The links to each individual chapter of the Color Atlas of Gonioscopy are available at the Chapters link, below.
Primary Angle Closure
Pupillary block
Pupillary block is the result of abnormal contact between the iris and the lens that prohibits the free flow of aqueous from the posterior chamber to the anterior chamber. Aqueous trapped in the posterior chamber pushes the iris forward (8‑1), giving it a convex appearance, which is termed “iris bombé” (8‑2). The outflow of aqueous is impaired when the iris is pushed on to the trabecular meshwork. Hyperopic eyes are at highest risk of angle closure. Such eyes have shallow anterior chambers and small corneas (Tornquist, 1956, 1957). Angle closure is uncommon in myopic eyes, although it is seen in myopic eyes with spherophakia, plateau iris, and retinopathy of prematurity. If a myopic patient presents with narrow angles, one should consider looking for a specific cause. The elderly are more susceptible because the growth of their lenses moves the lens-iris diaphragm forward. Angle closure is especially prevalent among Asians. Pupillary block gives rise to three forms of primary angle closure: acute angle closure, intermittent angle closure, and chronic angle closure.
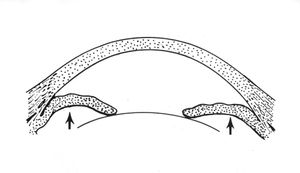
8-1 Sketch of pupillary block. A relative seal between iris and lens traps aqueous in the posterior chamber. The aqueous pushes the peripheral iris forward, causing iris bombé and eventual angle closure.
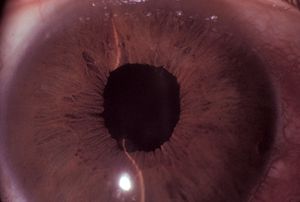
8-2 Pupillary block with iris bombé. Note that the midperipheral iris is bowed forward and that the central anterior chamber is relatively deep.
Acute angle-closure glaucoma is the most dramatic. On the basis of symptoms it was classified as acute congestive glaucoma until the late 1930s, when Barkan was able to classify it by its mechanism (Barkan, 1938). Patients experience sudden, severe pain with blurred vision and may have nausea and vomiting. Attacks generally occur in dark environments or during periods of intense emotion. Pupillary block is most likely to occur in middilation, when there is greatest lens-iris contact. A patient who does not develop pupillary block when the pupil is fully dilated in the physician’s office may develop an attack later in the day when the pupil returns to middilation.
In acute angle-closure glaucoma, the affected eye is dramatically injected, showing corneal edema and a shallow anterior chamber (8‑3). It may be difficult to examine the angle during an acute attack due to corneal haze (8‑4). On gonioscopic examination, the iris bows forward and obscures the view of angle structures (8‑5).
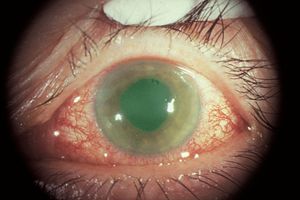
8-3 Acute angle-closure glaucoma with marked injection and a steamy cornea.

8-4 Gonioscopic view after an attack of acute angle closure. The cornea is edematous, which limits visualization. The trabecular meshwork has a ruddy appearance.
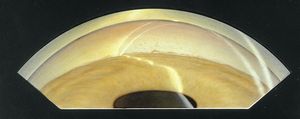
8-5 Iris bombé. No trabecular structures are visible. Note that the inner and outer lines of the corneal wedge do not meet in the anterior chamber, meaning that Schwalbe’s line and the trabecular meshwork are hidden by the iris.
Indentation gonioscopy is difficult during an acute attack owing to the high intraocular pressure. The other eye should be examined; in most cases of primary angle closure the contralateral eye will also have a markedly narrowed angle. Although asymmetric angles are unusual, they can be seen in eyes with unilateral mature cataracts, anisometropia, or trauma. If the attack of angle closure breaks spontaneously, there may be an anterior chamber cellular reaction and aqueous hyposecretion. The patient may be diagnosed erroneously as having iritis. This may be seen more frequently in patients classified as having intermittent angle closure. Eyes that have undergone previous episodes of angle closure may show evidence on slit-lamp examination of damage from high pressure, such as atrophy of the iris (8‑6), cataract (8‑6), or spiral changes of the radial iris fibers (8‑7). Very high pressures can also cause necrosis of the lens epithelium, which appears as opaque areas (glaukomflecken) beneath the anterior lens capsule (8‑8).
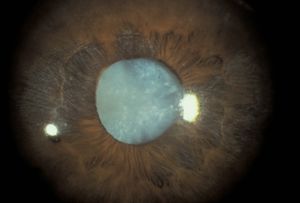
8-6 Dense cataract and patchy iris atrophy in an eye after an attack of acute angle-closure glaucoma.
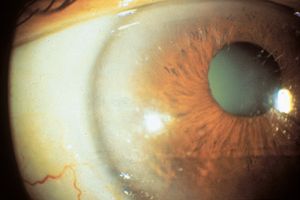
8-7 Eye after an attack of acute angle closure. Note that some of the iris fibers now take a spiral, rather than radial, course.
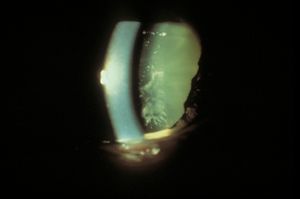
8-8 Glaukomflecken under the anterior lens capsule after an attack of acute angle closure. These lens changes are caused by necrosis of the lens epithelium.
Intermittent (subacute) angle closure presents less dramatically. Patients may be asymptomatic or report episodic blurred vision, colored haloes around lights, and headache. Because intermittent headache may be the only symptom, many of these patients are misdiagnosed as migraineurs. If left untreated, intermittent angle closure can progress to chronic angle closure.
Chronic angle closure results in an insidious rise in intraocular pressure without symptoms. In both conditions gonioscopy will reveal iris bombé with very narrow angles (see 8‑5). Trabecular structures may be visible only with indentation (Chapter 4). Synechiae may be present—especially superiorly, where the angle is narrowest. There may be a dusting of pigment on the cornea from contact with the iris, which might cause one to mistake it for the pigmented trabecular meshwork (8‑9; see also 4‑20). Indentation gonioscopy and the corneal wedge are most helpful in determining the true location of the trabecular meshwork. Changes in the optic nerve and visual field may be noted, especially in chronic angle closure.
The treatment of angle closure due to pupillary block is surgical iridectomy or laser iridotomy. Medical management using aqueous suppressants is used to temporize and to clear the cornea sufficiently to permit laser iridotomy. Once an iridotomy (or surgical iridectomy) has been performed in an eye with narrow angles, the anterior chamber should be noticeably deeper (8‑10). Although they deepen, the angles do not usually attain a normal depth. After iridotomy, it is unusual to see grade 3 or 4 angles (Shaffer scale), or to see beyond the scleral spur. If angle closure persists after such treatment, the possibility of synechial angle closure or plateau iris syndrome should be considered. Both are discussed later in this chapter.
It is important to recognize that markedly elevated intraocular pressure is not always associated with angle closure. One should perform gonioscopic examination first and should not proceed directly to iridectomy or iridotomy. Other processes, such as glaucomatocyclitic crisis, can cause acute and marked pressure elevations. It is also not safe to assume that a marked pressure rise after dilation is due to angle closure. Patients with open angles may show pressure spikes after dilation. These spikes can be quite high, especially in patients with open-angle glaucoma who are taking cholinergic drops (Shaw and Lewis, 1986).
Video Clips
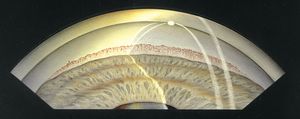
8-9 Pigment accumulation on the corneal endothelium could be mistaken for trabecular meshwork in this eye with synechial angle closure. The corneal wedge shows that Schwalbe’s line and the trabecular meshwork are hidden by the iris.
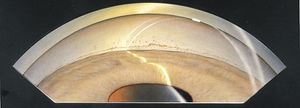
8-10 Gonioscopic view of an eye with angle closure following surgical iridectomy. This is the same eye as in 8‑5. There are extensive synechiae and only the most anterior portion of the trabecular meshwork is seen in some areas with the slit-lamp beam.
Plateau iris
Plateau iris configuration refers to a condition in which the iris exhibits a flat approach to the angle. The iris appears to be on a plane with Schwalbe’s line or the anterior trabecular meshwork and has a prominent last roll that blocks the view of the angle, especially on dilation. As the angle is approached, the iris drops off sharply to insert just below the scleral spur (8‑11 and 8‑12). The central anterior chamber is relatively deep, especially in comparison to patients with a convex iris associated with pupillary block. Patients with plateau iris configuration can also develop pupillary block, which can be treated with peripheral iridotomy. If the angle remains compromised, the patient would be diagnosed as having plateau iris syndrome.
Plateau iris syndrome is an unusual form of primary angle closure that is not caused by pupillary block. The angle is closed by the prominent last roll of the iris and the abnormal approach of the iris to the angle. A patent peripheral iridectomy or iridotomy must be present for a diagnosis of plateau iris syndrome. In such eyes, the ciliary processes are abnormally far forward. After iridotomy, the iris is held forward by the ciliary body (Pavlin et al, 1992). On indentation, the central iris is pushed back but the peripheral iris is held up by the ciliary processes (8‑13 and 8‑14). Anterior segment imaging techniques are also helpful in supporting the diagnosis of plateau iris.
Video Clip: Gonioscopy: Plateau Iris
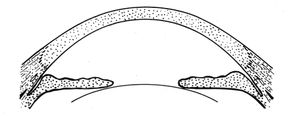
8-11 Sketch of plateau iris configuration. There is a relatively deep central chamber. The iris takes a flat approach towards Schwalbe’s line. There is a prominent last roll of the iris before a steep drop-off into the chamber angle.
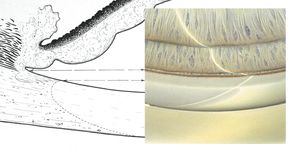
8-12 Gonioscopic view of an eye with a plateau iris configuration. Note the prominent peripheral rolls of the iris and the steep drop-off into the chamber angle.
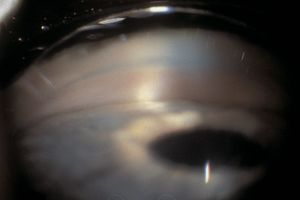
8-13 Narrow angle in a patient with a patent laser iridotomy. Plateau iris configuration.
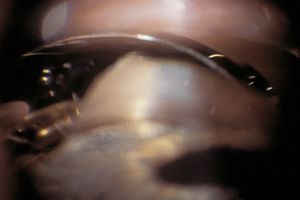
8-14 Same eye as shown in 8‑13. When indented, the middle portion of the iris moves back, but the iris over the lens and periphery stays forward. The peripheral iris is held forward by an abnormally anterior ciliary body (arrow).
Aqueous misdirection (ciliary block, malignant glaucoma)
Aqueous misdirection is an uncommon form of glaucoma in which aqueous is misdirected into the vitreous cavity, causing the lens and iris to be pushed forward (8‑15). The central anterior chamber tends to be very shallow or flat (8‑16). Gonioscopy is difficult to perform in aqueous misdirection because of the extreme shallowing of the anterior segment, but an example is shown in figure 8‑17. The mechanism of aqueous misdirection is uncertain. It may be that apposition of the ciliary body to the lens or vitreous body creates a seal that directs aqueous humor posteriorly (Weiss and Shaffer, 1972). The involvement of the vitreous body in aqueous misdirection is supported by the fact that definitive treatment involves breaking through the anterior vitreous face with either vitrectomy or Nd:YAG laser. Annular swelling of the choroid may represent a concurrent or alternate mechanism of aqueous misdirection.
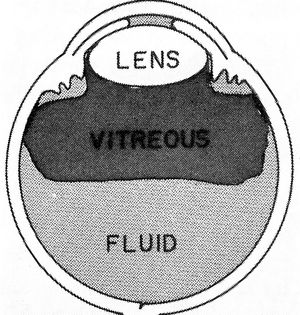
8-15 Mechanism of aqueous misdirection. Aqueous is diverted into the vitreous cavity and causes vitreous, lens, and iris to be pushed forward, collapsing the anterior chamber. (Reprinted with permission from the BMJ Publishing Group. Simmons RJ. Malignant glaucoma. British Journal of Ophthalmology [1972;56:263–272].)
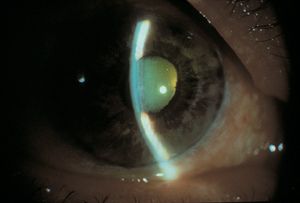
8-16 An eye with aqueous misdirection following a trabeculectomy. Note that the entire chamber is flat, with lenscornea contact. This should be contrasted with 8‑2, which shows a flat peripheral chamber but a relatively deeper central chamber in iris bombé.

8-17 Gonioscopy of a patient with aqueous misdirection. Because of the narrow angle and dilated pupil, the ciliary processes are very easily seen.
Aqueous misdirection occurs most frequently after intraocular surgery, particularly after filtration surgery in patients with narrow angles. It can occur at an early postoperative stage or later when anticholinergics are discontinued or cholinergics are begun. Aqueous misdirection must be differentiated from other causes of flat chamber with high intraocular pressure. Pupillary block can be difficult to distinguish from aqueous misdirection. In the former the central chamber is usually deeper than the peripheral chamber (see 8‑2), whereas in aqueous misdirection the central chamber is very shallow or flat (see 8‑16). If there is a patent iridectomy or iridotomy, pupillary block can be ruled out; if there is no iridectomy or iridotomy, one should be performed prior to a diagnosis of aqueous misdirection. Suprachoroidal hemorrhage can present with a shallow chamber and high pressure after surgery. Patients with suprachoroidal hemorrhage often give a history of abrupt-onset, severe pain. The choroidal elevation can be seen with the ophthalmoscope or by echography.
Secondary Angle Closure
Secondary angle closure can occur with or without pupillary block. In eyes without pupillary block, the iris can be pushed over the trabecular meshwork by a mass or swelling in or behind the iris. The iris can also be pulled over the trabecular meshwork by inflammation, neovascularization, or membranes in the anterior chamber. In eyes with secondary angle closure but without pupillary block, peripheral iridectomy or iridotomy is of no benefit and should not be performed.
Secondary pupillary block
Pupillary block can develop secondarily if the iris becomes adherent to the lens or vitreous body or if lens or vitreous becomes trapped within the pupil. Peripheral iridectomy or iridotomy can relieve angle closure in secondary pupillary block.
Central posterior synechiae are inflammatory adhesions of the iris to the anterior lens capsule or face of the vitreous body. If synechiae develop over 360°, the pupil becomes secluded. Aqueous is trapped in the posterior chamber, driving the iris forward (8‑18 and 8‑19). Dilation can break the synechiae if they are of recent onset. Iridotomies performed in patients with iris bombé associated with uveitis are more likely to fail (Spencer et al, 2001). It is usually appropriate to make these iridotomies larger if possible, but it is equally important to use topical corticosteroids afterward to avoid excessive inflammation.
A partially dislocated lens can move forward and directly shallow the anterior chamber or cause pupillary block (8‑20 and 8‑21). A dislocated lens in the anterior chamber can lead to pupillary block (8‑22). In pseudoexfoliation syndrome, the loose zonules allow the lens to move forward slightly, potentially narrowing the angle and causing pupillary block. Microspherophakia describes the condition in which there is an abnormally round, thick lens and short axial eye length, resulting in lenticular myopia (8‑23 and 8‑24). The condition can be isolated or may occur with the Weill-Marchesani syndrome. Less commonly, microspherophakia has been seen in Marfan syndrome or homocystinuria. Eyes with microspherophakia can develop lens subluxation resulting in pupillary block. Alternatively, the abnormally thick lens can cause a direct pupillary block from its native position. Pupillary block can occur in aphakia when vitreous (8‑25) or an intraocular lens (8‑26) is trapped by the iris.
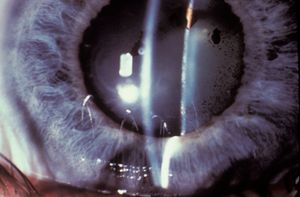
8-18 Secondary pupillary block due to central posterior synechiae. Inflammatory synechiae have formed over 360°, trapping aqueous in the posterior chamber and leading to iris bombé.
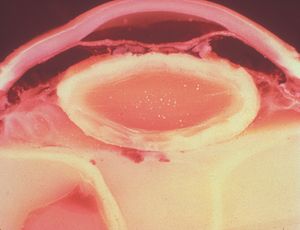
8-19 Gross photograph of eye with secondary pupillary block and iris bombé. There is adhesion of the iris to the lens capsule, causing aqueous to be trapped in the posterior segment. Broadbased peripheral anterior synechiae are noted (between arrows). (Courtesy of the National Museum of Health and Medicine, Armed Forces Institute of Pathology.)
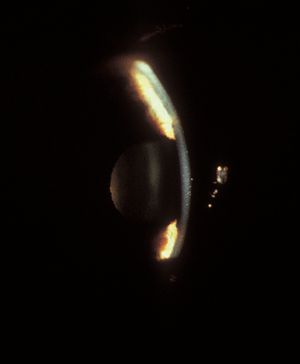
8-20 The lens is dislocated and has moved forward, causing pupillary block. Note how the iris drapes over the lens, giving a “volcano” appearance. (Courtesy of Robert Ritch, MD, New York Eye and Ear Infirmary.)

8-21 Gonioscopic view of eye shown in 8‑20. The iris is draped across the lens. The anterior position of the lens provides a view of the ciliary processes. (Courtesy of Robert Ritch, MD, New York Eye and Ear Infirmary.)
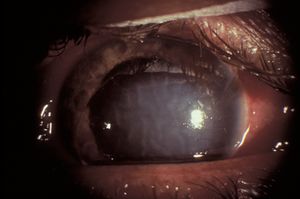
8-22 Ectopia lentis with the lens dislocated into the anterior chamber. If the lens becomes trapped in the pupillary space, pupillary block glaucoma can develop.

8-23 Pupillary block secondary to microspherophakia. This patient is an 18-year-old female with progressive myopia and elevated intraocular pressure. Note that the angle is closed and the anterior chamber is very shallow.

8-24 Same patient as in 8‑23. After dilation the anterior chamber is substantially deeper. The trabecular meshwork is still not visible due to 360° of synechiae. Note that the lens is small and round, with the zonules being visualized through the pupil.
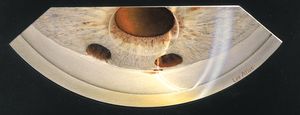
8-25 Prolapse of vitreous body through pupil. In aphakic patients the vitreous body can prolapse into the pupil, causing a pupillary block unless there are patent peripheral iridectomies, as in this patient.
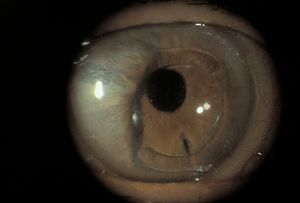
8-26 Secondary pupillary block due to an anterior chamber intraocular lens. Note ballooning of iris around intraocular lens. There is a patent iridectomy at 5 o’clock, but this is covered by the optic of the intraocular lens.

8-27 Phacomorphic glaucoma with pupillary block. Morgagnian cataract with a sunken lens nucleus (N). (Courtesy of the National Museum of Health and Medicine, Armed Forces Institute of Pathology.)
Closure of the angle by synechiae
Peripheral anterior synechiae develop when the iris becomes adherent to the ciliary body, trabecular meshwork, or peripheral cornea (8‑28 and 8‑29). Synechiae may be small, or they can be so extensive that they close the entire angle. They must be distinguished from iris processes. Synechiae are thick and opaque, whereas iris processes are usually delicate and lacy (see 5‑19 and 5‑20). Iris processes are rarely sufficiently numerous to obscure the scleral spur. Synechiae tether the iris to the angle and interfere with the posterior motion of the iris during indentation gonioscopy; iris processes do not. Synechiae bridge the angle recess, while processes tend to follow the recess. The cornea may be pigmented anterior to synechiae (8‑30).
Most synechiae attach to the scleral spur or trabecular meshwork. Although it is unusual for the iris to adhere to the cornea, this does happen in the iridocorneal-endothelial syndromes or after prolonged contact between the two bodies.
When synechiae are of recent origin, they can sometimes be broken with the laser or by surgery (goniosynechialysis), although successful synechialysis is not a guarantee that the underlying trabecular meshwork will regain any function. Some of the many conditions that can lead to the growth of synechiae are described on the following pages.
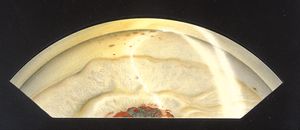
8-28 Extensive angle closure in chronic granulomatous uveitis. Trabecular meshwork can be seen only in the left-hand portion of this illustration, the remainder of the angle having been closed by synechiae. There are also central posterior synechiae at the pupil. Keratic precipitates are visible on the corneal endothelium.
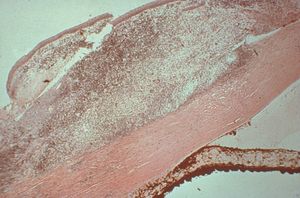
8-29 Histopathologic view of angle closed by peripheral anterior synechiae (between arrows). There is a large granuloma on the surface of this eye with a syphilitic infection. (Courtesy of the National Museum of Health and Medicine, Armed Forces Institute of Pathology.)
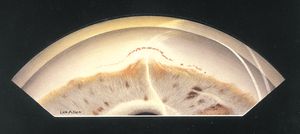
8-30 Gonioscopic view of eye with peripheral anterior synechiae due to inflammation of unknown etiology. Peripheral anterior synechiae have developed over 360°. Pigment has been deposited anterior to the peripheral anterior synechiae at 6 o’clock.
Neovascularization Blood vessels are frequently seen in normal angles. The vessels are circumferential at the base of the iris or are radial. Normal vessels do not cross the scleral spur on to the trabecular meshwork. Abnormal vessels can cross the scleral spur (8‑31 and 8‑32) and may arborize into fine branches that dive into the trabecular meshwork. While the larger branches are seen as small twigs, smaller vessels may appear only as a red blush. Neovascular vessels have an associated membrane that frequently impairs aqueous outflow despite an apparently open angle (8‑33). With time this fibrovascular membrane may contract, leading to closure of the angle by synechiae (8‑34). Many patients with neovascularization of the anterior segment will first demonstrate new vessels near the pupillary margin (8‑35) although they can be found anywhere on the iris.
Neovascularization can result from many processes. Most cases are due to retinal disease—such as diabetic retinopathy, central retinal vein occlusion retinopathy of prematurity, tumor, chronic retinal detachment, Eales disease, or sickle cell disease. Ocular ischemia, either in isolation, or associated with nonembolic central retinal artery occlusion, can also cause neovascularization of the anterior chamber Chronic inflammation can make normal angle vessels become more prominent or can cause true neovascularization. The neovascularization of Fuchs heterochromic iridocyclitis rarely leads to the development of synechiae. This disease is therefore described under open-angle mechanisms in Chapter 9.

8-31 Gonioscopic view of eye with neovascularization of the angle. Twigs of vessels are seen crossing the scleral spur, but the angle is open.

8-32 Gonioscopic view of eye with moderate neovascularization of both the iris and angle.
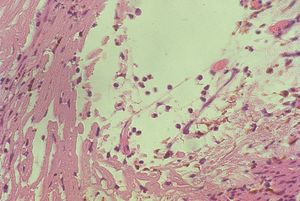
8-33 Neovascular membrane (arrow) lines an open angle. (TM, trabecular meshwork; I, iris).

8-34 Neovascular glaucoma with 360° of peripheral anterior synechiae. Note that the corneal wedge does not join into a single line in the anterior chamber. This finding confirms that the iris is pulled anterior to Schwalbe’s line.

8-35 Iris of a patient with neovascularization of the iris (rubeosis iridis). Note the vessels around the pupillary margin, which is a common location for neovascularization to occur.
Iridocorneal-endothelial syndromes The iridocornealendothelial (ICE) group of diseases have in common changes in the corneal endothelium and the formation of peripheral anterior synechiae (8‑36). They appear to be the result of an abnormal growth of corneal endothelium throughout the anterior segment. The corneal endothelium demonstrates a characteristic hammered silver appearance on examination by slit lamp (8‑37). Specular microscopy reveals markedly abnormal endothelial cells (8‑38). The ICE syndromes are unilateral and are most prevalent in women in their 30s and 40s. They frequently cause a secondary angle-closure glaucoma that can be very difficult to treat, both medically and surgically. This appears to be due to the persistent growth of the abnormal corneal endothelium across the anterior chamber and angle structures, as well as glaucoma surgical sites and drainage devices.
The ICE syndromes are usually divided into three types, but there is substantial overlap. In Chandler syndrome the corneal changes described above predominate (see 8‑37). There may be corneal edema. Abnormalities of the iris are minimal. In essential iris atrophy the iris changes predominate. The iris demonstrates evidence of melting and membrane contraction. These result in the development of stretch holes and melt holes (polycoria) and a displaced pupil (corectopia) (8‑39 and 8‑40). In iris-nevus (Cogan-Reese) syndrome nevi-like elevations develop on the face of the iris; these consist of normal iris protruding through a membrane that coats the iris (8‑41 and 8‑42). The unilateral, acquired, and progressive presentation of ICE syndrome allows it to be easily distinguished from Axenfeld- Rieger syndrome, in which many of the iris findings can be similar, but are bilateral and congenital. No definitive cause of ICE syndrome has been identified, although herpes simplex virus has been detected in the aqueous of affected eyes (Groh et al, 1999).
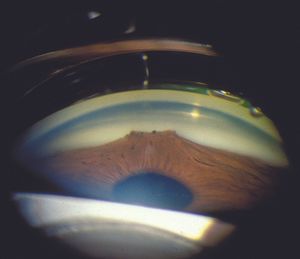
8-36 High peripheral anterior synechiae in iridocornealendothelial syndrome.
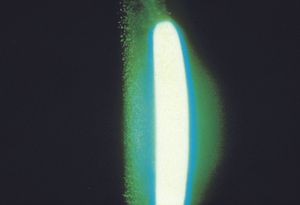
8-37 Iridocorneal-endothelial syndrome with the typical hammered metal appearance of the corneal endothelium. This patient has Chandler’s syndrome.

8-38 Specular microscopy of a patient with Chandler’s syndrome (upper portion), showing marked disruption of the endothelial pattern. The endothelium of the patient’s normal eye is shown in the lower portion of the figure.
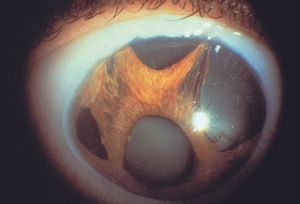
8-39 Essential iris atrophy showing corectopia with the pupil displaced inferiorly. Also visible are a stretch hole in the upper right part of the figure and a melt hole in the lower left corner of the figure.
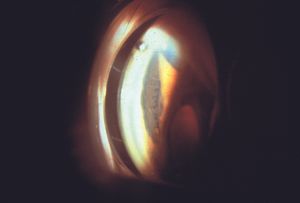
8-40 Gonioscopic view of eye shown in 8‑39, demonstrating anterior synechiae with the ciliary body drawn up towards the angle.
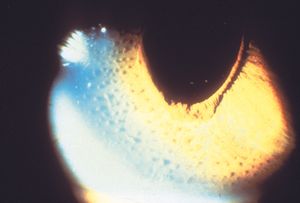
8-41 Iris-nevus syndrome showing what appear to be nodular lesions on the surface of the iris. (Courtesy of Elizabeth A. Hodapp, MD, Bascom Palmer Eye Institute.)
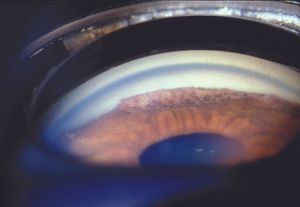
8-42 Gonioscopic view of iris-nevus syndrome, showing the same nodular iris appearance as seen in figure 8‑41, but in this patient the iris nevus pattern extends up to a broad peripheral anterior synchiae.
Posterior polymorphous dystrophy Posterior polymorphous dystrophy (PPMD) is an uncommon bilateral corneal dystrophy. It is usually an autosomal dominant disease but may be sporadic. Mutations in the gene TCF8, a transcription factor involved in collagen production, have been identified as a cause of approximately one-half of PPMD cases (Krafchak, 2005). PPMD is characterized by the presence of endothelial vesicles (8‑43); these may appear in groups, sometimes with a linear orientation (8‑44). Often the vesicles are surrounded by a corneal haze. There can be opacification of the posterior cornea. Glaucoma develops in about 14% of cases and is usually due to a membranous overgrowth of the trabecular meshwork (8‑45). Open angles with high iris insertions have been seen, suggesting a primary angle abnormality (Krachmer, 1985).
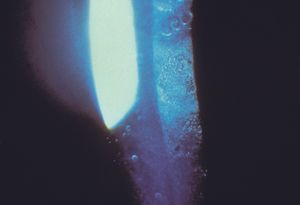
8-43 Slit lamp photograph of posterior polymorphous dystrophy in a seven-year-old child showing vesicular changes on the corneal endothelium. (Reprinted with permission from Trans Am Ophthalmol Soc [1985;83:413–475]. Krachmer JH. Posterior polymorphous corneal dystrophy: a disease characterized by epithelial-like endothelial cells which influence management and prognosis.)
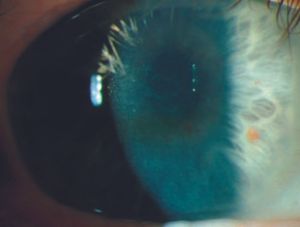
8-44 Slit lamp photograph showing a different vesicular pattern in posterior polymorphous corneal dystrophy than that seen in 8‑43.

8-45 Gonioscopic view of posterior polymorphous dystrophy. To the left/inferior the angle is open, but to the right/superior there is a large area of synechiae.
After surgery and trauma Flat anterior chambers following surgery can lead to the development of peripheral anterior synechiae (8‑46), the location of which may be anterior to Schwalbe’s line (8‑47).
Epithelial downgrowth is a rare outcome of penetrating trauma or surgery, particularly since the transition to small-wound cataract surgery. It may occur particularly when there is inadequate wound apposition. Epithelium seeded into the anterior chamber may form a cyst (8‑48) or a sheet of epithelium (8‑49). The sheets are clear and can be difficult to identify. Their presence on the iris can be detected by blanching when heated with argon laser energy; normal iris does not demonstrate blanching. As the epithelium coats the inner surfaces of the eye it covers the chamber angle (8‑50), leading to intractable glaucoma.
The synechiae formed by argon laser trabeculoplasty are usually small, conical adhesions of peripheral iris to the posterior meshwork (8‑51), but they can be extensive if the laser is aimed far to the posterior and high energy levels are used (8‑52) (Rouhiainen et al, 1988).
Inflammation Inflammation can lead to the formation of synechiae as consolidation of the inflammatory material in the angle draws the iris over the trabecular meshwork. Synechiae may develop when the iris is apposed to the trabecular meshwork for a prolonged period. If there is both apposition and inflammation, as in a flat chamber following surgery, the tendency for synechiae to form becomes marked.
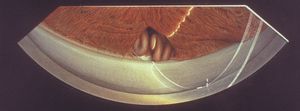
8-46 Formation of synechiae in the superior angle following filtration surgery. Ciliary body processes are incarcerated within the filtration fistula. On the right of the figure is a broad synechia. On the left, the trabecular meshwork is open with scattered low synechiae to the scleral spur. The painting is positioned to show the superior angle viewed through an indirect lens. The artist’s name therefore appears upside down.
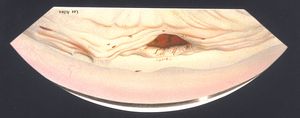
8-47 Extensive synechiae and iris adherent to wound (arrow) in patient with prolonged flat chamber following extraction of a cataract. No trabecular meshwork is visible. The painting is positioned to show the superior angle viewed through an indirect lens. The artist’s name therefore appears upside down.
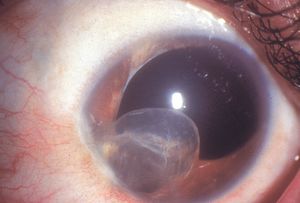
8-48 Epithelial inclusion cyst in the anterior chamber of a young man who had previously been struck in the eye with a knife.
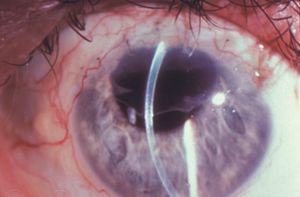
8-49 Sheet of epithelial downgrowth over the superior portion of the corneal endothelium following extraction of a cataract. (Courtesy of William W. Culbertson, MD, Bascom Palmer Eye Institute.)
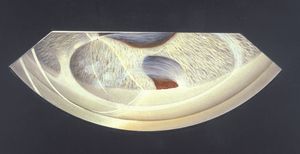
8-50 Extensive membranes within the anterior chamber causing complete synechiae. This epithelial downgrowth occurred following extraction of a cataract.

8-51 Low-lying, tent-like synechiae following argon-laser trabeculoplasty.

8-52 Extensive formation of synechiae in a myopic patient who had received laser trabeculoplasty. The energy was delivered very far posteriorly. (Courtesy of Robert Ritch, MD, New York Eye and Ear Infirmary.)
Posterior pressure
Cysts and tumors in the peripheral iris and ciliary body can lead to closure of segments of the angle (8‑53). They do not usually close enough of the angle to result in glaucoma. Some iris cysts can be opened with a laser to relieve compromise of the angle (8‑54 and 8‑55). Dislocated lenses can also cause the closure of angle segments.
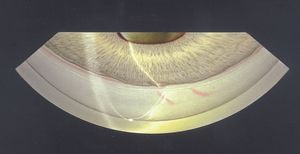
8-53 Narrowing of segmental angle due to ciliary body melanoma. Note that the iris is pushed forward in the center of the figure and obscures the trabecular meshwork, which is visible both to the right and to the left of this area. This patient has a rather prominent Schwalbe’s line and has blood in Schlemm’s canal.
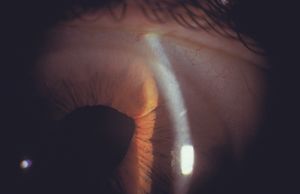
8-54 Cyst of the iris with segmental angle closure.
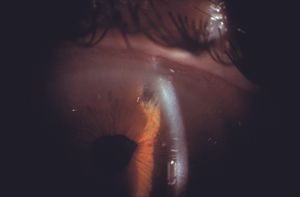
8-55 Same patient as in 8‑54, showing deepening of the chamber following laser opening of the iris cyst.
Swelling of the ciliary body and choroid
Swelling of the ciliary body (or anterior choroid) can result in angle closure (8‑56). Compromise of venous drainage or inflammation of the ciliary body may lead to such swelling. Swelling can also occur after scleral buckling procedures, after extensive panretinal photocoagulation and with ciliochoroidal detachment as a result of inflammatory pseudo-tumor (Gass, 1967). Other choroidal inflammatory processes such as Vogt-Koyanagi-Harada or posterior scleritis can also result in angle closure. Angle closure associated with topiramate (Topamax) is due to ciliochoroidal detachment as well (Banta, 2001). Angle closure due to swelling of the ciliary body and/or anterior choroid does not respond to iridectomy or iridotomy. Cholinergic agents can worsen the condition by moving the lens-iris diaphragm forward and breaking down the blood-aqueous barrier. Anticholinergic and steroid therapy along with aqueous suppression is the preferred management.

8-56 Shallow anterior chamber due to swelling of the ciliary body, the result of extensive panretinal photocoagulation.
Iridoschisis
Iridoschisis is a rare iris disorder of the elderly. In this disease there is a bilateral separation of the stroma, especially of the inferior iris. Early in the disease an intact sheet of anterior stroma may split and balloon forward (8‑57 and 8‑58). The anterior iris can obstruct the angle. Later the anterior layer of the iris may fragment (8‑59 and 8‑60). The fragments may come into contact with the corneal endothelium, leading to localized corneal edema. Glaucoma can arise as a result of angle closure either because of the anterior iris layer covering the trabecular meshwork or as a result of pupillary block (Rodrigues et al, 1983). Open-angle glaucoma can result from the presence of fragments of pigment and iris in the trabecular meshwork.
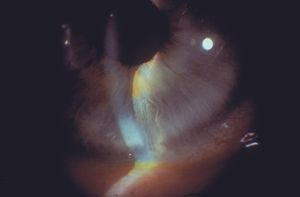
8-57 Eye of patient with iridoschisis; the anterior and posterior portions of the inferior iris have split, causing the anterior iris to bulge forward and segmentally close the angle (right eye).
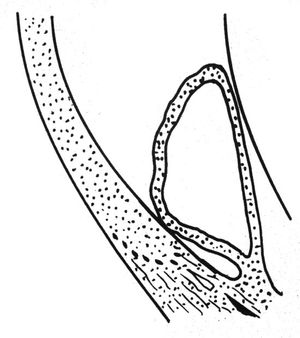
8-58 Illustration of the process shown in 8‑57, demonstrating how iridoschisis can cause angle closure.
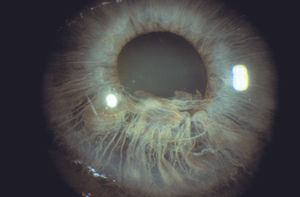
8-59 Same patient as 8‑57. The left eye showed fragmentation of the anterior iris. These iris strands were touching the cornea but had not yet caused corneal decompensation.
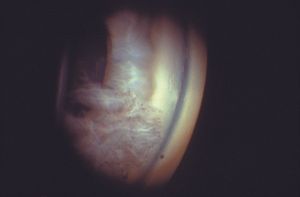
8-60 Gonioscopic photograph of eye shown in 8‑59. Fragments of the iris touch the corneal endothelium in an eye with an open angle.
Fuchs endothelial dystrophy
Fuchs endothelial dystrophy is a slowly progressive degeneration of the corneal endothelium. The loss of endothelial function leads to an accumulation of water in the cornea accompanied by an increase in its thickness (8‑61). In rare cases the thickening of the cornea can cause angle closure, particularly in an eye with already narrow angles.

8-61 Fuchs endothelial dystrophy. Marked thickening of the cornea with extensive guttata. Rarely, these patients will develop angle closure. (Courtesy of Jay H. Krachmer, MD, University of Minnesota.)