturbance occurs bilaterally involving thickening of choroid and multiple serous retinal detachments (see Figure 1A). Inflammation eventually becomes diffuse and extends to the anterior segment. Optic disc swelling or hyperemia is seen. This phase usually lasts for several weeks before the transition to the convalescent phase, which is characterized by depigmentation of the choroid several months later and an orange-red discoloration of the fundus known as "sunset glow fundus." The optic disc sometimes appears pale.
While most cases of VKH can be diagnosed by clinical examination, fluorescein angiography (FA) can be useful for diagnosis (see Figure 1B), as can lumbar puncture.1,2
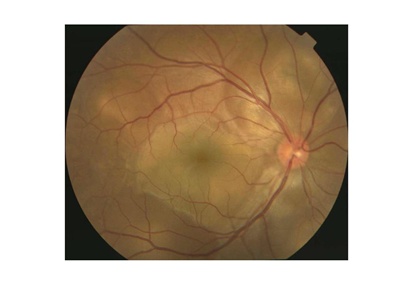
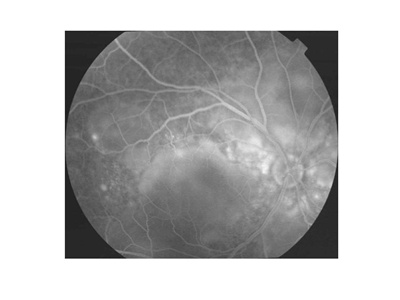
Figures 1A and 1B. Typical fundus photograph of a patient with Vogt-Koyanagi-Harada disease. This optic disc is slightly hyperemic, with multiple exudative retinal detachments visible (A). Fluorescein angiography reveals multifocal dye leakage and staining of the optic disc (B).
Optic disc involvement
Optic disc hyperemia or swelling is a common finding in the prodromal and uveitic phases of VKH. Optic nerve staining by FA is one of the major findings of VKH. However, studies have found that among Hispanic VKH patients, only 9 percent of eyes showed optic nerve swelling in the acute phase4 compared with 39 percent of Japanese patients and a much higher incidence of 71 percent among elderly patients.5
The mechanism causing optic disc hyperemia/swelling is unclear. However, mechanical compression of the optic disc by inflammation of the choroid and/or disc may play a major role in this change. In most cases, optic disc change resolves without sequelae, although a pale optic disc is sometimes seen in the convalescent phase. Researchers have reported several noteworthy VKH disease cases in which optic disc changes were seen.
Optic disc change may mask VKH
Optic neuritis may appear as the initial symptom of VKH in some patients, which can make diagnosis of VKH difficult. One article reported the cases of three Hispanic patients with sudden onset of moderate to severe bilateral visual disturbance preceded by extraocular symptoms, such as headache, cold or rhinorrhea.6 Fundus examination revealed swelling of the bilateral optic disc. However, the initial work-up, including CT or MRI, was unremarkable. A post-viral inflammatory type of optic neuritis was suspected. Subsequently, multifocal serous retinal detachments became evident, and FA revealed optic disc leakage and multiple foci of hyperfluorescent spots. Ultrasound examination showed choroid thickening. A diagnosis of VKH disease was subsequently made, and oral corticosteroid therapy was started. After several months, extraocular symptoms resolved and visual acuity improved.
In these cases, the initial diagnosis was bilateral optic neuritis. The differential diagnosis included Devic disease, immune-mediated optic neuropathy, nutritional amblyopia and Leber's hereditary optic neuropathy. It would not be difficult to correctly diagnosis an Asian or Hispanic patient presenting with the complete form of VKH, although it might not be easy to reach the correct diagnosis for the incomplete form.
VKH should be considered in differential diagnoses of cases with bilateral optic disc swelling, even in the absence of extraocular manifestations. This is particularly important when a neurological imaging examination does not show an abnormality and cerebrospinal fluid pleocytosis is present. In such cases, FA, indocyanine green angiography and ultrasonography are useful for making the diagnosis.
VKH may mask optic disc disease
Although optic disc hyperemia/swelling is common in VKH disease, visual field loss from optic disc involvement is uncommon. In our series of 52 consecutive Japanese patients with VKH disease, 15 patients showed bilateral optic disc swelling.7 Among them, six developed irreversible visual field defects, a much higher number than expected. While no permanent visual field defects could be demonstrated in the patients without optic disc swelling, all six patients with optic disc swelling noticed acute bilateral visual loss. They all had bilateral panuveitis associated with meningismus and/or cerebrospinal pleocytosis. Additionally, four of them had peripapillary hemorrhage (see Figure 2). FA showed multifocal leakage in the posterior pole in these patients and both filling delay and late leakage of the optic disc (see Figure 3). A Humphrey visual field analysis revealed various degrees of visual field defects (see Figure 4). The critical flicker frequency was abnormal.
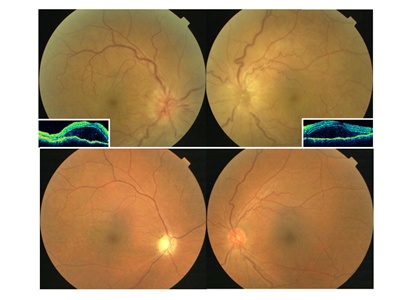
Figure 2. Ocular fundus of a Vogt-Koyanagi-Harada disease patient with associated bilateral anterior ischemic optic neuropathy. Bilateral optic disc swelling associated with hemorrhage and exudative serous retinal detachment in the acute phase (top). Optical coherence tomography reveals serous retinal detachment (insets). After corticosteroid treatment, these changes subsided, and a pale optic disc is seen in the right eye in the convalescent phase (bottom).
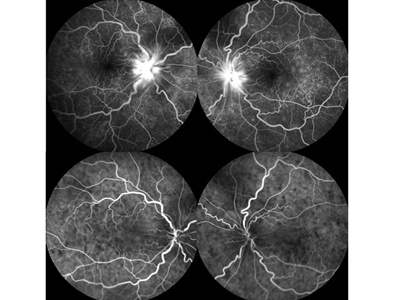
Figure 3. Fluorescein and indocyanine green angiography of a Vogt-Koyanagi-Harada disease patient with associated with bilateral anterior ischemic optic neuropathy in the acute phase. Fluorescein angiography shows leakage of dye from the optic disc or choroid (top). Indocyanine green angiography shows multiple hypofluorescent spots in the posterior fundus (bottom).
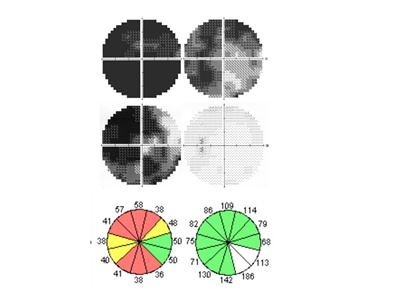
Figure 4. Visual fields and retinal nerve fiber layer thickness of a Vogt-Koyanagi-Harada disease patient with associated with anterior ischemic optic neuropathy. Humphrey 30-2 visual fields in the acute phase (top) and convalescent phase (middle). Visual fields defects were evident in the acute phase. Even after high corticosteroid therapy, some visual field defects remained during the convalescent phase (middle). Retinal nerve fiber layer thickness analysis showed the decrease corresponding to the visual field defects (bottom).
These patients were treated with high-dose corticosteroid therapy. Several weeks later both uveal inflammation and optic disc swelling disappeared. The visual fields showed some improvement as retinal detachments and disc swelling resolved, but the visual field loss remained. Subsequently, one optic disc developed pallor. Retinal nerve fiber layer (RNFL) thickness measured by optical coherence tomography decreased (see Figure 4), but there was no choroidal atrophy. These findings suggest that anterior ischemic optic neuropathy (AION) developed almost simultaneously with VKH.
In the literature, there are several cases of AION and VKH developing simultaneously.7,8 It has been reported that patients with Posner-Schlossman syndrome, Behcet's disease, HLA-B27-associated anterior uveitis or birdshot chorioretinopathy developed AION, indicating that uveitis may play a role in its development.
The mechanism whereby AION develops in VKH disease is also uncertain. The arterial supply to the optic disc is derived from the ciliary circulation. The region of the lamina cribrosa is supplied by centripetal fine branches from the so-called arterial circle of Zinn or more often directly from the branches of the posterior ciliary arteries. The prelaminar region of the optic disc is supplied mainly by centripetal branches from the peripapillary choroidal vessels.
Histopathological study of sympathetic ophthalmia, the pathological identical twin of VKH, shows severe uveitis at the juxtapapillary choroid. There is also obliteration of the choriocapillaris and inflammatory infiltration surrounding the emissary vessels and nerves through the sclera canals, which are associated with the severity of choroidal involvement. Severe inflammation at the juxtapapillary choroid may lead to interference of the posterior ciliary artery supply to the optic nerve head and the retrolaminar part of the optic nerve, resulting in AION development.
AION eyes show a variety of optic nerve-related visual field defects. The range of these defects varies depending on the involvement of the branches of the posterior ciliary artery. If the visual field defects caused by AION are small, they may be overlooked when there are larger field defects caused by exudative retinal detachment.
However, it should be noted that AION is associated with the acute phase of VKH. Optic disc swelling in VKH may therefore be a sign of complications of AION, particularly in elderly patients with diabetes mellitus and small optic discs with either absent or small cups.
Figures 2, 3 and 4 reprinted with permission from Springer from Nakao K, Mizushima Y, Abematsu N, Goh N, Sakamoto T. Anterior ischemic optic neuropathy associated with Vogt-Koyanagi-Harada disease. Graefes Arch Clin Exp Ophthalmol. 2009;247(10):1417-1425.
References
- Moorthy RS, Inomata H, Rao NA. Vogt-Koyanagi-Harada syndrome. Surv Ophthalmol. 1995;39(4):265-292.
- Damico FM, Kiss S, Young LH. Vogt-Koyanagi-Harada disease. Semin Ophthalmol. 2005;20(3):183-190.
- Read RW, Holland GN, Rao NA, et al. Revised diagnostic criteria for Vogt-Koyanagi-Harada disease: report of an international committee on nomenclature. Am J Ophthalmol. 2001;131(5):647-652.
- Sukavatcharin S, Tsai JH, Rao NA. Vogt-Koyanagi-Harada disease in Hispanic patients. Int Ophthalmol. 2007;27(2-3):143-148.
- Kiyomoto C, Imaizumi M, Kimoto K, Abe H, Nakano S, Nakatsuka K. Vogt-Koyanagi-Harada disease in elderly Japanese patients. Int Ophthalmol. 2007 ;27(2-3):149-153.
- Rajendram R, Evans M, Khurana RN, Tsai JH, Rao NA. Vogt-Koyanagi-Harada disease presenting as optic neuritis. Int Ophthalmol. 2007;27(2-3):217-220.
- Nakao K, Mizushima Y, Abematsu N, Goh N, Sakamoto T. Anterior ischemic optic neuropathy associated with Vogt-Koyanagi-Harada disease. Graefes Arch Clin Exp Ophthalmol. 2009;247(10):1417-1425.
- Yokoyama A, Ohta K, Kojima H, Yoshimura N. Vogt-Koyanagi-Harada disease masquerading anterior ischaemic optic neuropathy. Br J Ophthalmol. 1999;83(1):123.