Download PDF
This month, News in Review highlights selected papers from the original papers sessions at AAO 2020 Virtual. Each was chosen by the session chairs because it presents important news or illustrates a trend in the field. For details of presentation times, check the Virtual Meeting Guide at either aao.org/2020 or in the virtual meeting platform.
Artificial intelligence can now be used to help detect the most common cornea diseases. A deep learning algorithm, which uses anterior segment optical coherence tomography (AS-OCT), has been developed to simultaneously screen for keratoconus (KC), dry eye syndrome (DES), and Fuchs endothelial dystrophy (FED). In a study at Bascom Palmer Eye Institute in Miami, it performed nearly as well as cornea specialists.1
“This is the first deep learning multidisease algorithm for the autonomous diagnosis of common cornea diseases with significant morbidity and economic burden,” said Mohamed Abou Shousha, MD, PhD, at Bascom Palmer in Miami.
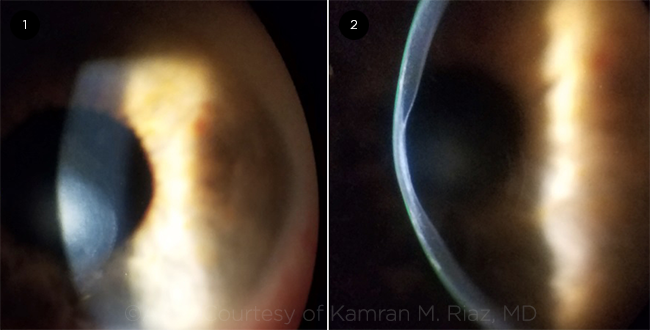 |
POSTERIOR KERATOCONUS. (1) Stromal opacity associated with (2) posterior excavation of the cornea in the visual axis.
|
Teaching the machine to classify. For this prospective study, the researchers created a dataset of 158,220 AS-OCT images. The researchers then trained the algorithm using 134,460 of those images; the images were obtained from 747 eyes that were either healthy or diagnosed with KC, DES, or FED. In addition, they tested the model in a second clinical study, using 23,760 images of 132 eyes that had not been diagnosed by the study’s cornea specialists.
High specificity and sensitivity. Overall, the algorithm was able to both accurately diagnose KC, DES, and FED and confirm the lack of cornea disease in healthy eyes. It had sensitivity values ranging from 93% to 100% and specificity values ≥96%.
However, while the algorithm was able to distinguish between diseased and healthy corneas, it didn’t always agree with the experts’ clinical diagnoses. It made seven diagnostic errors in the independent clinical study. Those included five diagnoses that confused dry eye and healthy eyes; one in which dry eye was misclassified as keratoconus; and one in which an eye with keratoconus was misclassified as healthy. This misclassification of keratoconus was the only error considered to be serious, said Dr. Shousha. However, the algorithm correctly diagnosed keratoconus in the other eye of the same patient, so the diagnostic error presumably would have been caught on referral.
A win-win. “These autonomous diagnostic algorithms can allow nonspecialists to detect disease with a sensitivity approximating that of a specialist,” said Dr. Shousha, who plans to file for FDA approval of the algorithm. “Using such algorithms translates into patients who will be diagnosed and referred early enough for care to prevent irreversible vision loss.”
—Miriam Karmel
___________________________
1 Shousha MA et al. Multidisease deep learning neural network for the diagnosis of corneal diseases. Presented at: AAO 2020 Virtual; Nov. 13-15, 2020.
___________________________
Relevant financial disclosures—Dr. Shousha: NEI: S; Research to Prevent Blindness: S; Resolve Ophthalmics: O. Related patents and PCT are owned by the University of Miami and licensed to Resolve Ophthalmics.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Nakazawa Canon: S; Daiichi-Sankyo: S; Kowa: S; Nidek: S; Rhoto Pharmaceutical: S; Santen: C,L,S; Senju Pharmaceutical: C,L,S: Tomey: S; Topcon: S; Wakamoto Pharmaceutical: S.
Dr. Riemann AGTC: S; Alcon: C,L,S; Alimera: C,LS; Alimera Deutschland: C,L; Allergan: L,S; Aniridia Foundation International: C; Animal Eye Institute: C; Aerpio: S; Bausch + Lomb/Valeant: C,L; BioTime/Lineage: C,S; BMC/Eyetube: C; Chengdu Kanghong: S; CSTLII: L; Chruman Research: O; Clearside: S; Clovernook Center for the Blind and Visually Impaired: C; CVP (CEI Vision Partners): O; Digital Surgery Systems: O; Gore: C; Genentech/Roche: S; Gyroscope: C, S; Haag-Streit: C; Haag-Streit Surgical: C; Haag-Streit USA: C,L,P; HumanOptics: C; IamC2: C,P; iVeena: C,O; Janssen /Johnson & Johnson: C,P,S; Kaleidoscope Engineering: C,P; Lowy-MacTel Registry: S; Med One: C,P; Macor Industries: O; Northmark Pharmacy: O; Neurotech: S; Nightstar/Biogen: S; Novartis: S; Notal Vision: C,S; Novartis: L; Opthotech/Iveric: S; Orbit BioMedical: C; Regeneron: L,S; Reliance Industries: C,L,P; SalutarisMD: C,L; Spark: S; TrueVision: C,L,P; VEO: O; Vortex Surgical: C,O,P.
Dr. Shousha NEI: S; Research to Prevent Blindness: S; Resolve Ophthalmics: O. Related patents and PCT are owned by the University of Miami and licensed to Resolve Ophthalmics.
Dr. Yu-Wai-Man GenSight Biologics: C.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|
More from this month’s News in Review