By Wei Yan Ng, MD, MMed (Ophth), Ranjana Mathur, FRCS (Ed), and Daniel Shu Wei Ting, MD, PhD
Edited By: Ingrid U. Scott, MD, MPH, and Sharon Fekrat, MD
Download PDF
Retinal arterial macroaneurysm (RAM) is an acquired focal aneurysmal dilation of an arteriole, usually within the first 3 orders of the retinal arterial system (Fig. 1). RAM is uncommon (approximately 1 in 4,500 people)1 and has a predilection for elderly, hypertensive women. RAM typically occurs along the temporal branches, often at points of bifurcation or arteriovenous crossing.
The thickening of the arteriolar vessel wall is similar to arteriosclerotic changes elsewhere, resulting in focal areas of ischemia, remodeling of the greater intimal collagen, and, finally, dilation of the vessel diameter.2 Breaks within the arteriolar wall result in a fusiform dilation of the wall, increasing the risk of exudation and rupture.
Signs and Symptoms
The onset of RAM is often insidious, but patients occasionally present with acute, severe visual loss. Lavin and colleagues classified RAM into 3 clinical forms: quiescent, hemorrhagic, and exudative.3
Quiescent RAM. This is an incidental finding on routine examination; it seldom results in visual symptoms.
Hemorrhagic RAM. The hemorrhagic form tends to present acutely, with rapid visual deterioration. On clinical examination, a saccular or fusiform dilation along the first- or second-order arteriole might be seen. It often presents with a characteristic multilayered hemorrhage involving the the vitreous, as well as the preretinal, intraretinal, and subretinal spaces.
Exudative RAM. Compared to hemorrhagic RAM, the exudative form has a more gradual, indolent course. It is characterized by the presence of lipid deposition in a circinate pattern surrounding the lesion, as well as intraretinal edema and accumulation of subretinal fluid. If the accumulation of hard exudates and retinal edema occurs at the fovea, permanent loss of central vision may result.
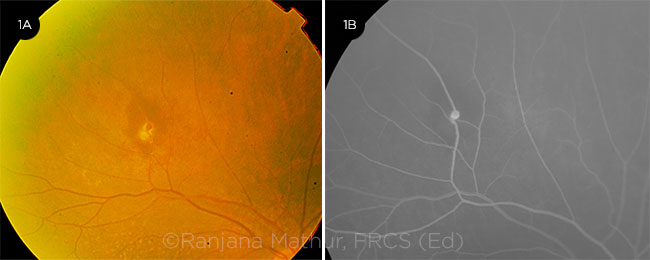 |
|
EVIDENCE OF RAM. (1A) Fundus photograph of eye with a retinal arteriole macroaneurysm, evidenced by exudation and subretinal blood in the area of an arteriolar bifurcation. (1B) FFA in the early phase highlights the focal hyperfluorescent dilation of the arteriole.
|
Clinical Evaluation
A complete assessment of RAM requires a combination of clinical examination and imaging studies, such as B-scan ultrasonography, fundus fluorescein angiography (FFA), indocyanine green angiography (ICGA), and optical coherence tomography (OCT).
B scan. If extensive vitreous hemorrhage obscures the view of the fundus, ultrasound B scan can be used to exclude conditions such as retinal tears, detachments, and breakthrough hemorrhage from polypoidal choroidal vascuolopathies.
FFA. This is the ancillary study of choice to diagnose and delineate the suspected lesion. The typical RAM fills up uniformly in the early phase, revealing a fusiform dilation of the arteriole with late leakage. The presence of a thrombus may result in filling defects. Areas of blocked fluorescence due to the overlying blood may also be present. With the aid of dynamic FFA, pulsatility of the macroaneurysm may be observed in 10% of cases.
ICGA. If significant hemorrhage renders FFA inconclusive, ICGA may be a useful alternative because it has an absorption and emission peak in the near-infrared range, allowing deeper penetration through the areas of hemorrhage (Fig. 2). However, both FFA and ICGA are considered invasive and relatively time-consuming.
OCT. Spectral-domain OCT (SD-OCT) enables visualization of the lesion as well as associated complications. The macroaneurysm will appear as a round or oval hyperreflective lesion in the inner retinal layers. In addition, SD-OCT may demarcate the extent of lipid deposition and accumulation of intraretinal and/or subretinal fluid. However, SD-OCT is significantly affected by the presence of media opacities such as vitreous hemorrhage. In such circumstances, ICGA may be more informative.
Diagnostic challenges. Despite several imaging options at our disposal, diagnosing RAM may prove challenging when extensive hemorrhage is present. If a clear focal lesion cannot be identified, the patient may be misdiagnosed with other conditions such as polypoidal choroidal vasculopathy or exudative age-related macular degeneration.
In such cases, examining the fellow eye closely may help guide the physician. Important diagnostic clues for RAM include absence of drusen and the presence of significant hypertensive retinopathy in the fellow eye in the setting of an asymmetric vascular lesion with lipid or blood concentrated around the bifurcation of an arteriole in the affected eye.
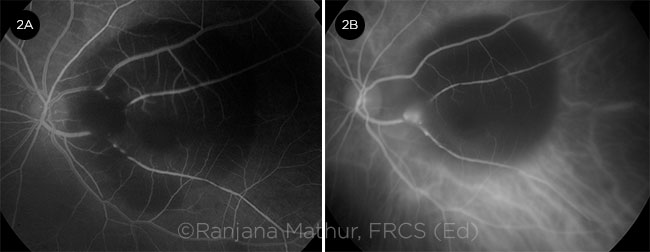 |
|
FFA VS. ICGA. In the presence of excessive blood with significant blocked hyperfluorescence, ICGA may be superior to FFA. RAM is completely obscured by blood on FFA (2A) but can be seen clearly on ICGA (2B).
|
Treatment
Hypertensive patients with RAM should be referred to their primary care physician to optimize blood pressure control. Management of the ocular manifestations of RAM depends on the visual acuity in the affected eye, as well as the location of the lesion and associated consequences.
Observation. Observation is generally recommended for eyes with good visual acuity, with the RAM located inferiorly or nasally, and minimal exudation or hemorrhage; in most of these cases, spontaneous involution and resolution will occur. Often, RAM with associated retinal or breakthrough vitreous hemorrhage will thrombose and subsequently regress without further intervention.
Argon laser photocoagulation. If the patient develops significant hemorrhage or exudation threatening the macula, especially when the RAM is located superotemporally, early retinal argon photocoagulation is recommended.
RAMs complicated by subfoveal hemorrhage tend to have a worse visual prognosis due to the disorganization of the retinal inner layers and associated neurosensory detachment.4 Subsequent fibrosis and atrophy may result in permanent central vision loss.
Direct application. Direct argon laser to the entire macroaneurysm should be applied cautiously, with relatively low power, long burn duration, and large spot size to avoid rupture of the RAM. Potential complications of laser photocoagulation of the RAM include breakthrough hemorrhage, development of choroidal neovascularization, vascular occlusion at the site of treatment, and early increase in exudates following laser application. It has been reported that only 16% to 27% of RAMs are successfully thrombosed after treatment with laser photocoagulation.5
Indirect application. Indirect retinal laser photocoagulation is thought to reduce oxygen consumption and thus blood flow to the RAM as well as decreasing exudation from surrounding capillaries. This technique avoids direct thermal energy to the lesion, which could result in arteriolar occlusion and rupture. However, it has not been established which of these methods is superior, and some physicians prefer a combination of both direct and indirect laser.6
Threshold versus subthreshold. Battaglia Parodi and colleagues compared subthreshold laser versus threshold laser treatment. They found comparable visual and anatomic outcomes between the groups, with reduced epiretinal membrane formation in the subthreshold group.7 It is hypothesized that retinal hyperthermia below the cell-death threshold results in selective damage to retinal pigment epithelial cells, with improved balance of angiogenic factors and cytokine release.
Anti–vascular endothelial growth factor (anti-VEGF). Intravitreal anti-VEGF may provide an alternative treatment option.8 In a case series of 37 patients with complicated RAM, Pichi et al. reported that 3 monthly injections of intravitreal bevacizumab were associated with rapid improvement in best-corrected visual acuity and reduction of central retinal thickness. Inhibition of VEGF may close the permeable arteriole and normalize its function.8
YAG laser hyaloidotomy. If a large, nonresolving premacular hemorrhage is present, YAG laser hyaloidotomy may be performed to promote dispersion of the blood into the vitreous, where it may be more readily absorbed. This approach remains controversial due to the possible complications of macular hole, retinal detachment, or vitreous hemorrhage requiring vitrectomy.9
Intravitreal gas. Prolonged submacular hemorrhage may be toxic to the underlying photoreceptor and retinal pigment epithelial cells. Displacement of the hemorrhage may be achieved by injection of expansile intravitreal gas, with or without the aid of adjunctive tissue plasminogen activator.10
Pars plana vitrectomy. In cases involving a breakthrough vitreous hemorrhage that does not clear, pars plana vitrectomy can be considered. Coexisting submacular hemorrhage may be displaced at the same time.11
Conclusion
RAMs generally involute spontaneously and have a favorable visual prognosis. However, severe complications such as extensive hemorrhage and exudation involving the fovea can lead to severe, permanent central visual loss. Therefore, it is important for the treatment regimen to be tailored for each patient according to the lesion’s characteristics.
___________________________
1 Xu L et al. Br J Ophthalmol. 2007;91(6):840-841.
2 Fichte C et al. Am J Ophthalmol. 1978;85(4):509-518.
3 Lavin MJ et al. Br J Ophthalmol. 1987;71(11):817-825.
4 Cahuzac A et al. Eur J Ophthalmol. 2016;26(1):36-43.
5 Meyer JC et al. Graefes Arch Clin Exp Ophthalmol. 2015;253(4):537-541.
6 Koinzer S et al. Br J Ophthalmol. 2015;99(10):1345-1353.
7 Battaglia Parodi M et al. Invest Ophthalmol Vis Sci. 2012;53(4):1783-1786.
8 Pichi F et al. Am J Ophthalmol. 2013;155(2):287-294 e281.
9 Ulbig MW et al. Arch Ophthalmol. 1998;116(11):1465-1469.
10 Johnson MW. Curr Opin Ophthalmol. 2000;11(3):201-206.
11 Wu TT et al. Acta Ophthalmol. 2011;89(2):194-197.
___________________________
Dr. Ng is an ophthalmology resident at the Singapore National Eye Centre. Dr. Mathur is the senior consultant at the Singapore National Eye Centre and assistant professor at the Duke-National University Singapore (Duke-NUS). Dr. Ting is the associate consultant at the Singapore National Eye Centre and assistant professor at Duke-NUS Medical School. Financial disclosures: None.