By Fouad F.E. Sayyad, MD, and Carol L. Karp, MD
Edited by Ingrid U. Scott, MD, MPH, and Sharon Fekrat, MD
Download PDF
Pigmented lesions that arise from the conjunctiva include nevus, complexion-associated melanosis (CAM), primary acquired melanosis (PAM), and malignant melanoma.1,2 All of these lesions arise from melanocytes. However, a number of other lesions have a similar appearance but a different source, such as pigment deposits from silver and iron.
It is essential to differentiate these lesions. An undiagnosed melanoma, for instance, can be deadly. This review provides an overview of presentation and management of melanocytic conjunctival pigmented lesions.
Initial Workup
Medical history. Patients should be asked about the following: 1) their age when they first noticed the lesion, 2) their history of sun exposure, 3) their personal or family history of skin cancer, and 4) any recently noted changes in the appearance of the lesion.
Exam. Features such as lesion color, thickness, and location are important in prognosis. Lesions on the bulbar conjunctiva are more common and apparent; they are also more easily detected by the patient than are those in the fornix. Meticulous examination of the conjunctiva by slit lamp is key, as it is easy to miss a small lesion in the fornix or tarsus. Always evert the eyelids.
With regard to imaging, optical coherence tomography (OCT) and ultrasound have been used to evaluate a variety of anterior segment lesions.3 OCT can be especially useful in differentiating amelanotic melanoma from ocular surface squamous neoplasia (OSSN). In OSSN, the epithelium is thickened, and there is an abrupt transition from the normal zone to hyperreflective thickened epithelium. In melanoma, the epithelium is usually thin, and a subepithelial lesion is present. Ultrasound can be used to assess the depth of the lesion and scleral invasion.
Nevi
Presentation. Nevi represent more than 50 percent of conjunctival lesions and are the most benign of the pigmented lesions. Nevi usually appear in childhood. Conjunctival nevi are very similar to those of the skin and are categorized as junctional, compound, or subepithelial. They are typically located on the interpalpebral conjunctiva, near the limbus, and they remain relatively stationary throughout life.
Conjunctival nevi can first appear as a nonpigmented sessile mass and become darker later on, especially with puberty or pregnancy. This increase in pigmentation may be misleading, as it may give a false impression that the lesion is growing. On slit-lamp examination, most nevi are well circumscribed with a cystic appearance.
Management. Nevi should be monitored regularly. When they are stable, this is usually yearly, but examinations may occur more frequently if warranted. A slit-lamp photo should be taken on the first visit for baseline purposes. If the nevus enlarges, or if there is increased vascularity, prompt evaluation is recommended. A lesion should be removed if there is a change in color or increase in size. Nevi may change with pregnancy or puberty but otherwise should remain stable.
(click to expand)
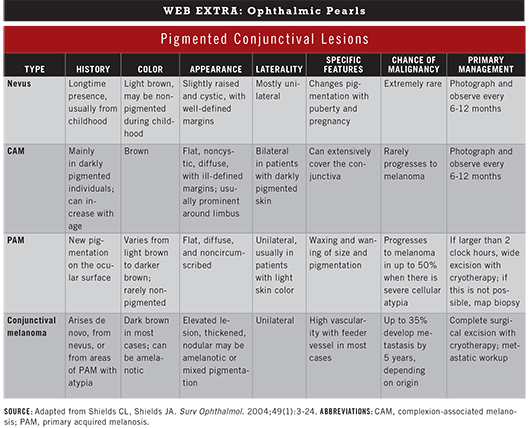
Complexion-Associated Melanosis
Presentation. CAM, also known as racial melanosis, is a benign lesion found among darkly pigmented individuals.
It is typically observed around the limbus. On examination, the pigmentation appears flat and noncystic. It can cover the conjunctiva extensively and increase in size with age; contrary to nevi and PAM, it is usually bilateral.1
Management. Although CAM has not been shown to progress to a melanoma, darkly pigmented individuals can develop melanomas, so yearly observation is recommended.
Primary Acquired Melanosis
Presentation. PAM is most likely to be observed in fair-skinned individuals who are middle-aged or older. On slit-lamp examination, a flat, unilateral, patchy, golden yellow to brown area of pigmentation is seen. Examination of the cornea may also reveal pigment.
PAM is almost always unilateral. The age at which the pigmentation was first noticed is important; if it is discovered at a young age, it is more likely to be a nevus. Furthermore, clinical examination can differentiate PAM from nevi. Nevi usually present with cysts and are well circumscribed. They are often thicker than PAM, as they have an epithelial and subepithelial component. PAM, on the other hand, is a diffuse intraepithelial disease, so it appears as a thin dusting of pigment and is usually not well circumscribed.
PAM with atypia can progress to malignant melanoma, while PAM with no atypia theoretically carries no risk. This differentiation requires histologic analysis.4
Management. If the lesion is small (occupying 1 or 2 clock hours), yearly follow-up is recommended unless change is noted. If any nodularity, thickening, or vascularity of the lesion is observed, a full excisional resection is recommended. For moderate-sized PAM (2 to 5 clock hours), lesions should be excised and margins treated with cryotherapy (see “Surgery” below). For large lesions (occupying more than 5 to 6 clock hours), thickened or suspicious areas should be removed. In addition, an incisional map biopsy of all quadrants is indicated, even for clinically uninvolved areas.
Rarely, PAM can occur without pigmentation. Further treatment depends on the histologic diagnosis. Medium and larger lesions are treated more aggressively, as the risk of melanoma conversion is directly related to the area of the pigmented lesions.5
Surgery. Excision with cryotherapy is the preferred treatment for PAM with atypia. The lesion is removed with a tumor-free margin of 4 to 5 mm. Double-freeze, slow-thaw cryotherapy is applied to the conjunctival edges. If the pigmentation extends onto the cornea, we recommend application of absolute alcohol intraoperatively for one minute and then epitheliectomy.
For diffuse disease that cannot be excised, cryotherapy or topical chemotherapy may be employed. Double-freeze, slow-thaw cryotherapy, the technique used at the margins of excisions, may also be used for cases in which the pigmentation cannot be excised, such as diffuse pigment on the tarsal conjunctiva. Topical chemotherapy is not used as a primary therapy for pigmented lesions. It can, however, be used postoperatively when the lesion is so extensive that it cannot be excised. The most commonly used topical drug is mitomycin C 0.02 percent or 0.04 percent. The typical cycle is treatment four times a day for one week, followed by a pause of one to two weeks and then by another treatment cycle. Punctal plugs are used, and the cornea is managed for toxicity with artificial tears and topical steroids.
The entire treatment-pause-treatment cycle is repeated until the pigment resolves. This usually requires two to three cycles.
Malignant Melanoma
Presentation. Patients are typically 60 to 70 years old and present with a nodular mass arising either de novo, from a nevus, or from PAM with atypia.5 The most common location of these tumors is the limbus, but they also can appear in the caruncle, tarsus, and fornix. Nonlimbal locations portend a poorer prognosis. A raised mass, often accompanied by feeder vessels, may be observed.
Management. If malignant melanoma is suspected, incisional biopsy is avoided to prevent tumor seeding. The mass is excised, using a dry, no-touch technique6 with a safety margin zone of 4 to 6 mm. After excision, double-freeze, slow-thaw cryotherapy is applied to the conjunctival margins and limbus. Alcohol is used for the corneal epithelium to debride abnormal cells. We use a sclerectomy when the tumor is adherent to the sclera, and we apply cautery and cryotherapy to the base of the lesion. With clean instruments, an amniotic graft or primary closure is used to cover the defects. Histopathology helps to determine the prognosis. Sentinel node biopsy should be considered for lesions larger than 2 mm or have high-risk factors on histopathology.
Prognosis. Lesion origin is a critical factor: De novo melanomas tend to have the worst prognosis.5 Other factors predictive of poor survival include older age, male gender, nonwhite race, and tumors with nodularity or ulceration. Local recurrence is common and can be 45 percent at five years and 59 percent at 10 years. Melanoma-related death rates are 5 to 17 percent at five years and 9 to 35 percent at 10 years, depending on the precursor lesion.5
___________________________
1 Shields CL, Shields JA. Surv Ophthalmol. 2004;49(1):3-24.
2 Oellers P, Karp CL. Ocul Surf. 2012;10(4):251-263.
3 Bianciotto C et al. Ophthalmology. 2011;118(7):1297-1302.
4 Jakobiec FA et al. Ophthalmology. 1989;96(2):147-166.
5 Shields CL et al. Ophthalmology. 2011;118(2):389-395.
6 Shields CL et al. Am J Ophthalmol. 2001;132(4):576-578.
___________________________
Dr. Sayyad is a research associate, and Dr. Karp is professor of ophthalmology; both are in the department of ophthalmology at Bascom Palmer Eye Institute. The authors have no personal financial interests to declare.