By John P. Berdahl, MD, Frank J. Martin, MD, and April Y. Maa, MD
Download PDF
Because the end of the year is a fitting time to take stock of recent clinical developments, EyeNet has asked three of its editorial board members to review their areas of expertise and to consider recent trends and news that have the greatest potential to shape their subspecialty over the next several years. Perhaps not surprisingly, all of their observations involve the use of technology to improve patient care. Below are their perspectives.
DR. BERDAHL ON GLAUCOMA
Glaucoma Treatment’s Three Waves
For years, traditional tube shunt surgeries and trabeculectomy had been the mainstay therapies for effectively lowering intraocular pressure (IOP) in patients with glaucoma. But results from the 2012 Tube Versus Trabeculectomy (TVT) Study corroborated some of our safety concerns—surgeons were witnessing early- and late-term failures, devastating infections, excessively low IOP, and other complications. Here was an unmet need: How can ophthalmologists provide an alternative to traditional surgery? In stepped microinvasive glaucoma surgery (MIGS).
The first wave: MIGS. With its FDA approval the same year as the TVT Study, the iStent (Glaukos) kicked off the first new wave of innovation for treating glaucoma. It provided ophthalmologists with precise and personalized management of mild to moderate disease through bypassing the resistance of the trabecular meshwork via a tiny stent that increases outflow. This microincisional technique resulted in reduced physiological trauma, rapid post-op recovery, and minimal need for follow-up. Additionally, it allowed patients to reduce their medication burden and avoid the complications associated with traditional procedures.
In eight years on the market, MIGS has become a vital part of the treatment continuum, with a range of different devices now available. However, as with the evolution of other innovative therapies, there have been challenges along the way. Approved by the FDA in 2016, the CyPass Micro-Stent (Alcon) was withdrawn from the market and then recalled two years later because of results from the COMPASS XT study indicating a troubling rate of endothelial cell loss. Nevertheless, we’ve experienced a proliferation of safe and innovative products—such as the Kahook Dual Blade (New World Medical), Omni (Sight Sciences), and Hydrus (Ivantis)—as well as innovative techniques—like endoscopic cyclophotocoagulation—to target and manipulate the eye’s outflow pathways and aqueous dynamics.
The second wave: drug delivery. Innovation didn’t end with MIGS. For years, studies have shown that glaucoma patients are nonadherent to their medications—whether because of difficulty with drop application, trouble sticking with a regimen that requires frequent administration, or side effects. Here was another unmet need: How can ophthalmologists help deliver glaucoma drugs to patients exactly where they need it, when they need it? In came the next new wave of treatment, sustained delivery.
Durysta. March 2020 marked a significant milestone in drug delivery when the FDA approved the first sustained-release, biodegradable, IOP-lowering implant, Durysta (Allergan). Placed in the anterior chamber via a preloaded, single-use applicator, this implant delivers 10 μg bimatoprost—similar to a single drop of the ophthalmic 0.03% solution. In one of two phase 3 studies, Durysta reduced IOP by close to 30% during a 12-week primary efficacy period. These results also suggested, somewhat surprisingly, the potential for many patients to remain treatment free for at least one year following implantation. The one caveat? The FDA approved the implant for only a single use in each eye. However, Allergan is compiling additional data to expand the label for multiple administrations. (For more about Durysta, see “Bimatoprost Implant: What Role Will It Play in Glaucoma Management?”)
In the pipeline. A number of other sustained-release technologies are in the works as well, including punctal plugs (Ocular Therapeutix), internally placed reservoirs (Glaukos), and external rings (Allergan). Taken together, these options represent a paradigm shift in how ophthalmologists can combat patient noncompliance. We’re now able to deliver medication at much lower concentrations directly to targeted tissues without having to pass through the tear film and the cornea. As a result, these minute dosages reduce side effects by eliminating effects to the ocular surface.
The third wave: treating severe cases. Both MIGS and enhanced drug delivery are dramatically changing the approach to treating mild-to-moderate glaucoma. Yet, innovation for patients with severe and difficult-to-treat disease, such as normal-tension glaucoma, has been more incremental.
FDA approved. Modifications of existing approaches are helping. For example, technologies that bypass episcleral venous pressure can help hit low target IOPs. New World Medical, for instance, has recently improved filtration shunt technology with the Ahmed ClearPath. Its flexible, contoured plate allows for less-intrusive placement as well as a more natural fit in the eye. Because it’s sutured more anteriorly than previous filtration devices, it provides the surgeon with a better view when securing the implant to the eye. The Xen Gel Stent (Allergan) also allows for a less-invasive approach to transscleral surgery via a preloaded ab interno injection technique.
In the pipeline. Two intriguing shunts are also in the FDA approval process. The PreserFlo MicroShunt (Santen) involves the creation of a full-thickness fistula from the anterior chamber across to the subconjunctival space and is designed to prevent hypotony. The Beacon Aqueous Microshunt (MicroOptx), on the other hand, shunts aqueous humor from the anterior chamber to the ocular surface. Both products look to be promising new takes on traditional filtration devices.
In addition, exciting noninvasive, nonpharmaceutical approaches are on the horizon for patients running out of options. For example, the Mercury Multi-Pressure Dial (Equinox; note that I helped found the company and helped create the product) consists of a pair of goggles that draw a vacuum around each eye’s periorbital area. Each goggle is connected to a pressure-modulating pump, which, when activated, establishes a targeted negative pressure in the microenvironment surrounding the eye. This release of atmospheric pressure placed on the eye results in the almost-instantaneous lowering of IOP and is titratable even at low pressures.
A multiplicity of treatment options. Glaucoma surgeons are indeed experiencing a renaissance. MIGS filled an important gap in the treatment algorithm between medications, selective laser trabeculoplasty, tubes, and trabeculectomy. Drug delivery is now helping us target drugs more precisely, avoid ocular surface complications, and sidestep compliance issues. And the third wave of glaucoma treatment promises to improve upon traditional transcleral approaches and provide the first noninvasive IOP-lowering therapies to help those patients with the most difficult-to-treat disease.
 |
|
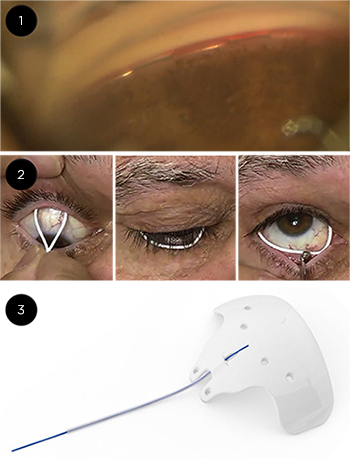
EXAMPLES. In the first wave of new technology, myriad MIGS devices became available, including (1) the Hydus implant. For master class on implanting the Hydrus, visit aao.org/master-class-video/hydrus-implant. In the second wave, sustained release devices are becoming available or are in development like (2) bimatoprost rings from Allergan. The third wave has brought transscleral approaches like (3) the Ahmed ClearPath.
|
DR. MARTIN ON PEDIATRIC OPHTHALMOLOGY
Advances in Amblyopia
In pediatric ophthalmology, the most high-profile developments are in myopia and in gene therapy (both mentioned later). However, an emerging new treatment for amblyopia may be just as transformative: dichoptic therapy.
Problems with traditional therapy. Until recently, first-line treatment options for amblyopia have focused on refractive correction with glasses, patching, or pharmaceutical penalization of the better eye, with successful protocols largely established by the Pediatric Eye Disease Investigator Group (PEDIG) investigations.
However, these methods—even when combined—do not always achieve ideal visual acuity outcomes, even when patient adherence is good, which is often not the case. About half of children treated with traditional methods from about age 3 to age 7 have some amblyopia at age 10, and older children demonstrate even greater degrees of residual amblyopia on long-term follow-up.1,2
Further, studies have shown that patching—the gold standard—increases the likelihood of bullying by 37%,3 suggesting that this type of treatment be completed before school age, as bullying may have long-term psychological consequences.4
Pharmacological approaches. Growing in popularity following the PEDIG studies, atropine 1% works as well as patching. However, potential side effects include glare, reversal of amblyopia, and even systemic side effects.5
Investigations are also under way to test pharmacological approaches, including levodopa-carbidopa, dopamine, and citocoline, some of which neurosensitize the brain, enhancing its responsiveness to traditional therapies.
We clearly need new options that are accessible, affordable, and acceptable for both children and adults, given that the prevalence of amblyopia is between 2% and 5%.2 Often considered untreatable in the past, adults are a prime target, particularly given that the lifelong chance of vision loss in the better eye is higher in amblyopes.6
Addressing a Binocular Problem
In a major step forward, amblyopia was recognized as a binocular problem—in that the stronger eye actively suppresses the weaker eye. Activating binocular neural circuits in patients with amblyopia has the potential to “awaken” an amblyopic eye in adults as well as children.1
Dichoptic treatment. A newer type of therapy, dichoptic training, gradually optimizes binocularity by presenting independent stimuli to each eye while the patient with amblyopia looks at a screen. The dominant eye may initially receive less contrast, which is adjusted over time so the nondominant eye improves with training.1
Benefits. Better tolerated than patching, binocular dichoptic training is suitable for anisometropic amblyopia, the most common type seen in clinical practice. The 2015 PEDIG study found that one hour a day was as effective as two hours a day of patching. Although adherence was initially good, children became bored and stopped using dichoptic training because it was limited to a single game, called Falling Blocks, which wasn’t stimulating enough to repeat daily.7
Coming soon? More engaging dichoptic therapies were needed to address this problem. In addition to iPad-based platforms, developers have introduced passive viewing of dichoptic movies, head-mounted virtual reality displays, and shutter glasses paired with dichoptic technology.1
In April 2020, Novartis acquired Amblyotech, a U.S.-based software company, to help pursue a digital therapy for amblyopia. (While I serve on the Amblyotech advisory board, my interest in this approach predates the company’s existence.) It uses active gaming and passive video technology with 3-D glasses and a dichoptic display that presents each eye with different images, gradually training the eyes to work together. With improvement, the lazy eye is forced to work more and more. In both children and adults, early clinical studies demonstrated faster visual improvements than with standard treatments.8,9
Another product called BinoVision (Medisim) offers dichoptic training with similar efficacy, but it uses goggles instead of glasses and manipulates streaming videos to perform audiovisual brain stimulation that increases blood flow and strengthens synaptic connections in the brain to improve the amblyopic eye’s response. It uses brightness/contrast oscillations and flickering, superimposed objects, and audio cues to encourage brain plasticity.10
Given the variety of viewing options available with both devices, chances are greater for maintaining the viewer’s interest and adherence. I predict that dichoptic training will reach its full potential in the near future, becoming a very viable treatment for amblyopia.
Other Growing Fields
Myopia treatment and retinal gene therapy are poised to become bigger areas of focus.
Myopia surge from pandemic? A change in lifestyle with children using computer screens from an early age is likely a key factor leading to the epidemic in myopia worldwide. With COVID-19 lockdowns, online learning has taken precedence, increasing still further the amount of time spent on screens.
More myopia treatment options. With an ophthalmology-driven evidence base, a wide range of treatments has become available to slow the progression of myopia, including outdoor activity, contact lenses, ortho-K lenses, peripheral defocus lenses and spectacles, multifocals, and bifocals. Following the ATOM1 and ATOM2 studies and LAMP study, low-dose atropine also gained acceptance as a pharmacological treatment for myopia.
Myopia treatment will become a larger and larger part of pediatric ophthalmology practice, requiring equipment to measure axial length accurately and to monitor the progression of myopia.
Retinal gene therapy. The diagnosis of retinal dystrophy is devastating not only for the child but also for the whole family. In the past, pediatric ophthalmologists had nothing to offer.
Now, gene therapy for Leber congenital amaurosis (LCA) with RPE65 has been approved by the FDA, as well as in Europe and, more recently, in Australia. Gene therapy for other congenital retinal dystrophies, including Stargardt disease, choroideremia, and retinitis pigmentosa, is also progressing and will pick up over the next five to 10 years, providing new hope for the future.
Pediatric and general ophthalmologists will need to learn how to recognize and diagnose these conditions, work with geneticists to counsel parents and children, familiarize themselves with available treatments, and refer appropriate patients to tertiary, high-level institutes that can perform the treatment. Retinal gene therapy will likely become a subspecialty within pediatric ophthalmology and ocular genetics.
___________________________
1 Kraus CL, Culican SM. Br J Ophthalmol. 2018;102(11):1492-1496.
2 Yen M-L. Taiwan J Ophthalmol. 2017;7(2):59-61.
3 Horwood J et al. Invest Ophthalmol Vis Sci. 2005;46(4):1177-1181.
4 Williams C et al. Br J Ophthalmol. 2006;90(6):670-671.
5 McLendon K, Preuss CV. StatPearls Publishing. 2020. www.ncbi.nlm.nih.gov/books/NBK470551/.
6 Rahi J et al. Lancet. 2002;360(9333):597-602.
7 Manh VM et al. Am J Ophthalmol. 2018;186:104-115.
8 Kelly KR et al. JAMA Ophthalmol. 2016;134(12):14020-1408.
9 Novartis press release. “Novartis acquires Amblyotech, pursuing novel digital therapy for children and adult patients with ‘lazy eye.’” www.novartis.com/news/media-releases/novartis-acquires-amblyotech-pursuing-novel-digital-therapy-children-and-adult-patients-%22lazy-eye%22.
10 BinoVision: “Binocular Treatment—Binovision.” www.bino-vision.com/for-eye-care-professionals/binocular-treatment-binovision/.
 |
|
DICHOPTIC TRAINING. Specialized glasses or goggles coupled with dichoptic movies or games that are designed to engage kids may become mainstream amblyopia treatment.
|
DR. MAA ON TECHNOLOGY
Ophthalmologic Telemedicine and Eye Care Delivery
COVID-19 has disrupted every level of society and raised many questions about social disparities, including health care inequities. As the pandemic continues, new safety standards to protect patients and staff have arisen, profoundly impacting both access to care and practices’ business strategies. A few illustrations: Patients may be reluctant to come to the physician’s office, even at the risk of significant vision loss; hospital systems may be looking for ways to reduce the number in-person visits to conserve PPE; and clinics may not be able to accommodate the same patient volume given social distancing requirements.
Telemedicine’s past. Telemedicine, while not new to ophthalmology, had been used only sparingly in ophthalmology prior to COVID-19. Ophthalmology typically used asynchronous telehealth, the most well-known program being diabetic teleretinal screening. It’s worth noting that store-and-forward telehealth is especially well-suited to ophthalmology because clinical practice is based on pattern recognition, and we routinely use photographs or other data reports such as Humphrey visual fields (HVF) or optical coherence tomography (OCT) images. We also routinely gather discrete data elements such as visual acuity and eye pressure, which can easily be forwarded for interpretation later. Yet, despite the suitability of eye care for telehealth, ophthalmologic telemedicine has traditionally been infrequently used due to barriers such as technological connectivity, licensure issues, and low reimbursement rates.
Ophthalmologic telemedicine’s future. Now is the perfect opportunity for ophthalmologists to incorporate telemedicine as part of their practice offering. Patient buy-in for telemedicine is at an all-time high because the quality of care is comparable to an in-person visit, and it can help keep them safe when accessing medical care.
Even though most ophthalmology visits cannot be done 100% virtually, we can still integrate synchronous and asynchronous telemedicine techniques to enhance practice and potentially reach patients who normally live too far from the clinic. Better yet, telemedicine has received payment equity, with CMS reimbursing telehealth visits at the same rate as face-to-face care, for the foreseeable future during the public health emergency.
Reaching more patients. There are many ways that telemedicine can be incorporated into an eye clinic. For example, a retina specialist may collaborate with a local comprehensive ophthalmologist and provide telehealth consults for select patients, a type of shared-care model used in Canada. Such an arrangement allows the patient to receive specialty care without having to travel to the retina clinic for evaluation, especially if a procedure is not required. The retina specialist can bill for the telemedicine consult visit and generate more referrals from their telemedicine partnerships for the patients who need procedures for face-to-face care. In addition, ophthalmologists can help alleviate some of the health care disparities exacerbated by the pandemic by bringing subspecialty care directly to high risk and poor access populations, such as rural or indigent patients.
Practices may begin utilizing telemedicine testing visits, where patients come to the clinic and receive testing such as HVF or OCT, to be interpreted by their provider later (teleglaucoma or telemacula). These models have been piloted successfully in the United Kingdom, even before the pandemic, to improve patient access to subspecialty care. This asynchronous visit can then be combined with a one-on-one video call or telephone follow-up for medical counseling. This hybrid telemedicine model means that patients spend less time in person in the clinic, but the ophthalmologist can still maintain a patient-provider relationship.
Remote-only care. Post-op follow-up one-on-one video visits for subspecialties like oculoplastics may be utilized more frequently than in other subspecialties because these post-ops do not usually require common procedures such as a slit-lamp exam. However, companies are developing sophisticated home-monitoring technologies for wet AMD conversion, home IOP checks, and home-based OCT. These technologies can be incorporated into a patient’s care plan, and the results can help prioritize which patients need in-person appointments and reduce frequency of unnecessary face-to-face follow-ups. Accurately identifying the highest risk patients will help facilitate social distancing and appointment access in the pandemic era.
Future directions. Moreover, research may create new tele-eye programs such as coverage for ophthalmic emergencies using telemedicine techniques for hospitals and clinics that don’t have access to ophthalmology on call. Finally, as the field of artificial intelligence (AI) and “big data” continues to evolve, the efficiency of interpreting asynchronous telemedicine data will increase, and risk stratification of patients has the potential to become more precise. One of my retina colleagues imagines using big data one day to provide evidence-based individualized follow-up, a definite future possibility with AI. In addition, big data in ophthalmology has the potential to help physicians in other medical specialties, for instance, by prediction of stroke risk through a combination of epidemiologic factors (smoking, race, age) and retinal vessel caliber in fundus photographs.
Conclusion. Telemedicine will play an increasingly important role in the pandemic era to provide quality eye care and improve access for patients. Use of telemedicine techniques, big data, and AI will ultimately benefit patients and reduce disparities for vulnerable patient populations over the near future.
The views expressed here are solely those of the author and do not in any form represent the views or policies of the VA or United States government.
Definitions
Telemedicine is a method of health care delivery in which the provider and the patient are separated by distance, time, or both. There are two broad categories:
Asynchronous (“store-and-forward”): Patient information is gathered at one time point and sent for interpretation at a second time point.
Synchronous: Provider and patient are connected at the same time using video and audio. Synchronous telemedicine can further be subdivided into two modes: one-on-one video/audio (e.g., Zoom call), or video exam facilitated by a tele-presenter.
- In the one-on-one video visit, the care is often delivered directly to the patient’s home, and the patient or family member is responsible for carrying out the physician’s instructions. For example, patients may be required to bring their eye to the phone camera so that the physician can see it.
- In a video exam facilitated by a tele-presenter, a trained individual conducts parts of the exam while supervised and directed by the ophthalmologist. The tele-presenter could be a nurse, a medical assistant, or even a primary care practitioner. The care may be delivered at the patient’s home, or the patient may receive care from an outside location (e.g., in the primary care clinic).
|
Meet the Experts
JOHN P. BERDAHL, MD Glaucoma and cornea specialist at Vance Thompson Vision in Sioux Falls, S.D. Relevant financial disclosures: Alcon: C; Allergan: C; Equinox: C,O,P,S; Glaukos: C; MicroOptx: C; New World Medical: C; Ocular Therapeutix: C; Kedalion: C; Sight Sciences: C.
APRIL Y. MAA, MD Associate professor of ophthalmology at Emory Eye Center and clinical director of TECS, Regional Telehealth Services, Veterans Integrated Service Network (VISN) 7, Department of Veteran Affairs, in Atlanta. Relevant financial disclosures: Regional Telehealth Services, Department of Veteran Affairs in Atlanta: E.
FRANK JOSEPH MARTIN, MD Clinical professor at the University of Sydney; visiting ophthalmologist at the Sydney Children’s Hospitals Network, Sydney Eye Hospital, and Royal Darwin Hospital; and chairman of the Board of the Children’s Medical Research Institute. Relevant financial disclosures: Novartis/Amblyotech: C.
Full Financial Disclosures
John P. Berdahl, MD Aerie.: C; Aerpio: C; Alcon: C; Allergan: C; Aurea: C,O; Avedro: C; Bausch + Lomb: C; Calhoun Vision.: C; Clarvista: C; DigiSight: C,O; Envisia: C; Equinox: C,O,P,S; Glaukos: C,O; Imprimis: C,P; iRenix: C; Johnson & Johnson: C; Kedalion: C; MicroOptx: C; New World Medical: C; Ocular Surgical Data: C,O; Ocular Theraputix: C; Omega Ophthalmic: C,O; Oyster Point: C,O; RxSight: C; Sight Sciences: C; ViaLase: C; Vittamed: C.
April Y. Maa, MD Click Therapeutics: C; Regional Telehealth Services, Department of Veteran Affairs in Atlanta: E. Warby Parker: C.
Frank Joseph Martin, MD Novartis: C.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|