Download PDF
With the publication of the BEAT-ROP study results in 2011, some clinicians were inclined to jump on the bevacizumab bandwagon, while others held back, citing the well-documented success of laser.
Randomizing infants to receive either retinal laser or intravitreal anti-VEGF therapy, the BEAT-ROP (Bevacizumab Eliminates the Angiogenic Threat of Retinopathy of Prematurity) study, published in the New England Journal of Medicine, found a lower rate of recurrent neovascularization for zone I ROP in patients receiving Avastin compared with laser.1
"That's a big reason there s been so much enthusiasm for anti-VEGF therapy,” said David K. Wallace, MD, MPH, at Duke Eye Center in Durham, N.C. “But that study was not ideal, and recurrent neovascularization is not the most relevant outcome. We need additional studies to assess the relative effectiveness of these two types of treatment and to assess the safety of anti-VEGF therapy in these fragile premature infants."
Michael T. Trese, MD, at William Beaumont Hospital in Detroit, couldn't agree more. BEAT-ROP does not provide the level of evidence offered by the ETROP (Early Treatment for Retinopathy of Prematurity) study, he said, where ablation reduced unfavorable structural outcomes from 15.6 to 9.0 percent at 9 months.2
In addition, confounders might have skewed the BEAT-ROP results, said Dr. Trese. For example, 67 percent of the patients in the BEAT-ROP study were Hispanic. “For reasons we don't entirely understand, ROP is more difficult to treat in these patients,” he said. "They may require more than one laser treatment but the BEAT-ROP protocol did not allow this."
The visual outcomes and benefits of laser have been validated in multicenter clinical trials over the years. Thus far, however, BEAT-ROP is the only randomized, comparative anti-VEGF trial that has produced results. So where does this leave clinicians today? Five ROP experts weigh in.
Advantages of Laser Therapy
The most commonly used ROP treatment, peripheral laser ablation (Fig. 1) burns the peripheral retina to stop neovascularization in its tracks. For doctors like Dr. Trese, laser is still the treatment of choice, with anti-VEGF therapy playing a secondary role. Following are some advantages cited for laser.
One and done. With transpupillary laser, the disease typically regresses and doesn’t come back, said Richard A. Saunders, MD, at Medical University of South Carolina in Charleston. “If laser is applied at an appropriate time,” he said, “the success rate at our institution is well over 90 percent—at least with regard to avoiding retinal
detachment.”
Finite follow-up. This translates into a well-defined follow-up period. “With a peak incidence of treatment at 35 to 36 weeks postmenstrual age,” said Dr. Trese, “I can follow a child to 50 weeks and be very confident they won’t develop a retinal detachment, if they haven’t already.”
Long-term effects. During their life times, people born prematurely have an approximately 14 percent risk of retinal detachment from a retinal tear, compared with 1.5 to 3.0 percent in the general population, he said. “By doing peripheral laser treatment, we may provide lifelong protection from development of those tears.”
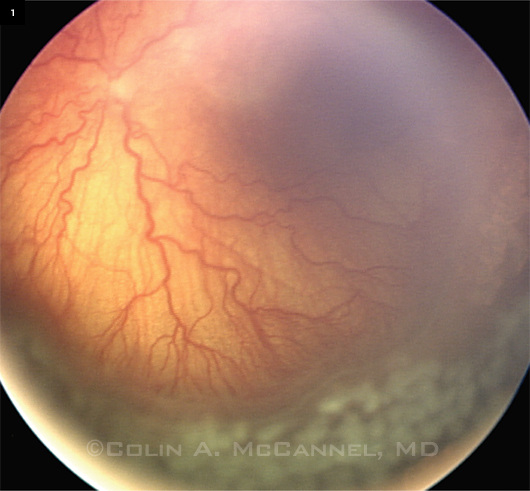 |
|
PERIPHERAL LASER. Wide-angle view of fundus after laser photocoagulation for ROP with plus disease.
|
Disadvantages of Laser Therapy
Laser therapy is not easy to learn, said Dr. Wallace. Some pediatric ophthalmologists—even after fellowship training—are not completely comfortable using a laser on infants with ROP, in either the neonatal intensive care unit (NICU) or operating room (OR).
NICU. Some institutions require that laser be delivered in the OR under general anesthesia, while others use sedation in the NICU, said Michael B. Yang, MD, at Cincinnati Children’s Hospital Medical Center. “But some neonatologists don’t like the fluctuations in oxygen saturation and alertness that occur with sedation, which requires that the dose of sedating medication be closely monitored.” With the procedure lasting one to one and a half hours, added Dr. Wallace, sedation also can be stressful to an infant.
OR. Despite challenges like these, some experts believe that sedation puts less wear and tear on the child than general anesthesia does. “In my opinion, the most underappreciated disadvantage of laser therapy is the need for general anesthesia at many institutions,” said Dr. Saunders, who implicates general anesthesia in a few cases of cardiopulmonary arrest occurring within 24 hours after his laser treatment.
“Multiple anesthesias in young kids may also be correlated with developmental issues later in life,” he said. “In addition, when children have been intubated for laser treatment, there is no assurance they will be successfully extubated once they’ve emerged from anesthesia.”
Outcomes and other risks. In roughly 5 percent of laser cases, treatable areas of retina may be missed, said Dr. Yang, which then requires retreatment. “And, with type 1 zone I ROP,” he added, “the risk of an unfavorable retinal structural outcome after laser may be nearly 30 percent, according to ETROP.”
“With posterior disease,” said Dr. Saunders, “you can easily ablate two-thirds of the retina. This can restrict the peripheral visual field, cause inflammation, and lead to specific complications such as anterior segment ischemia or, more rarely, cataract formation from the laser itself.”
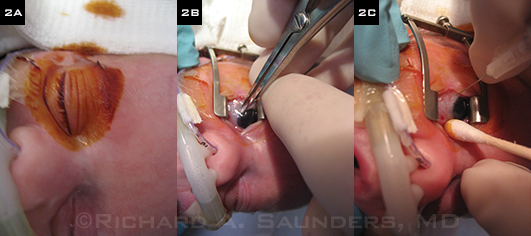 |
|
BEVACIZUMAB STEPS. (2A) Area is thoroughly prepped with povidone-iodine. (2B) Determining injection site with caliper. (2C) Preparing for injection.
|
Advantages of Anti-VEGF Therapy
VEGF plays a significant role in both the ischemic and vasoproliferative phases of ROP, and treating with anti-VEGF therapy offers certain advantages over laser.
Ease of use. Done at the bedside in less than five minutes, often without intubation, intravitreal injection offers ease of administration (Fig. 2), said Dr. Wallace, and it’s a technique most residents are learning with adult patients during their training. Treatment can be done at your convenience with modest preparation, staff time, and effort, added Dr. Saunders. All of these factors may make anti-VEGF therapy particularly attractive in developing countries, especially where ophthalmologists may not have easy access to an appropriate indirect laser delivery system.
Rapid response. It may take a week for laser treatment to stop progression of disease, but the antibody binds the VEGF inside the eye almost immediately, said Dr. Wallace. “By the time we look again—usually the next day—we see a marked response that we typically don’t see until much later with laser.”
Better visual fields? BEAT-ROP and other studies have shown that in patients with type 1 zone I ROP, the abnormal blood vessels regress, and the retina then undergoes more normal peripheral vascularization, said Dr. Yang. (See Fig. 3.) This offers the potential for larger visual fields with anti-VEGF than with laser, although it is not yet known how functional these areas of peripheral retina may be.
Visual acuity. Another potential advantage concerns long-term visual acuity. “Two-year follow-up data from the BEAT-ROP study show a striking reduction in the amount of myopia,” said Dr. Wallace. Lower myopia was also seen in a nonrandomized study conducted by investigators at Baylor College of Medicine. They found that mean myopia in children treated with anti-VEGF therapy was –0.9 D, compared with –4.4 D in the laser group. This could be significant, he said. In his 19 years of practice, Dr. Wallace has followed former premature patients into their teen years and has seen many of them struggle with very high levels of myopia—some with even more than 20 D.
Prospects for Prevention
Although preventing prematurity would be the best way to eliminate ROP, that goal remains elusive. Meanwhile, said Dr. Smith, “If we could completely replicate the intrauterine milieu, we wouldn’t have any complications of prematurity. Therefore, we’re trying to determine the critical factors so we can mimic this environment, if possible.” Some of the areas of current research include the following.
Inositol. Although its mechanism of action is not entirely clear, preliminary studies conducted by Dale Phelps, MD, have shown that inositol, a sugar, may help prevent ROP, said Dr. Wallace.
Omega-3s. “We’ve shown causality in a mouse model looking at omega-3 polyunsaturated fatty acids helping retinopathy,” said Dr. Smith. “There have also been a couple of studies looking at babies with ROP suggesting that a more normal level of omega-3 fatty acids is associated with better ROP outcomes.”
IGF-I. In a mouse model, Dr. Smith and colleagues also have found that insulin-like growth factor I (IGF-I) is required for normal vascularization of the retina. However, after preterm birth, infants undergo a drastic loss of IGF-I. “We found that this was very highly correlated with the development of ROP,” she said. “In fact, the lower the levels of IGF-I after preterm birth, the higher the risk.” Postnatal weight gain may play a critical role, said Dr. Yang, as the infant needs to gain enough weight after birth to produce adequate levels of IGF-I for normal vascularization.
Preliminary pilot data show that supplementation of IGF-I to normal intrauterine levels may help prevent ROP, said Dr. Smith, who is planning another trial in this area.
Light before birth. Although Dr. Yang said it’s “not ready for prime time,” he has conducted early studies in mice and infants suggesting that the amount of light positively affects the development of the retina in utero.1,2 He and Dr. Smith are collaborating to further study the role of light exposure.
__________________________
1 Rao S et al. Nature. 2013;494(7436):243-247.
2 Yang MB et al. Ophthalmology. 2013;120(12):2706-2713. |
Disadvantages of Anti-VEGF Therapy
Delayed vascularization. “The consensus is that anti-VEGF therapy decreases neovascularization and can work quite quickly,” said Lois Smith, MD, PhD, at Harvard Medical School. “However, it tends to increase the length of time required for normal vascularization of the retina.” This means there is avascular retina in the periphery, putting children at risk of disease long after they are discharged home, she added.
Recurrence. Perhaps 20 percent of these kids have recurrent disease that comes back weeks or even months after treatment, said Dr. Saunders. “It is not atypical to have recurrences in children who are 60 to 70 weeks postmenstrual age—or longer. If you’re going to treat with Avastin, you have to be prepared to monitor for a long time.”
Challenging follow-up. Once these babies have gone home, it isn’t easy to follow them, said Dr. Wallace. “These are also bigger babies, and it’s hard to see the retinal periphery well in the office without anesthesia.” Nobody really knows the ideal follow-up schedule for a child treated with anti-VEGF therapy, added Dr. Trese. Opinions abound, with some clinicians comfortable following up every month, said Dr. Yang, while others examine more frequently.
A double-edged sword. One of the more controversial issues involves establishing the right balance of VEGF in an infant at risk for ROP. “The biochemistry of a developing child is quite different from that of an adult,” said Dr. Trese. “For example, VEGF plays many positive roles related to neural, vascular, and lung development.”
Long-term effects: unknown. “We really do not know for sure that this drug is safe in these infants,” said Dr. Saunders. His pilot studies comparing developmental measures after both laser and Avastin therapy have not yet turned up evidence of harm. “But that’s a far cry from saying we have evidence that it does not cause problems.”
Systemic issues are very difficult to define in children who have many comorbidities, such as bronchopulmonary dysplasia, said Dr. Trese. In addition, systemic changes may not be apparent until children are well into their teens. “For these reasons,” he said, “it would take a very well-structured, randomized, prospective controlled clinical trial to sort this all out.” Although the five deaths in the anti-VEGF arm of the BEAT-ROP trial did not reach statistical significance compared with the laser arm, he added, it’s concerning that four of the five were reported as respiratory deaths.
Uncertainties about drug and dose. From a cost perspective, Avastin was a logical first choice, but for this patient population, other anti-VEGF therapies may be preferable. Bevacizumab has been shown to leak into the systemic circulation, suppressing systemic VEGF at least two weeks after intravitreal injection, which may cause changes in brain and other organ development in very preterm infants.3 However, ranibizumab (Lucentis) has a shorter half-life, said Dr. Saunders, which might reduce possible systemic effects.
The optimal anti-VEGF dose has not yet been determined. The dose used in BEAT-ROP was 0.625 mg—half of that typically used in adults with age-related macular degeneration. “There are estimates that this dose may provide up to 10,000 times as much anti-VEGF antibody as VEGF present on average in the vitreous of these infants,” said Dr. Smith.
Dr. Wallace is planning dosing studies for Avastin. “Instead of doing a typical escalating dose study, we will de-escalate the dose of Avastin,” he said, to avoid a starting dose that is too low to be effective at halting disease progression. Lucentis dosing studies are being planned in Germany by Tim U. Krohne, MD, at the University of Bonn, and Andreas Stahl, MD, at the University of Freiburg. Finding the lowest effective dose of anti-VEGF will be important to both minimize systemic effects and promote normal vascularization of the retina.
Other risks. “Endophthalmitis is not a good thing in adults, but in an infant, it could be a fatal event,” warned Dr. Saunders. To reduce the risk level “to as close to zero as possible,” he takes precautions such as ensuring that the area is completely prepped with Betadine (Fig. 2A) and that topical antibiotics are used before and after the injection. He noted that the small risk of hitting the lens with the needle during injection is also a concern.
ROP Classification: A Primer
With numbers representing zones, stages, and types, the ROP classification system can be confusing for physicians who don’t use it regularly. Here’s a brief refresher course to untangle the terminology. For a more detailed description, see the revised International Classification of Retinopathy of Prematurity.1
Location of disease involvement is described in zones.
- Zone I: Innermost circle; radius extends from the middle of the optic disc to twice the distance from the disc to the macula
- Zone II: Area extending centrifugally from the edge of zone I to the nasal ora serrata
- Zone III: The peripheral, temporal crescent of retina
Extent is expressed in clock-hours of involvement.
Severity is indicated by the following stages.
- Stage 1: Presence of line of demarcation between vascularized and nonvascularized retina
- Stage 2: Ridge of demarcation that rises above the retinal surface; in addition, small, isolated areas of neovascularization may be present on the retinal surface
- Stage 3: Extraretinal fibrovascular proliferation or neovascularization that extends from the ridge into the vitreous
- Stage 4: Partial retinal detachment
- Stage 5: Total retinal detachment
Plus disease denotes the presence of increased venous dilatation and arteriolar tortuosity; later signs include iris vascular engorgement, impaired pupil dilation, and vitreous haze. Plus disease may occur at various stages and is notated with a plus sign—for example, stage 3+ ROP.
Type. Although not part of the original classification schema, this was added based on findings from the ETROP study2 to help determine whether to treat or to observe.
Type 1 represents higher-risk disease.
Type 1 is defined as any of the following:
- Zone I, any stage ROP with plus disease
- Zone I, stage 3 ROP without plus disease
- Zone II, stage 2 or 3 ROP with plus disease
Type 2 is defined as any of the following:
- Zone I, stage 1 or 2 ROP without plus disease
- Zone II, stage 3 ROP without plus disease
__________________________
1 International Committee for the Classification of Retinopathy of Prematurity. Arch Ophthalmol. 2005;123(7):991-999.
2 Early Treatment for Retinopathy of Prematurity Cooperative Group. Arch Ophthalmol. 2003;121(12):1684-1694. |
Anti-VEGF Consensus to Come?
Given these many considerations, where does anti-VEGF therapy fit into these experts’ treatment plans?
Not primary. “I frankly don’t use anti-VEGF therapy as a primary therapy,” said Dr. Trese. “I envision it making the most sense in a child still at risk for retinal detachment and blindness after use of the safer peripheral laser ablation,” he said. “Anti-VEGF therapy is also a rational approach in cases where laser treatment would put foveal development at risk.” After first trying laser, Dr. Trese would also be more inclined to consider anti-VEGF therapy for Hispanic patients, who appear to have more recalcitrant disease.
Favored. Considering the appeal of pharmacologic treatment among physicians and parents, Dr. Saunders does not think laser will remain the gold standard indefinitely. “Although not an early proponent of anti-VEGF therapy, I’ve come to favor it, particularly in posterior or asymmetric disease, with certain important reservations,” he said. “I offer parents both types of treatment, explaining in some detail the risks—both known and unknown—and almost everybody chooses Avastin.”
Depends on status. At Dr. Yang’s institution, the policy thus far has been to follow the recommendations of the BEAT-ROP study, offering Avastin only to patients who have type 1 zone I disease. “Some parents choose laser, and others choose Avastin,” said Dr. Yang.
“It may be that infants with the most posterior, severe, rapidly progressing disease will be the most likely to benefit from Avastin,” said Dr. Wallace, “perhaps followed by an additional injection or laser, if needed, after blood vessels have had a chance to grow more into the periphery.” On the other hand, he doubts that other therapies will supplant laser for treatment of zone II ROP, where it is remains very successful.
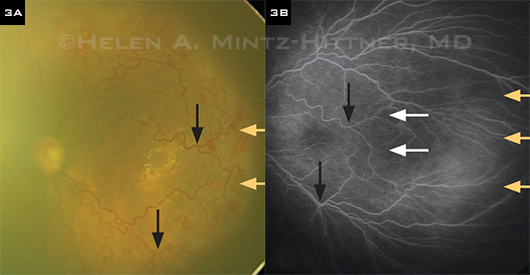 |
|
TREATMENT EFFECT. (3A) Pretreatment temporal fundus photo of left eye of infant at age 2.5 months (born at 24 weeks gestational age) with zone I aggressive posterior ROP. (3B) FA at 13 months after intravitreal bevacizumab monotherapy shows further vascularization of the peripheral retina. Black arrows indicate identical retinal points; yellow arrows show the temporal extent of retinal vessels; white arrows show the extent of the retinal vessels before treatment.
|
Combinations. In the future, Dr. Wallace thinks that laser and anti-VEGF therapies may be used increasingly in combination for ROP. Dr. Smith agreed that anti-VEGF therapy for ROP may find its niche as a rescue therapy. “The question is,” she said, “what’s the best place for it—after we treat with laser, or before?”
Earlier, better. Ideally, she said, we could treat earlier and with a lower dose—before reaching the point of plus disease and a higher risk for neovascularization. “This would work if we had a way to determine that VEGF levels were starting to go up. We could treat to bring levels back to normal, repeating injections, if needed, and with an anti-VEGF molecule that did not leak into the systemic circulation.”
__________________________
1 Mintz-Hittner HA et al. N Engl J Med. 2011;364(7):603-615.
2 Good WV; on behalf of the Early Treatment for Retinopathy of Prematurity Cooperative Group. Trans Am Ophthalmol Soc. 2004;102:233-250.
3 Sato T et al. Am J Ophthalmol. 2012;153(2):327-333.e1.
Meet the Experts
RICHARD A. SAUNDERS, MD Professor of ophthalmology and clinical vice chair of the department of ophthalmology at the Medical University of South Carolina in Charleston. Financial disclosure: None.
LOIS SMITH, MD, PHD Professor of ophthalmology at Harvard Medical School and associate in ophthalmology at Children’s Hospital Boston. Financial disclosure: None.
MICHAEL T. TRESE, MD Clinical professor of biomedical sciences at the Eye Research Institute at Oakland University, clinical associate professor at Wayne State University School of Medicine, and chief of pediatric and adult vitreoretinal surgery at William Beaumont Hospital in Rochester, Mich. Financial disclosure: Consults for Focus ROP, Genentech, Nu-Vue Technologies, Retinal Solutions, and Synergetics.
DAVID K. WALLACE, MD, MPH Professor of ophthalmology and pediatrics and director of clinical research at Duke Eye Center in Durham, N.C. Financial disclosure: Consults for Allergan.
MICHAEL B. YANG, MD Associate professor of ophthalmology at the University of Cincinnati College of Medicine and Abrahamson Pediatric Eye Institute, Cincinnati Children’s Hospital Medical Center. Financial disclosure: None. |