By Stephanie A. Dotchin, MD, Michael D. Penny, OD
Background
Blepharitis represents a chronic ophthalmologic disease characterized by eyelid inflammation, conjunctival redness, and persistent ocular discomfort. It is one of the most common disorders encountered by adult and pediatric eyecare providers alike.1 Mild blepharitis is often asymptomatic, while more severe forms may present with recurrent chalazia (occlusions of the meibomian glands), hordeola (infectious abscesses of the external glands of Zeis or Moll, or rarely the internal meibomian glands), and corneal inflammation.2 In the pediatric setting, blepharitis often induces a secondary immune-mediated keratitis and conjunctivitis, and the spectrum of the clinical manifestations has been termed blepharokeratoconjunctivitis (BKC). Sight-threatening corneal complications include neovascularization, scarring, and ulceration.3 BKC has also been referred to in the literature by names such as staphylococcal blepharokeratitis, meibomitis-related keratoconjunctivitis, childhood ocular rosacea, and phlyctenular keratoconjunctivitis.
BKC often presents bilaterally, though asymmetry is not uncommon. The blepharitis component can be classified anatomically into anterior and posterior divisions (Figure 1), in which anterior blepharitis primarily affects the eyelashes and anterior lid margin, while posterior blepharitis primarily affects the meibomian glands, meibomian gland orifices, and the cutaneous mucous junction of the posterior eyelid.4 The exact etiology of BKC is largely unknown but likely multifactorial. Proposed mechanisms include chronic low-grade infection with naturally occurring bacterial skin flora, Demodex mite infestation of the eyelash follicles, and inflammatory dermatologic conditions such as seborrheic dermatitis, rosacea, or atopy.5
Treatment often begins with a frequent regimen of eyelid hygiene including premedicated eyelid wipes, baby shampoo on a cotton-tipped swab or washcloth, and/or warm compresses. Topical antibiotics, anti-inflammatories, or steroid-antibiotic combinations can be added for mild to moderate cases. Oral antibiotics such as erythromycin should be considered in moderate cases or in cases with corneal involvement. Treatment for associated chalazia and hordeola includes placing a warm compress over the lesion and lightly massaging it for 10 minutes 4 times daily and using topical antibiotics if the lesion is draining or to suppress blepharitis. Systemic doxycycline may be considered in older children for its anti-inflammatory and antibacterial properties, and steroid injections or incision and curettage may be performed after 3 to 4 weeks if medical therapy fails.6
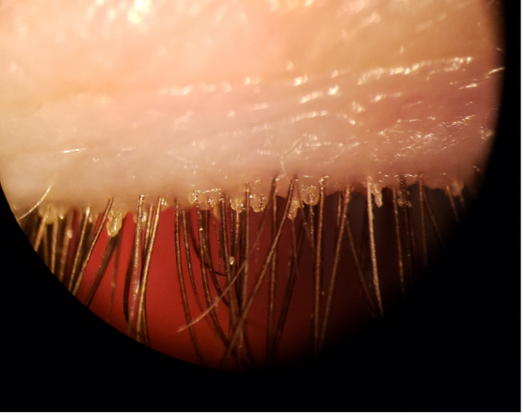
Figure 1. Anterior blepharitis with significant eyelash base collarettes signifies Demodex infestation.
Epidemiology
Multiple studies have shown that the onset of BKC in the pediatric setting is typically between 3 and 5 years of age; however, the diagnosis is often delayed 1 to 3 years, due to a high frequency of missed diagnosis or misdiagnosis.7,8 In children, the true incidence and prevalence of BKC is controversial; however, 1 study reports that BKC is the reason for referral to pediatric ophthalmology in 12% to 15% of cases.1 Children of Asian descent may be the most affected, while children of Indo-Pakistani and Middle Eastern descent often have more severe presentations.3 Eyelid margin disease is also common in the general population; 1 survey of US ophthalmologists and optometrists reported that 37% and 47% of all patients seen by those surveyed, respectively, had evidence of blepharitis.9
Risk Factors
The risk factors for BKC include poor hygiene, seborrheic dermatitis, childhood rosacea, and infestation with parasitic Demodex mites.10 The risk factors related to corneal involvement in patients with BKC may include female gender, diagnosis at an older age, asymmetric disease, and the presence of photophobia and hordeola.11
Etiology and Pathogenesis
Although the exact cause of BKC is largely unknown, proposed mechanisms based on the primary anatomic region are well supported. Anterior blepharitis can be subcategorized as staphylococcal or seborrheic blepharitis. Staphylococcal blepharitis typically presents with debris and collarette formation at the base of the eyelash cilia, along with anterior lid erythema, scaling, and prominent blood vessels. It is thought to be an overgrowth of coagulase-negative staphylococcus or Staphylococcus aureus on the ocular surface. One study revealed that patients diagnosed with staphylococcus blepharitis had a 40% positive S. aureus culture rate, compared to 9% in unaffected eyes.12 Symptoms may stem from cell-mediated immunity staphylococcal antigens and exotoxins, as enhanced-cell mediated inflammatory markers were observed in 40% of symptomatic staphylococcal blepharitis patients, but not among normal eyes in a study of ocular flora.13
Seborrheic blepharitis typically presents with scaly, itchy eyelids and a burning sensation.14 It is almost always associated with a dermatologic condition, and 1 study revealed that 95% of patients with seborrheic blepharitis had evidence of seborrheic dermatitis.15 Infantile seborrheic dermatitis (ISD) is a chronic, inflammatory scaling skin condition in infants and young children that presents with a greasy, patchy rash typically affecting the scalp (referred to as cradle cap, or formally Pityriasis capitis), eyebrows, or skin folds of the neck, arms, or diaper areas.16 A large study from Australia showed a high prevalence of seborrheic dermatitis in 44.5% of children younger than 12 months, although this number dropped substantially to 10.2% when all children under the age of 5 were examined. The majority of these cases were mild, being defined as “<5% surface area involvement and likely to respond to simple measures, such as emollients.”17 The exact cause of seborrheic dermatitis is unknown, but several associations are apparent. Studies have shown an increased culture rate of the yeast Malassezia, which is thought to release fatty acids, which in turn promotes an inflammatory reaction.18 Another suggestion is that maternal hormones overstimulate the sebaceous glands on the skin of newborns, producing a greasy layer, which prevents old skin cells from sloughing off, or that the increased fatty acids cause increased cell turnover and thus flaking of the skin. This hormonal theory is supported by the bimodal occurrence of seborrheic dermatitis in infancy and again in puberty.19
Posterior blepharitis is largely synonymous with meibomian gland dysfunction (MGD). The International Workshop on Meibomian Gland Dysfunction defines MGD as “a chronic, diffuse abnormality of the meibomian glands, commonly characterized by terminal duct obstruction and/or qualitative/quantitative changes in the glandular secretion. It may result in alteration of the tear film, symptoms of eye irritation, clinically apparent inflammation, and ocular surface disease.”20 They suggest that MGD is primarily caused by hyperkeratinization of the ductal epithelium and increased meibum viscosity, causing an altered meibum content and decreased volumes. Inadequate meibum concentration within the tear film may lead to increased evaporation, tear hyperosmolarity, increased susceptibility to lid margin bacterial growth, and ocular surface dryness and inflammation.20 MGD typically progresses from an asymptomatic pre-clinical stage with reduced meibomian gland expression to a symptomatic stage with obvious lid margin signs such as gland orifice opacities, hyperemia, and telangiectasias. With this correlation of signs and symptoms, the condition can be described as posterior blepharitis.21
The corneal findings in BKC, including punctate epithelial erosions, infiltrates, phlyctenules, pannus, and neovascularization are typically thought to be noninfectious in nature and more consistent with an inflammatory and immunological response to periocular bacteria.22 It is theorized that punctate epithelial erosions represent an effect of the bacterial exotoxins, while marginal infiltrates and ulcers represent an antigen-antibody reaction, and phlyctenules represent a delayed hypersensitivity reaction.23
Clinical Presentation
BKC typically presents as recurrent episodes of bilateral (often asymmetric) chronic red eyes, photophobia, epiphora, eye rubbing, and signs of blepharitis including inflamed lid margins with recurrent chalazia or hordeola, in an otherwise healthy child. Decreased visual acuity may occur in more advanced cases with corneal involvement.8
Symptoms
Children often present with conjunctival redness, discharge, epiphora, foreign body sensation, recurrent chalazia or hordeola, mild pain, and itching. Blurred vision and photophobia are more common with corneal involvement. Symptoms are typically worse on awakening, and chronic, but with periods of exacerbations and remissions.3,6
Signs
The clinical signs of BKC can be separated into those related to anterior or posterior blepharitis, although they often coexist. Findings consistent with anterior blepharitis include thick, inflamed eyelids, lid margin telangiectasias, scales or collarettes at the base of lashes, trichiasis (misalignment of lashes), madarosis (loss of eyelashes), or lid margin notching. Posterior blepharitis typically presents with meibomian gland opacification, pouting, capping, hypertrophy, or inflammation, posterior lid telangiectasias, or posterior lid margin scalloping.24 Recurrent chalazia, conjunctival hyperemia, chemosis, and phlyctenules are common in all forms of BKC.
Corneal sequelae are also common, but a high amount of variability in frequency exists across studies, from 5% to 81%.1,22 Common corneal findings include central or paracentral opacities with or without scarring, punctate epithelial erosions, marginal opacities with or without pannus formation, and sectoral or circumcorneal vascularization.22
Differential diagnosis
The main differential diagnosis for BKC is shown in Table 1.
Table 1. Differential diagnosis for blepharokeratoconjunctivitis
|
Disease
|
Clinical characteristics
|
Additional features
|
|
Chronic allergic conjunctivitis (atopic or vernal)
|
Marked itching, atopic facial and eyelid dermatitis. Significant conjunctival papillary reaction25
|
History of atopic disease (asthma or atopic dermatitis)
|
|
Bacterial conjunctivitis (gonococcal or non-gonococcal)
|
Copious purulent discharge with conjunctival erythema26
|
Abrupt onset. Must rule out associated corneal ulcer.
|
|
Viral conjunctivitis
|
Diffuse conjunctival injection with follicular reaction, serous tearing, and a burning sensation7
|
May present with fever, rash, and arthralgias
|
|
Herpes simplex eye disease
|
Unilateral tearing, burning, and photophobia with dendritic corneal ulcers and desensitization27
|
Evaluate for vesicular dermatologic lesions near eyelid margin. Corneal dendrites are best assessed with fluorescein staining.
|
|
Molluscum contagiosum (pox virus)
|
Chronic, follicular keratoconjunctivitis with or without obvious dermal lid lesions28
|
Lesions appear as round, raised flesh-colored bumps with a small central indentation
|
|
Phthirus pubis (crab lice)
|
Chronic low-grade conjunctivitis and irritation. Appearance mimics that of anterior blepharitis25
|
Child should be screened for possibility of sexual abuse
|
Investigations
The diagnosis of blepharitis is clinical and often apparent with a thorough history and physical examination. Fluorescein staining (demonstrated in Figure 2) should be performed to better visualize corneal abrasions, infiltrates, and neovascularization. Lid margin and ocular surface bacterial culture swabs are not routinely done in practice, although they may be necessary in severe or persistent cases.29
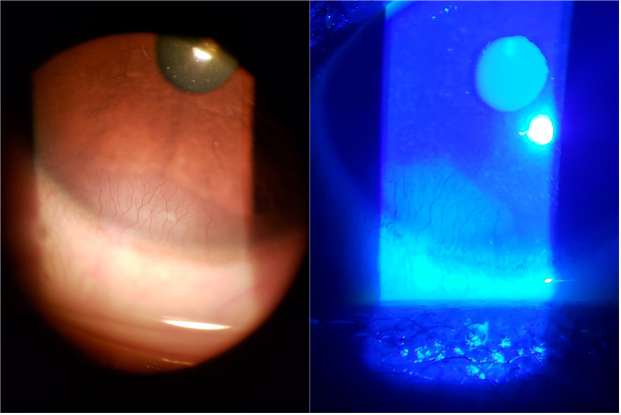
Figure 2. Corneal neovascularization, shown before and after fluorescein staining.
Treatment
The mainstay of therapy for BKC targets the inflammatory and infectious components of the underlying blepharitis and is often prolonged indefinitely, due to the chronic nature of the condition. The general treatment algorithm for BKC begins with a frequent regimen of eyelid hygiene for all patients. Depending on severity, topical or oral antibiotics, topical anti-inflammatories, oral omega-3 fatty acids, and topical immunosuppression agents may be considered.
Eyelid hygiene is thought to directly decrease the amount of bacterial flora on the periocular tissues, as well as liquefying and expressing stagnant meibum from the blocked meibomian glands. This involves a routine of daily eyelid and eyelash cleansing with a mild baby shampoo, as well as use of a warm compress for 10 to 15 minutes 2 to 4 times daily, depending on the presence of chalazia. Light massage with a finger or cotton swab may also be performed, as it is thought to improve meibomian gland outflow. Although this is difficult to enforce and maintain with children and their parents, it must be stressed as the foundation for a successful treatment.30 Lubricant eyedrops are also considered standard of care for these patients, as they provide symptomatic relief, support the integrity of the tear film, dilute proinflammatory markers, reduce osmolarity toward physiologic levels, and reduce friction between the tarsal conjunctiva and corneal epithelium. It is thought that to prevent preservative toxicity, preservative-free artificial tears should be considered when use exceeds 4 to 6 times daily.31
Patients with significant MGD may also benefit from omega-3 supplementation, although the evidence for use in pediatric BKC is limited. Omega-3 fatty acids are thought to reduce the overall inflammatory state of the eyelid margin by competitively inhibiting omega-6 pro-inflammatory metabolism, and by reducing meibum stagnation through a decreased secretion viscosity.32 One teaspoon, or 2.5 ml, of flax seed oil daily for 6 weeks has been suggested for pediatric BKC, although newer literature would recommend fish oil as a better source of omega-3s.22 This is because flax seed contains the omega-3 alpha-linolenic acid (ALA), which must be converted by limited enzymes into eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), which are the omega-3s believed to provide the anti-inflammatory benefits. This conversion means that only 10%-15% of the omega-3s from flax seed provides benefit, while the remaining ALA is metabolized in other ways.33 A recent randomized control trial has also suggested that krill oil, which contains omega-3s in a phospholipid form, is superior to fish oil, which contains omega-3s in a predominantly triacylglyceride form, when used in the treatment of dry eye disease on meibomian gland dysfunction.34 The ideal dosing for pediatric patients has not been well established, but 1 suggested regimen is 1 teaspoon of fish or krill oil daily for younger children, 2 teaspoons daily for school age children, and 1 tablespoon daily for adolescents.
Topical and oral antibiotics with broad-spectrum gram-positive coverage are used to reduce the bacterial colonization of the eyelash bases, eyelid margin, and conjunctival tissue.35 The decrease in lipase and lipolytic exoenzyme-producing bacterial flora is thought to decrease the meibomian lipid breakdown products, which contributes to widespread improvement of the clinical picture.36 Oral tetracyclines and macrolides also display anti-inflammatory properties, which decrease concentrations of inflammatory cytokines, and activity of matrix metalloproteinases, phospholipase-A2, and collagenases. This anti-inflammatory effect of oral tetracyclines and macrolides can be observed in a wide variety of ocular tissues, including the corneal epithelium.37,38
Commonly used topical antibiotic agents include tobramycin 0.3% drops or ointment, azithromycin 1% drops, and erythromycin 0.5% ointment. Topical chloramphenicol is not readily available in the United States but is commonly used elsewhere, representing significant regional variability in treatment. Drops are typically instilled twice daily; however, a recent study showed azithromycin dosed as a 3-day treatment (1 drop bid) initially prescribed every 10 days for at least 1 month, then tapered to 1 treatment every 15 days, then 1 treatment monthly, was a safe and effective strategy for BKC. The author of this study attributes the efficacy of azithromycin to its long half-life and significant tissue accumulation.39 If the BKC is moderately severe, azithromycin gel or erythromycin ointment can be added to the eyelids every night before bed.6
Commonly used oral antibiotics for moderate to severe BKC or BKC with corneal involvement include tetracyclines and macrolides, although it should be noted that tetracyclines are contraindicated in children under the age of 8 years, due to the potential to affect bone growth and tooth development. The dosage for tetracycline ranges from 250 mg once to 4 times daily, and the dosage for doxycycline ranges from 50 mg to 100 mg, once or twice per day. Most authors suggest use for 6 to 8 weeks, with a slow taper.35 Common side effects of oral tetracyclines include diarrhea, nausea, headache, photosensitization, and vaginal or oral candidiasis.31 Although erythromycin is the most commonly used oral macrolide in pediatric BKC, azithromycin and clarithromycin have also been used successfully.40 The dosage for erythromycin ranges from 12.5 mg/kg to 40 mg/kg body weight, divided into 2 or 3 doses, for 7-12 months. The pediatric dosage for azithromycin is not well established; however, 1 small study reports a dosage of 5 mg/kg daily was effective at treating BKC.41 Gastrointestinal upset is common with oral macrolides, although it occurs less frequently with azithromycin and clarithromycin than erythromycin.42
Topical corticosteroids are advised in moderate to severe BKC to reduce sight-threatening conjunctival and corneal inflammation. For control of acute exacerbations, prednisolone 0.3%-0.5% or dexamethasone 0.1% can be used 4 to 6 times per day. For chronic inflammatory control, fluorometholone 0.1%, or loteprednol 0.5%, is typically dosed 4 times daily for 4 to 6 weeks, with a slow taper.11 These lower-potency agents carry less risk of inducing intraocular pressure (IOP) spikes, compared to prednisolone and dexamethasone.43 Frequent IOP checks should be performed for all patients to monitor for steroid-induced ocular hypertension. Other side effects of topical corticosteroids include cataract formation and increased susceptibility to infections such as herpetic keratitis.44
Topical antibiotic-steroid combinations are indicated for “steroid-responsive inflammatory ocular conditions for which a corticosteroid is indicated and where superficial bacterial ocular infection or a risk of bacterial ocular infection exists,” and are thus frequently prescribed for pediatric BKC.45 Commonly used drops include dexamethasone 0.1%-tobramycin 0.3%, or loteprednol 0.5%-tobramycin 0.3% dosed 4 times daily. Unsurprisingly, reducing the amount of drops a patient or caregiver must instill through the use of combination medications has been shown to improve compliance and treatment outcomes.46 Limiting the number of drops is also particularly important in children, as eyedrop administration can have a significant psychological impact that can result in poor cooperation, a negative effect on the doctor-patient relationship, and greater difficulty with subsequent examinations.47
Steroid-sparing immunosuppressant agents, such as the calcineurin inhibitor cyclosporin-A, have been suggested for use in pediatric BKC patients with severe corneal involvement. One study suggests that twice-daily topical unpreserved 0.05% cyclosporin-A emulsion for a minimum of 3 months allows for adequate control of inflammation, although the study noted a high dropout rate due to intolerance and high costs associated with the drug.11 Common side effects of topical cyclosporin-A include blurred vision, burning, and itching.
Complications
Vision-threatening complications associated with advanced disease include corneal neovascularization, scarring, thinning, and perforation. Failure to recognize or control the disease despite diagnosis has been cited as a main contributing factor for severe disease, highlighting the importance of specialist involvement.29 Amblyopia is another potentially devastating complication of corneal involvement. Refractive amblyopia is possible due to induced irregular astigmatism from neovascularization or scarring, and deprivation amblyopia is possible with dense corneal opacities. Cycloplegic refractions are essential to monitor for changes in visual acuity, and amblyopia should be treated promptly with proper spectacle correction, patching, or pharmacological methods.48
Chalazion and Hordeolum
Frequent chalazion and hordeolum are commonly seen in pediatric BKC and eyelid margin disease. They typically present as an acute or chronic focal eyelid swelling, which can be painless or tender. These nodules are often well-defined, visible, and palpable.6 A chalazion is a localized lipogranulomatous inflammation within the eyelids, often secondary to a noninfectious obstruction of the meibomian gland or gland of Zeis. It is the most common inflammatory lesion of the eyelid.49 A hordeolum is an acute bacterial infection of the external eyelid glands (glands of Zeis or Moll), commonly known as styes, or infections of the internal meibomian glands. They are typically caused by a staphylococcal infection and may spread to surrounding tissues, causing a preseptal cellulitis, although most cases drain spontaneously and resolve untreated within 1 to 2 weeks.50
Treatment begins with a warm compress over the lesion for 10 minutes, followed by light massage over the lesion 4 times daily, with the intent of liquefying the stagnant secretions to allow for drainage through the gland orifices. Since a towel soaked in warm water only stays warm for 30 to 60 seconds, commercially available eye masks that can be heated in the microwave provide longer, more consistent heat therapy. Home remedies include a boiled egg, a microwaved potato, or a sock filled with dry rice. Although the suggested treatment duration of 10 minutes may be difficult for children, caregivers should make their best effort. If there is significant aversion to a warm compress, caregivers may resort to simply getting warm water in the eyes during bath or shower time (although this is not likely to be as effective).
Eyelid hygiene using a clean cloth and a warm, dilute solution of baby shampoo (ie, 1 tsp baby shampoo per cup of warm water) rubbed back and forth across the eyelashes and eyelash margins may also help to reduce accumulated debris.51 Topical and oral antibiotics may be prescribed for internal and external hordeolum and associated blepharitis but are ineffective toward the treatment of noninfectious chalazia. Commonly used topical antibiotic agents include erythromycin ointment or azithromycin gel twice daily. Although contraindicated for patients under the age of 8 years, oral doxycycline may be prescribed for its antibacterial and anti-inflammatory actions.6 For noninfectious chalazia, topical corticosteroids for 3 to 4 weeks can help to blunt the immune and inflammatory response, shrinking the size of the lesion so that the child’s body can eventually reabsorb the granulation tissue. Unlike in adults, chronic chalazia in children can resolve on their own over long periods of time (10-12 months) if reinfection does not occur. For treatment resistant, chronic, or progressive chalazia and hordeola, incision and curettage or intralesional triamcinolone steroid injection may be warranted. However, due to reports of choroidal and retinal vascular occlusion, accidental globe perforation, and skin depigmentation associated with triamcinolone injections, some providers reserve this treatment only for cases in which incision and curettage cannot be performed. These patients include those with allergies to local anesthetic, and those with chalazia overlying or close to the lacrimal drainage system.52
Prognosis
The diagnosis of BKC should be considered in all children with chronic ocular discomfort and signs of eyelid inflammation. Frequent follow-up is required to monitor for corneal involvement and disease exacerbations. Although the management of BKC in children can be challenging, prompt diagnosis and aggressive treatment can minimize sight-threatening corneal inflammation and scarring.
References
- Gupta N, Dhawan A, Beri S, D’souza P. Clinical spectrum of pediatric blepharokeratoconjunctivitis. J AAPOS. 2010 Dec;14(6):527-529. doi:10.1016/j.jaapos.2010.09.013
- Blepharitis. Accessed February 12, 2021. https://www-dynamed-com.ezproxy.lib.ucalgary.ca/condition/blepharitis
- Viswalingam M, Rauz S, Morlet N, Dart JKG. Blepharokeratoconjunctivitis in children: diagnosis and treatment. Br J Ophthalmol. 2005;89(4):400-403. doi:10.1136/bjo.2004.052134
- Guillon M, Maissa C, Wong S. Eyelid margin modification associated with eyelid hygiene in anterior blepharitis and meibomian gland dysfunction. Eye Contact Lens. 2012;38(5). doi:10.1097/ICL.0b013e318268305a
- Lindsley K, Matsumura S, Hatef E, Akpek EK. Interventions for chronic blepharitis. Cochrane Database Syst Rev. 2012 May 16;2012(5):CD005556. doi:10.1002/14651858.CD005556.pub2.www.cochranelibrary.com
- Gerstenblith AT, Rabinowitz MP, eds. The Wills Eye Manual: Office and Emergency Room Diagnosis and Treatment of Eye Disease. 6th ed. Wolters Kluwer/Lippincott Williams & Wilkins; 2012.
- Beal C, Giordano B. Clinical Evaluation of Red Eyes in Pediatric Patients. J Pediatr Heal Care. 2016;30:506-514. doi:10.1016/j.pedhc.2016.02.001
- Hammersmith KM, Cohen EJ, Blake TD, Laibson PR, Rapuano CJ. Blepharokeratoconjunctivitis in children. Arch Ophthalmol. 2005;123(12):1667-1670. doi:10.1001/archopht.123.12.1667
- Lemp MA, Nichols KK. Blepharitis in the United States 2009: A Survey-Based Perspective on Prevalence and Treatment From the Department of Ophthalmology. Accessed February 16, 2021. www.theocularsurface.com
- Amescua G, Akpek EK, Farid M, et al; American Academy of Ophthalmology Preferred Practice Pattern Cornea and External Disease Panel. Blepharitis Preferred Practice Pattern®. Ophthalmology. 2019;126(1):P56-P93. doi:10.1016/j.ophtha.2018.10.019
- Rodríguez-García A, González-Godínez S, López-Rubio S. Blepharokeratoconjunctivitis in childhood: corneal involvement and visual outcome. Eye (Lond). 2016;30(3):438-446. doi:10.1038/eye.2015.249
- Dougherty JM, McCulley JP. Comparative bacteriology of chronic blepharitis. Br J Ophthalmol. 1984;68:524-528. doi:10.1136/bjo.68.8.524
- Ficker L, Ramakrishnan M, Seal D, Wright P. Role of cell-mediated immunity to staphylococci in blepharitis. Am J Ophthalmol. 1991;111(4):473-479. doi:10.1016/S0002-9394(14)72383-9
- Bernardes TF, Bonfioli AA. Blepharitis. Semin Ophthalmol. 2010 May;25(3):79-83. doi:10.3109/08820538.2010.488562
- McCulley JP, Dougherty JM, Deneau DG. Classification of chronic blepharitis. Ophthalmology. 1982;89(10):1173-1180. doi:10.1016/S0161-6420(82)34669-2
- Victoire A, Magin P, Coughlan J, van Driel ML. Interventions for infantile seborrhoeic dermatitis (including cradle cap) 2019 Mar 4;3(3):CD011380. doi:10.1002/14651858.CD011380.pub2
- Foley P, Zuo Y, Plunkett A. The frequency of common skin conditions in preschool-aged children in Australia: seborrheic dermatitis and pityriasis capitis (cradle cap). 2003 Mar;139(3):318-322. Accessed February 23, 2021. https://jamanetwork.com/
- Gupta AK, Nicol K, Batra R. Role of Antifungal Agents in the Treatment of Seborrheic Dermatitis. 2004;5(6):417-422.
- Ro BI, Dawsonw TL. The Role of Sebaceous Gland Activity and Scalp Microfloral Metabolism in the Etiology of Seborrheic Dermatitis and Dandruff. Vol 10.; 2005. doi:10.1111/j.1087-0024.2005.10104.x
- Nichols KK, Foulks GN, Bron AJ, et al. The international workshop on meibomian gland dysfunction: executive summary. Invest Ophthalmol Vis Sci. 2011;52(4):1922-1929. doi:10.1167/iovs.10-6997a
- Tomlinson A, Bron AJ, Korb DR, et al. The international workshop on meibomian gland dysfunction: report of the diagnosis subcommittee. 2011;52:2006-2049. doi:10.1167/iovs.10-6997f
- Jones SM, Weinstein JM, Cumberland P, Klein N, Nischal KK. Visual outcome and corneal changes in children with chronic blepharokeratoconjunctivitis. Ophthalmology. 2007;114:2271-2280. doi:10.1016/j.ophtha.2007.01.021
- Tetz MR, Klein U, Völcker HE. Staphylokokken-assoziierte Blepharokeratokonjunktivitis. Ophthalmologe. 1997;94(3):186-190. doi:10.1007/s003470050099
- Wong IBY, Nischal KK. Managing a child with an external ocular disease. J AAPOS. 2010 Feb;14:68-77. doi:10.1016/j.jaapos.2009.11.021
- Daniel CM, O'Gallagher, Hingorani M, Dahlmann-Noor A, Tuft S. Challenges in the management of pediatric blepharokeratoconjunctivitis / ocular rosacea. Expert Review of Ophthalmology. 2017; 11:4, 299-309. doi:10.1080/17469899.2016.1209408
- Smith G. Differential diagnosis of red eye. Pediatr Nurs. 2010;36(4):213-215.
- Vadoothker S, Andrews L, Jeng BH, Levin MR. Management of Herpes Simplex Virus Keratitis in the Pediatric Population. Pediatr Infect Dis J. 2018;37(9):949-951. doi:10.1097/INF.0000000000002114
- Schornack MM, Siemsen DW, Bradley EA, Salomao DR, Lee HB. Ocular manifestations of molluscum contagiosum. Clin Exp Optom. 2006;89(6):390-393. doi:10.1111/j.1444-0938.2006.00073.x
- Hamada S, Khan I, Denniston AK, Rauz S. Childhood blepharokeratoconjunctivitis: characterising a severe phenotype in white adolescents. doi:10.1136/bjophthalmol-2011-300771
- Donaldson KE, Karp CL, Dunbar MT. Evaluation and treatment of children with ocular rosacea. 2007;26(1):42-46.
- Geerling G, Tauber J, Baudouin C, et al. The international workshop on meibomian gland dysfunction: report of the subcommittee on management and treatment of meibomian gland dysfunction. Invest Ophthalmol Vis Sci. 2011; Mar 30;52(4):2050-2064. doi:10.1167/iovs.10-6997g
- Macsai MS. The role of omega-3 dietary supplementation in blepharitis and meibomian gland dysfunction (an AOS thesis). Trans Am Ophthalmol Soc. 2008; 106:336-356.
- Why not flaxseed oil? - Harvard Health. Accessed June 4, 2021. https://www.health.harvard.edu/heart-health/why-not-flaxseed-oil
- Deinema LA, Vingrys AJ, Wong CY, Jackson DC, Chinnery HR, Downie LE. A randomized, double-masked, placebo-controlled clinical trial of two forms of omega-3 supplements for treating dry eye disease. Ophthalmology. 2017;124(1):43-52. doi:10.1016/j.ophtha.2016.09.023
- O’Gallagher M, Banteka M, Bunce C, Larkin F, Tuft S, Dahlmann-Noor A. Systemic treatment for blepharokeratoconjunctivitis in children. Cochrane Database Syst Rev. 2016 May 30;(5):CD011750. doi:10.1002/14651858.CD011750
- Aronowicz JD, Shine WE, Oral D, Vargas JM, McCulley JP. Short term oral minocycline treatment of meibomianitis. Br J Ophthalmol. 2006;90:856-860. doi:10.1136/bjo.2006.091579
- Solomon A, Rosenblatt M, Li DQ, et al. Doxycycline inhibition of interleukin-1 in the corneal epithelium. Invest Ophthalmol Vis Sci. 2000;41(9):2544-2557. doi:10.1016/s0002-9394(00)00755-8
- Sadrai Z, Hajrasouliha AR, Chauhan S, Saban DR, Dastjerdi MH, Dana R. Effect of topical azithromycin on corneal innate immune responses. Invest Ophthalmol Vis Sci. 2011;52(5):2525-2531. doi:10.1167/iovs.10-5658
- Doan S, Gabison E, Chiambaretta F, Touati M, Cochereau I. Efficacy of azithromycin 1.5% eye drops in childhood ocular rosacea with phlyctenular blepharokeratoconjunctivitis. J Ophthalmic Inflamm Infect. 2013;3(1):38. doi:10.1186/1869-5760-3-38
- Suzuki T. Meibomitis-Related keratoconjunctivitis: Implications and clinical significance of meibomian gland inflammation. Cornea. 2012;31(11 Suppl 1):41-44. doi:10.1097/ICO.0b013e31826a04dd
- Choi DS, Djalilian A. Oral azithromycin combined with topical anti-inflammatory agents in the treatment of blepharokeratoconjunctivitis in children. J AAPOS. 2013;17(1):112-113. doi:10.1016/j.jaapos.2012.10.015
- Klein JO. History of macrolide use in pediatrics. Pediatr Infect Dis J; 1997; Apr; 16(4):427-431. doi:10.1097/00006454-199704000-00025
- Nuyen B, Weinreb RN, Robbins SL. Steroid-induced glaucoma in the pediatric population. J AAPOS. 2017; Feb;21(1) 1-6. doi:10.1016/j.jaapos.2016.09.026
- Srinivasan M, Mascarenhas J, Rajaraman R, et al; Steroids for Corneal Ulcers Trial Group. Corticosteroids for bacterial keratitis: The steroids for corneal ulcers trial (SCUT). Arch Ophthalmol. 2012;130(2):143-150. doi:10.1001/archophthalmol.2011.315
- TobraDex® Prescribing Information, 2005, Alcon Pharma- Ceuticals/Laboratories, Inc.: Fort Worth (TX), USA. Accessed June 2, 2021. www.fda.gov/medwatch.
- Gugleta K, Orgül S, Flammer J. Experience with Cosopt, the fixed combination of timolol and dorzolamide, after switch from free combination of timolol and dorzolamide, in Swiss ophthalmologists’ offices. Curr Med Res Opin. 2003;19(4):330-335. doi:10.1185/030079903125001820
- Sujuan JL, Handa S, Perera C, Chia A. The psychological impact of eyedrops administration in children. J AAPOS. 2015; Aug;19(4):338-343. doi:10.1016/j.jaapos.2015.05.010
- Rousta ST. Pediatric blepharokeratoconjunctivitis: Is there a “right” treatment? Curr Opin Ophthalmol. 2017;28(5):449-453. doi:10.1097/ICU.0000000000000399
- Özdal PÇ, Codère F, Callejo S, Caissie AL, Burnier MN. Accuracy of the clinical diagnosis of chalazion. Eye (Lond). 2004;18:135-138. doi:10.1038/sj.eye.6700603
- Lindsley K, Nichols JJ, Dickersin K. Non-surgical interventions for acute internal hordeolum. Cochrane Database Syst Rev. 2017 Jan 9;1(1):CD007742. doi:10.1002/14651858.CD007742.pub4
- Skorin L. Hordeolum and Chalazion Treatment The Full Gamut. Accessed March 2, 2021. www.optometry.co.uk
- Lee JWY, Yau GSK, Wong MYY, Yuen CYF. A comparison of intralesional triamcinolone acetonide injection for primary chalazion in children and adults. ScientificWorldJournal. 2014;2014. doi:10.1155/2014/413729