By M. Tariq Bhatti, MD, with Larry Frohman, MD, and Gideon Nesher, MD
Download PDF
Giant cell arteritis (GCA) remains a baffling disease in terms of its cause and diverse clinical manifestations. Many patients with GCA have ocular or visual problems. Here, in part 1 of a 2-part series, M. Tariq Bhatti, MD, of the Duke Eye Center and Duke University Medical Center, Durham, North Carolina, hosts an MD Roundtable with Larry Frohman, MD, of Rutgers–New Jersey Medical School, and rheumatologist Gideon Nesher, MD, of Shaare-Zedek Medical Center and Hebrew University in Jerusalem. The group discusses GCA in terms of clinical manifestations and diagnostic approaches. Part 2 will appear in the July EyeNet.
Impact of GCA
Dr. Bhatti: Dr. Frohman, you recently published an excellent article addressing GCA.1 Can you discuss why ophthalmologists need to be aware of this disease?
Dr. Frohman: GCA is a potentially blinding disease, and it can cause strokes and heart attacks. Your initial management of GCA may determine whether the patient will ultimately see.
For patients with systemic symptoms but without visual loss, a large number will lose vision in at least 1 eye if they remain undiagnosed and untreated. For patients already with visual loss, the emphasis is on preventing further ischemic complications of the disease, including the most dreaded one: blindness in the second eye. A patient with undiagnosed, untreated GCA who has already suffered ischemic optic neuropathy in 1 eye has a chance of blindness in the second eye of somewhere between 20% and 62%. Recently, the risk was cited as even higher: 54% to 95%.2
Our main goals are to avoid second-eye involvement, which can occur rapidly, and to prevent further visual loss. In some acute cases of choroidal ischemia, if the patient is seen relatively early and steroids are initiated, some improvement in vision may be possible. However, steroid treatment usually doesn’t restore vision.
 |
|
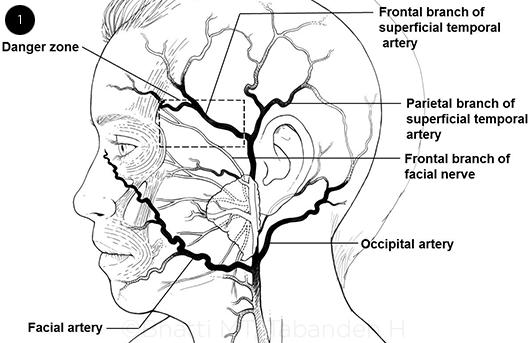
STEP BY STEP. (1) Anatomy of the superficial temporal artery and its branches. The biopsy should be performed ipsilateral to the side of visual symptoms and within an affected segment of artery as manifested by tenderness, decreased pulsation, or nodularity. Preoperatively, the course of the artery is identified and marked by either manual palpation or Doppler ultrasonography. It is preferable not to select a segment of artery within the danger zone (boxed area), which carries the greatest risk of inadvertent injury to a branch of the facial nerve.
|
Ocular Manifestations
Dr. Bhatti: What are some of the ocular manifestations of GCA? What should the ophthalmologist be looking for when an elderly patient comes in with visual problems or concerns?
Dr. Frohman: If a patient who is 50 years or older presents with diplopia, central retinal artery occlusion, or amaurosis fugax without visible emboli, and/or ischemic optic neuropathy, ophthalmologists must immediately consider GCA as a potential cause.
The best-recognized ocular manifestation of GCA is visual loss, and the most common form is ischemic optic neuropathy. Typically, we usethe term “arteritic anterior ischemic optic neuropathy” to differentiate from rare posterior cases. In anterior cases, acute changes in the fundus are observable, most classically a pallid swollen disc.
At least 2 other forms of visual loss may occur in a patient with GCA: arterial occlusion (central retinal or branch retinal) and choroidal ischemia. Research suggests that 10% of cases of ischemic visual loss in GCA actually result from retinal artery occlusion, not from disc infarction.
Although choroidal ischemia is associated with arteritic ischemic optic neuropathy, the incidence of choroidal ischemia causing visual loss in the absence of disc infarction is not well established in the literature.
In a review and metaanalysis, Goodman3 found that both amaurosis fugax and diplopia were seen in 12% of patients with GCA. Approximately 30% of patients with permanent visual loss have had antecedent amaurosis fugax. Although double vision from third, fourth, and sixth nerve palsies may occur in GCA, most cases of diplopia are from ischemia of the muscles due to vasculitic involvement. Many less common ocular manifestations of GCA also have been described, including ocular ischemic syndrome, Horner syndrome, and orbital inflammation.
 |
|
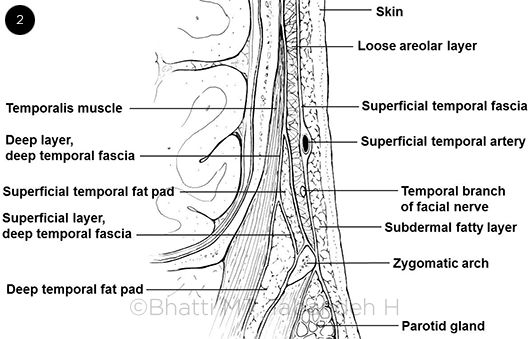
STEP BY STEP. (2) Cross-sectional view of the scalp at the level of the zygomatic arch illustrating the neurovascular anatomy and fascial planes. Branches of the superficial temporal artery course on top of the superficial temporal fascia. In comparison, branches of the facial nerve lie beneath the plane of the superficial temporal fascia.
|
Systemic Manifestations
Dr. Bhatti: What are some of the systemic manifestations of GCA that ophthalmologists should be asking about or looking for?
Dr. Frohman: I ask patients about anorexia and temporal or other scalp tenderness. In Goodman’s metaanalysis,3 he noted a 67% incidence of headache and a 47% incidence of polymyalgia rheumatica (PMR). He also found that a quarter of patients with GCA have depression. I’ve had patients describe onset of melancholy that started around the same time as other symptoms.
Goodman3 also noted a 36% incidence of jaw claudication in GCA. A few patients with GCA have presented to me with tongue claudication or trouble chewing or talking. Some have said that their tongue cramped while they were on the phone, and they suddenly couldn’t speak. I think this is a very specific sign.
In a retrospective review, Gonzalez-Gay et al.4 determined that roughly 7.5% of patients with GCA do not have many symptoms. If a patient presents with a visual manifestation suggestive of GCA but without overt systemic symptoms, the ophthalmologist should not be misled into excluding GCA as a possible cause.
Dr. Bhatti: These are all excellent points. The only comments that I can add are that tongue infarction is considered pathognomonic for GCA and that it is important for ophthalmologists to ask patients about their general health as well as their visual-ocular symptoms.
 |
|
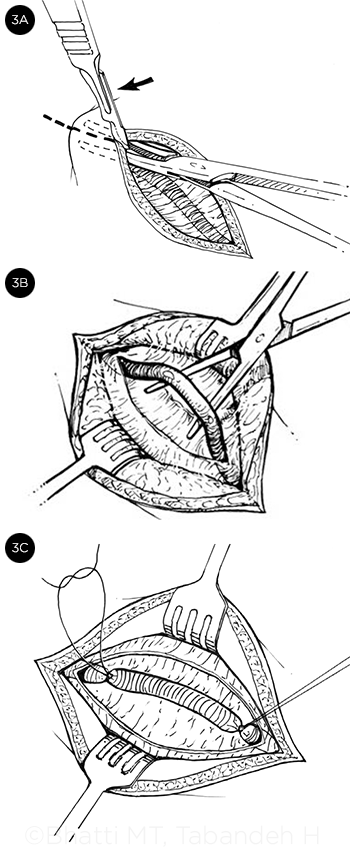
STEP BY STEP. (3) Surgical technique for isolating a branch of the superficial temporal artery. A) The skin incision is made parallel to the direction of the artery. Using a hemostat, the plane above the superficial temporal fascia is entered and the incision is completed with the skin and subcutaneous tissues tented up away from the underlying artery to avoid unintentional laceration. B) Blunt dissection is continued until the artery is isolated and all branches are identified. C) The proximal and distal ends of the artery are ligated with 2 4-0 silk ties. The specimen is excised with sharp scissors, and hemostasis is confirmed prior to skin closure.
|
Etiology
Dr. Bhatti: Do we know the underlying cause of GCA?
Dr. Nesher: The underlying cause is unknown. Because GCA develops in older individuals—the mean age is approximately 73 years—we assume that the etiology is associated with vascular aging. That is, alteration or formation of antigens in the vessel wall, combined with aging of the immune system, can result in an abnormal antigen response that involves vasculitis. There also may be some genetic causes. In a recent study,5 certain infectious agents, including varicella-zoster virus, were implicated in GCA, but an infectious etiology has not been proven.
We know more about the pathogenesis of GCA. An unidentified antigen activates dendritic cells in the vessel wall, which stimulate T cells to differentiate into Th1 or Th17 lymphocytes. Th1 lymphocytes produce interferon gamma, and Th17 cells produce interleukin 17. These cytokines activate macrophages to form giant cells and to produce growth factors, other interleukins, and proteolytic enzymes, which progressively damage the vessel wall. This results in narrowing and obstruction of the involved vessels as well as other manifestations of GCA.
 |
|
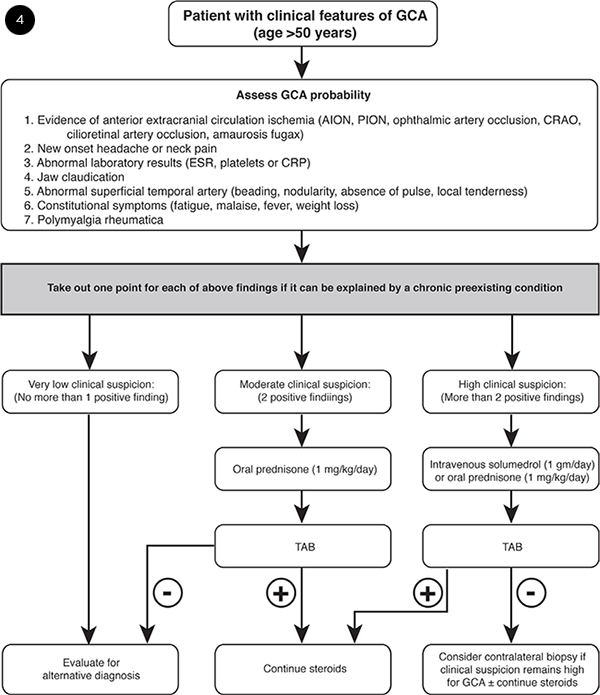
ALGORITHM TO DETERMINE PREBIOPSY RISK FOR GCA. Key: TAB, temporal artery biopsy; AION, anterior ischemic optic neuropathy; PION, posterior ischemic optic neuropathy; CRAO, central retinal artery occlusion; ESR, erythrocyte sedimentation rate; CRP, C-reactive protein. Source: El-Dairi MA et al. J Neuroophthalmol. 2015;35(3):246-253.
|
Spectrum of Disease
Dr. Bhatti: How does GCA relate to Takayasu arteritis and PMR?
Dr. Nesher: GCA and Takayasu arteritis are classified as separate diseases, based on differences in disease manifestations and in patient age and ethnicity (see Table 1). Most cases of Takayasu arteritis occur in patients younger than 40 years of age. However, there is some overlap regarding symptoms, histologic changes, and vascular distributions.
Patients with GCA may have a Takayasu-like presentation, with systemic symptoms but no headaches; they may have negative results for temporal artery biopsies, and there may be clinical or imaging evidence of involvement of other large vessels, especially the aorta and subclavian arteries. It is debatable whether these cases should be classified as large-vessel GCA or older-onset Takayasu arteritis.
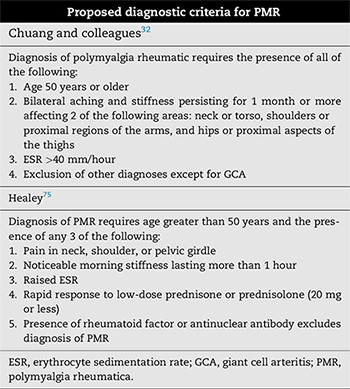
PMR is considered part of the spectrum of GCA (see Table 2). The frequency of PMR symptoms in patients with GCA ranges from 30% to 60%.6 Some patients have so-called isolated PMR—without symptoms of GCA. If you carefully evaluate these patients or perform imaging studies, you may find evidence of vasculitis in some of them, but it would be clinically silent.
If a patient with PMR has symptoms of vasculitis or has imaging findings to suggest vasculitis, we would evaluate the patient for GCA by performing temporal artery biopsy. We also might consider GCA in a patient with diagnosed PMR who has severe anemia or severe thrombocytosis or is not responding to treatment for PMR.
 |
|
TABLE 1. Source: Frohman L et al. Surv Ophthalmol. 2016;61(4):400-421.
|
Diagnosis
Dr. Bhatti: How do you diagnose GCA in your clinical practice?
Dr. Frohman: I obtain a temporal artery biopsy specimen that ideally is 1 to 2 cm long and involves appropriate sectioning, and I have it examined by elastin staining. I would emphasize that histologic analyses should be performed by a pathologist experienced in this disease—someone who is used to looking for vasculitis.
Dr. Nesher: Positive results of a temporal artery biopsy enable definitive diagnosis of GCA. However, GCA involves segments of inflammation in the blood vessel, with normal intervening segments. If the biopsy specimen is not long enough, you may miss the segment with inflammation. Approximately 20% to 30% of patients have what we call “biopsy-negative GCA.”
About 25 years ago, The American College of Rheumatology presented 5 classification criteria to differentiate GCA from other types of vasculitis. These include 1) 50 years of age or more, at the time of disease onset; 2) new headache; 3) temporal artery abnormality, such as local tenderness or decreased pulsation over the artery; 4) erythrocyte sedimentation rate ≥50 mm/hour; and 5) abnormal temporal artery biopsy showing vasculitis. However, the false-positive and false-negative rates when using these criteria have been approximately 25% each.7,8
Dr. Bhatti: Dr. Nesher, you recently coined the term “hidden GCA.”9 Can you please explain what you meant by using that term?
Dr. Nesher: The term refers to patients with GCA who present only with nonspecific symptoms, such as low-grade fever, anorexia, weight loss, and fatigue. Only when you further evaluate the patient—and sometimes only when you perform a temporal artery biopsy or imaging studies—can you diagnose GCA.
 |
|
TABLE 2. Source: Frohman L et al. Surv Ophthalmol. 2016;61(4):400-421.
|
Temporal Artery Biopsy
Dr. Bhatti: When should a temporal artery biopsy be done? And, from a logistical point of view, what particular biopsy features (timing, length of specimen, unilateral, bilateral, etc.) do you recommend?
Dr. Frohman: I was taught early in my career that if you have a high rate of positive biopsy results, it probably means you’re missing cases.
In a study of ophthalmic-presenting patients, Hayreh et al.10 found that approximately 20% of them had ocular signs and no systemic involvement. We know that approximately 15% of patients with GCA have negative erythrocyte sedimentation results, and a slightly smaller proportion have negative results for all lab tests. Therefore, approximately 1% of all GCA cases and 3% of cases with visual concerns will have no systemic symptoms and normal lab results. It is crucial that temporal artery biopsies be performed for these patients.
When I see a patient with nonarteritic ischemic optic neuropathy, I immediately recommend fluorescein angiography as an adjunct test. If I see a delay in choroidal perfusion or lobular nonperfusion, I would want the patient to undergo a temporal artery biopsy.
For diagnosing GCA, I advocate simultaneous bilateral biopsies of the temporal artery. When performed by an experienced surgeon, obtaining bilateral biopsy specimens as opposed to unilateral specimens involves only small increases in morbidity and time. Treatment for GCA can last 1 to 3 years and may cause serious iatrogenic disease, so I want to know for sure what I’m treating.
I avoid sequential biopsies because I have had patients refuse to undergo a second biopsy if results of the first one were negative. Also, by the time you get the result of the first biopsy and can schedule a biopsy for the second side, 1 to 2 weeks will have passed. Steroid treatment during this period could suppress some of the pathology, potentially yielding a false-negative result. Another major challenge is getting insurance approval for a second biopsy.
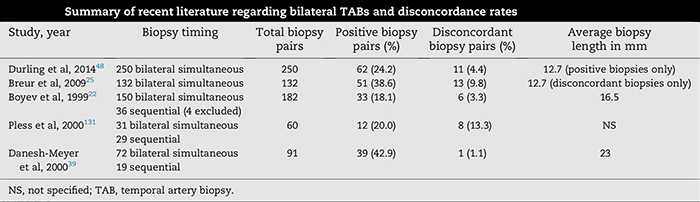
Dr. Nesher: In our center, we prefer to biopsy both sides at the same time. We found that the discordance rate between results of biopsies from the left and right sides was 25%, so approximately 12% of GCA cases would have been missed with unilateral biopsies.6 (See Table 3 for another set of discordance rates).
Dr. Bhatti: I tend to perform sequential biopsies of the temporal artery (Figs. 1-3C).11 I biopsy the temporal artery on the side of the patient’s symptoms, whether they be unilateral vision loss or headaches.
Very often I’m asked to do a biopsy by a rheumatologist or internist just to rule out GCA in a patient with vague symptoms. We developed an algorithm to predict the likelihood of a positive biopsy result, based on clinical signs and symptoms (see Fig. 4).12
 |
|
TABLE 3. Note: These discordance rates are based on all biopsied cases, not solely those with GCA. Source: Frohman L et al. Surv Ophthalmol. 2016;61(4):400-421.
|
Other Diagnostic Modalities
Dr. Bhatti: Can GCA be diagnosed without a temporal artery biopsy?
Dr. Nesher: If a patient has vision loss, jaw claudication, stroke, or a transient ischemic attack, we would perform bilateral biopsy of the temporal artery. If the suspicion of GCA is very low and the symptoms are vague and nonspecific, we would first perform duplex ultrasonography of the temporal arteries. The negative predictive value of this test is very high, approximately 90%. So if results of duplex ultrasonography are negative under these conditions, we would not perform a biopsy.
Duplex ultrasonography is available in most centers and is relatively inexpensive. However, the results are operator dependent, and you need some skill to diagnose vasculitis. The positive predictive value of duplex ultrasonography is approximately 50% to 60%. It is not a substitute for temporal artery biopsy, which has a positive predictive value of almost 100%, but these tests could be regarded as complementary.
Several other modalities can help establish the diagnosis of GCA. Fluorescein angiography was mentioned earlier. A less common modality is high-resolution magnetic resonance angiography (MRA) of the temporal or thoracic arteries, which enables evaluation of possible inflammation in the vessel wall. The diagnostic value of MRA is comparable to that of ultrasonography, but MRA is much more expensive. Positron emission tomography (PET) is another potential modality for GCA diagnosis, but PET is even more expensive than MRA and is not commonly available. PET allows for detection of large-vessel involvement in the thoracic, subclavian, or aortic areas. The predictive value of PET is still unclear, as some patients with atherosclerosis may have PET scan findings that resemble large-vessel vasculitis.
Dr. Frohman: Another modality is computed tomography angiography (CTA) of the great vessels, which also has utility for examining large-vessel vasculitis. Results of CTA are abnormal in approximately two-thirds of patients with GCA.
Periodically, you may see a patient with symptoms of GCA, but the patient says, “Five years ago they biopsied both sides, and the result was negative.” So, you can’t biopsy the temporal artery again, but you’d still want histologic confirmation. There are other superficial arteries, such as the occipital arteries, from which tissue can be obtained relatively easily.
Dr. Nesher: About 20% to 30% of patients with GCA have negative results of temporal artery biopsies, the so-called “biopsy-negative GCA.” In these cases, the final diagnosis is difficult to make and is based on all clinical laboratory and imaging findings consistent with GCA, as well as test results that rule out other conditions. We treat and monitor these patients very closely, for at least 6 months, to make sure that GCA is the right diagnosis.
___________________________
1 Frohman L et al. Surv Ophthalmol. 2016;61(4):400-421.
2 Chacko JG et al. Saudi J Ophthalmol. 2015;29(1):48-52.
3 Goodman BW Jr. Am J Med. 1979;67(5):839-852.
4 Gonzalez-Gay MA et al. Medicine (Baltimore). 2005;84(5):269-276.
5 Gilden D, Nagel MA. Curr Opin Infect Dis. 2016;29(3):275-279.
6 Nesher G. J Autoimmun. 2014;48-49:73-75.
7 Hunder GG et al. Arthritis Rheum. 1990;33(8):1122-1128.
8 Murchison AP et al. Am J Ophthalmol. 2012;154(4):722-729.
9 Nesher G, Breuer GS. Rambam Maimonides Med J. 2016;7(4):e0035.
10 Hayreh SS et al. Am J Ophthalmol. 1998;125(4):521-526.
11 Bhatti MT, Tabandeh H. Curr Opin Ophthalmol. 2001;12(6):393-399.
12 El-Dairi MA et al. J Neuroophthalmol. 2015;35(3):246-253.
___________________________
Dr. Bhatti is chief of the neuro-ophthalmology division at Duke Eye Center and professor of ophthalmology, neurology, and neurosurgery at Duke University Medical Center in Durham, N.C. Relevant financial disclosures: Novartis Pharmaceuticals: C,L; Receptos: C.
Dr. Frohman is vice chair of ophthalmology and professor and director of neuro-ophthalmology at Rutgers–New Jersey Medical School in Newark. Relevant financial disclosures: Quark Pharmaceuticals: S.
Dr. Nesher is a rheumatologist and head of the internal medicine department at Shaare-Zedek Medical Center and associate clinical professor at the Hebrew University in Jerusalem. Relevant financial disclosures: None.
For full disclosures and the disclosure key, see below.