Download PDF
Antitubercular therapy (ATT) has been shown to be effective in reducing recurrences of tubercular uveitis in nearly 86% of patients.1 Yet some physicians remain reluctant to initiate treatment.
“Ocular tuberculosis remains a challenge all over the world, as the diagnosis is largely presumptive due to lack of positive histopathologic confirmation,” said Vishali Gupta, MD, at the Advanced Eye Centre in Chandigarh, India. “In several countries, ophthalmologists have to refer these patients to infectious disease experts or physicians who refuse to start ATT for lack of confirmed diagnosis. This can lead to multiple recurrences [of disease], resulting in increased ocular morbidity and visual loss,” said Dr. Gupta.
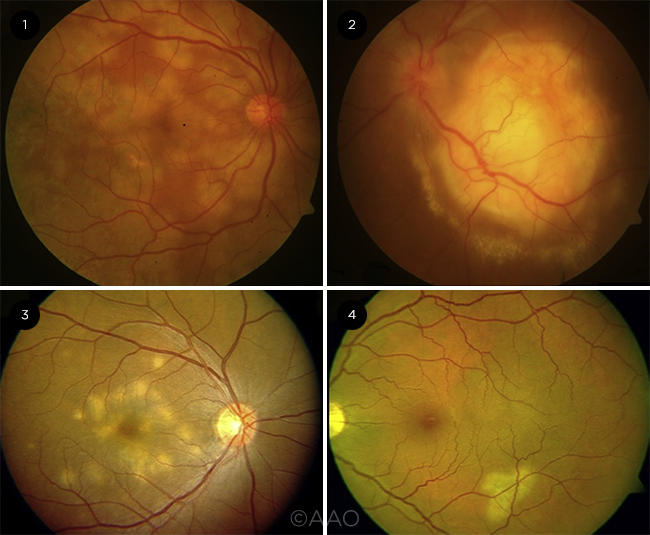 |
TUBERCULAR CHOROIDITIS. The spectrum of choroidal involvement ranges from (1) tubercular serpiginous-like choroiditis to (2) tubercular multifocal choroiditis, (3) tuberculoma, and (4) tubercular unifocal choroiditis.
|
Enter consensus guidelines on initiating ATT. To limit confusion, a team of international experts from the Collaborative Ocular Tuberculosis Study (COTS) has issued consensus guidelines on initiating ATT in several clinical scenarios.2 The guidelines suggest that clinicians take the following steps:
- First, ascertain whether the clinical presentation in the eye is suggestive of TB.
- Second, consider whether the patient lives in a TB-endemic region (defined as having an incidence of more than 100 cases per 100,000 persons). Consensus was more robust for endemic regions.
“It is also important to rule out other possible etiologies in the differential diagnosis, as TB can mimic several other varieties of uveitis,” both infectious and noninfectious, Dr. Gupta said.
- Once ocular TB is suspected, order an immunologic test—a tuberculin skin test and/or an interferon-gamma release assay.
- In the three subtypes of tubercular choroiditis—tubercular serpiginous-like choroiditis, tuberculoma, and tubercular focal or multifocal choroiditis—any positive immunologic test plus radiologic signs of active or healed pulmonary tuberculosis justifies initiation of ATT.
- In endemic regions, a positive result from a single immunologic test is sufficient to initiate treatment of tubercular serpiginous-like choroiditis or tuberculoma, even without radiologic features suggestive of tuberculosis.
When to use adjunctive therapy for inflammation. There is strong agreement to start oral corticosteroids with, or soon after, initiation of ATT in patients who have tubercular serpiginous-like choroiditis or tuberculoma (with no associated systemic infectious disease). But opinion is mixed regarding the timing of initiating oral corticosteroids in patients with tubercular multifocal or unifocal choroiditis.
And a caveat. Beware of potential drug interactions when combining ATT with various immunosuppressive drugs. When in doubt, consult the patient’s internist.
Impact on practice. Dr. Gupta now has more confidence in making decisions about initiating ATT, particularly in borderline situations. “Earlier, I was not sure whether or not I should start ATT. But after this consensus, I have started treating these patients, even though only one test is positive,” she said. “The guidelines have made a difference in my practice patterns.”
—Miriam Karmel
___________________________
1 Agrawal R et al. JAMA Ophthalmol. 2017;135(12):1318-1327.
2 Agrawal R et al. Ophthalmology. Published online Jan. 10, 2020.
___________________________
Relevant financial disclosures—Dr. Gupta: None.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Feng None.
Dr. Gibbons None.
Mr. Glassman None.
Dr. Gupta None.
Dr. Koch Alcon: C; Carl Zeiss Meditec: C; CapsuLaser: O; Johnson & Johnson Surgical Vision: C; Ivantis: O; Perfect Lens: C; Vivior: O.
Dr. Leung None.
Mr. Robbins None.
Dr. Wygnanski-Jaffe GoCheck Kids: C; NovaSight: C.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|
More from this month’s News in Review