Download PDF
Cases of atypical keratitis require a high degree of suspicion for Acanthamoeba keratitis (AK) and prompt referral for Acanthamoeba cultures, particularly in contact lens wearers, according to a study conducted at Wilmer Eye Institute in Baltimore.1 Because of delayed or inaccurate diagnoses prior to referral, several patients in this study required PK, and the average final visual acuity (VA) of those receiving a late diagnosis was 20/240.
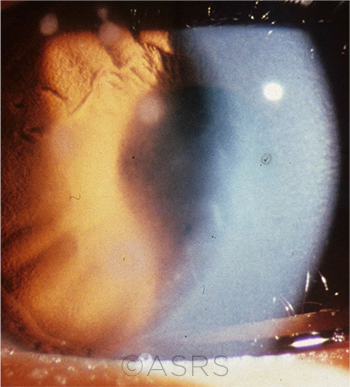 |
NOT CLEAR-CUT. Classic symptoms of Acanthamoeba keratitis—including severe pain—may not be present, potentially leading to an initial misdiagnosis. This image was originally published in the ASRS Retina Image Bank. David Callanan, MD. Acanthamoeba Keratitis. Retina Image Bank. 2014; Image Number 12851. © The American Society of Retina Specialists.
|
Easy to misdiagnose. This retrospective study included all patients (n = 43; 45 eyes) with culture-positive AK seen at Wilmer between 2012 and 2019. The patients’ mean age was 41 years, and they had been symptomatic for a mean of 52.6 days (range, 6-231). Before referral, 17 (37.8%) and nine (20%) eyes were misdiagnosed with herpetic and bacterial keratitis, respectively. An additional 13 (29%) received no initial diagnosis.
Risk factors and symptoms. Contact lens use was the strongest risk factor for AK, with nearly all eyes affected (95.6%). Notably, only five eyes (11%) presented with the classic ring infiltrate of AK.
Although 37 (82%) eyes were affected by pain or discomfort, only half had severe pain. “AK is typically characterized by pain out of proportion to findings,” suggesting that ophthalmologists should not rely solely on pain as a criterion, said Fasika A. Woreta, MD, MPH. Other common presenting symptoms included photophobia and blurred vision, and diffuse, multifocal, or deep infiltrate was present in roughly one-third of the patients.
Outcomes. At presentation, mean VA was 20/224 in those diagnosed within 28 days of symptom onset and 20/296 in those diagnosed more than 28 days after onset. At the final visit, VA was 20/92 in the early diagnosis group and 20/240 in the late diagnosis group.
The benefit of early diagnosis in this study did not reach statistical significance, possibly due to insufficient sample size. However, the trend was toward statistical significance, as late diagnoses yielded worse VA outcomes, Dr. Woreta noted.
PK was necessary in five eyes (11%). Although none of these eyes had the classic ring infiltrate, two presented with diffuse infiltrate. Three of the five were misdiagnosed with either bacterial or herpetic keratitis.
Need for easier testing. One reason for delayed diagnosis may be a lack of access to diagnostic testing and culturing outside of tertiary care centers. To that end, coauthor Nakul Shekhawat, MD, MPH, proposed development of a rapid polymerase chain reaction test or slide preparation that can be administered in clinicians’ offices. In the meantime, Dr. Woreta recommended that all contact lens wearers with any form of atypical keratitis be referred to tertiary centers for AK cultures “before making a presumed diagnosis of herpes keratitis, which can mimic AK.”
—Miriam Karmel
___________________________
1 Shah YS et al. Acta Ophthalmol. Published online Feb. 14, 2021.
___________________________
Relevant financial disclosures: Dr. Shekhawat: NEI: S. Dr. Woreta: None.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Korenfeld Encore Vision: S; EyeGate: C; EyePoint: C; Novartis: C,S; Orasis: C.
Dr. Shekhawat NEI: S.
Dr. Teo Bayer: L; Novartis: L; Topcon: L.
Dr. Woreta None.
Dr. WuDunn Allergan: S.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|
More from this month’s News in Review