Download PDF
Ectropion uveae (EU) is defined by the presence of iris pigment epithelium on the anterior surface of the iris. Although EU typically is an acquired condition, it may occur as an isolated congenital anomaly or in association with systemic diseases such as neurofibromatosis.
This review focuses on various types of EU, with an emphasis on secondary glaucoma and its management.
Overview
Acquired. Acquired EU is often observed with neovascularization of the iris and neovascular glaucoma; it also may be associated with any inflammatory, ischemic, or neoplastic process involving the iris.
This type of ectropion is commonly progressive. A contracting fibrovascular membrane with atrophy of the iris stroma causes a tractional curling of the posterior pigment epithelium, iris sphincter muscle, and stroma around the pupillary margin.
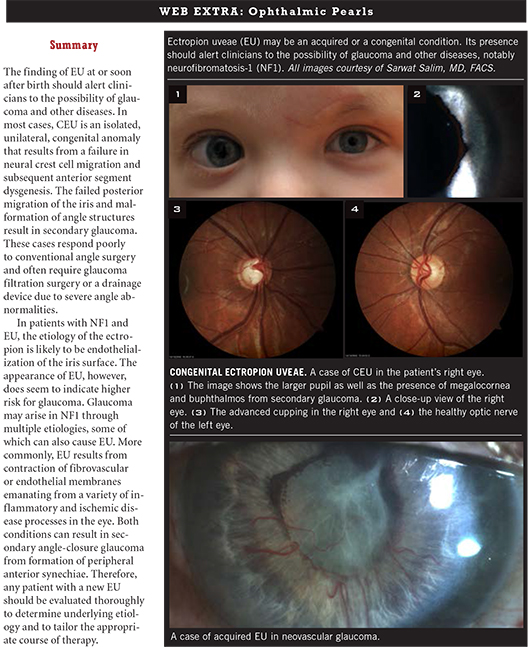
Congenital. Congenital EU (CEU) is a rare disorder, and epidemiological data on incidence are scarce. Although bilateral cases have been reported, the disease is usually unilateral. In addition, it tends to be nonprogressive.
CEU is characterized by iris pigment hyperplasia onto the anterior surface of the iris around the pupillary margin. In contrast to acquired EU, the iris sphincter muscle and stroma are not affected in CEU and are not everted on histopathologic examination.1 The hyperplasia is thought to be induced by a primordial endothelium, an embryological remnant that fails to fully regress in the anterior chamber.2 This anomaly may be caused by a late developmental arrest of neural crest tissue in utero.1
Typical clinical findings include a glassy, smooth, cryptless iris surface, iris stromal atrophy, proliferation of iris pigment epithelium onto the anterior surface of the iris, and glaucoma. The pupil is typically round and reactive to light but may grossly appear irregular because of the abnormal iris pigment epithelium. The affected eye may exhibit mild to moderate ptosis with good levator function. This finding is most likely related to the neural crest cell origin of Mueller’s muscle.
Systemic diseases. Although EU has been associated with several systemic diseases, including neurofibromatosis, facial hemiatrophy, and Prader-Willi syndrome, the condition is most often associated with neurofibromatosis-1, or NF1 (see below).
Secondary Glaucoma and CEU
Glaucoma is a frequent complication of CEU. In one report, glaucoma occurred in seven of eight cases of CEU.3 In another, glaucoma was found in nine of 10 patients with CEU, and the 10th patient was expected to ultimately develop glaucoma.1
Age of onset. In the studies cited above, glaucoma was most commonly diagnosed in childhood or early adolescence, but the patients’ ages at the time of diagnosis ranged from 7 months to 42 years. The variation in onset and severity of glaucoma may be explained by the degree of arrest in iris migration and subsequent trabecular meshwork malformation. The amount of endothelial remnant has also been shown to vary but does not seem to be associated with glaucoma severity.2
Findings. In eyes with CEU, gonioscopy typically reveals an anterior iris insertion, which in some cases extends almost to Schwalbe’s line.1,3 The failed posterior migration of the iris is thought to cause drainage angle dysgenesis and incomplete formation of trabecular meshwork and Schlemm’s canal.2 This dysgenesis is the primary mechanism of glaucoma in CEU.
Differential diagnosis. The differential diagnosis of a patient with CEU and glaucoma includes Axenfeld-Rieger syndrome (ARS) and iridocorneal endothelial (ICE) syndrome.
- ARS, like CEU, is associated with an arrest in neural crest tissue development. Findings include a high iris insertion, angle dysgenesis, EU, and glaucoma. However, the ectropion may manifest later in ARS and tends to be progressive. Furthermore, the syndrome is associated with other clinical findings such as posterior embryotoxon, iris atrophy, and corectopia or polycoria. Facial abnormalities—such as maxillary hypoplasia and dental defects—may be noted. ARS is autosomal dominant, frequently hereditary, and bilateral. Glaucoma is noted in 50 percent of patients with ARS.4
- ICE syndrome is typically unilateral and is an acquired condition (serologic studies indicate an association with herpes simplex virus or Epstein-Barr virus infection). Although ICE syndrome predominantly affects middle-aged women, an occurrence in a child has been reported. In ICE syndrome, the altered corneal endothelial cells grow over the iris and trabecular meshwork and result in formation of a cellular membrane. The contraction of this membrane leads to formation of peripheral anterior synechiae and secondary angle-closure glaucoma.5
Treatment. Treatment of glaucoma in patients with CEU usually requires surgery, although medical management may be attempted initially. Visual outcomes depend on prompt diagnosis and treatment of the glaucoma.
Effective surgical interventions for CEU-associated glaucoma differ from the techniques used in primary congenital glaucoma. Although the success rate with early goniotomy in primary congenital glaucoma is greater than 80 percent, this procedure’s success appears to be much lower in eyes with CEU, presumably due to severe angle dysgenesis in this disorder. Glaucoma filtration surgery or drainage device placement may be necessary.4,6
In one study, seven of nine patients with glaucoma associated with CEU required trabeculectomy, and a total of six goniotomies in three patients did not control intraocular pressure (IOP), even with the adjunctive use of medications.1 In another study, six of seven patients required trabeculectomy.3
Follow-up data on treatment success rates over an extended period in patients with CEU-associated glaucoma are not available. However, data are available for other types of secondary pediatric glaucomas. Trabeculectomy alone has a 30 to 35 percent success rate over extended follow-up. With the use of the antimetabolite mitomycin C (MMC), the procedure is 52 to 95 percent successful at lowering IOP, with a 60 percent success rate at two years. A 7 to 14 percent risk of late postoperative endophthalmitis, however, is associated with this procedure.
Glaucoma drainage devices have a 44 to 95 percent success rate, with 60 to 65 percent success at two years.4 The risk of endophthalmitis is lower with drainage implants, but adjunctive medical therapy is often necessary to maintain IOP control as opposed to trabeculectomy with MMC. In addition, complications that require surgical intervention may occur, including tube-cornea touch, implant exposure, tube blockage, or dislocation.
In refractory cases, cyclodestructive procedures may become necessary, but these procedures may have limited success and a high complication rate.4
(click to expand)
Ectropion Uveae and NF1
Glaucoma and NF1. Glaucoma in patients with NF1 is typically diagnosed early in childhood.3,7-8 In CEU associated with NF1, endothelialization of the iris surface may provide the forces needed to develop CEU and also cause iridocorneal adhesions that result in glaucoma.
The finding of EU on exam seems to be closely correlated with an increased glaucoma risk in patients with NF1, specifically in those with orbital involvement. Although glaucoma is considered a rare complication of NF1, it has been noted in 23 to 50 percent of patients with orbitofacial involvement and an ipsilateral lid plexiform neurofibroma.8
Morales et al. found glaucoma in 13 of 56 patients with NF1 with orbital involvement, and eight of these 13 were diagnosed with EU. Of the 43 patients who had NF1 and orbital involvement but no glaucoma, only four had EU, and these eyes had partial angle closure.8 In another study, all five patients with orbitofacial NF1 and glaucoma demonstrated histopathological EU.7
The pathogenesis of glaucoma in NF1 is likely multifactorial, involving infiltration of the anterior chamber angle by neurofibromas, neurofibromatous thickening of the ciliary body and choroid leading to angle closure, secondary angle closure by anterior synechiae, and developmental angle anomalies.8
Visual prognosis is poor in these patients. In the study by Morales et al., all of the patients with NF1 and glaucoma who were able to give a reliable visual acuity level were either legally or totally blind, and one-third required either enucleation or evisceration of the affected eye.8
Treatment strategies for glaucoma in patients with NF1 and EU are similar to those discussed above for secondary pediatric glaucomas.
___________________________
1 Dowling JL Jr et al. Ophthalmology. 1985;92(7):912-921.
2 Wilson ME. J Pediatr Ophthalmol Strabismus. 1990;27(1):48-55.
3 Ritch R et al. Ophthalmology. 1984;91(4):326-331.
4 Beck AD. Ophthalmol Clin North Am. 2001;14(3):501-512.
5 Salim S, Shields MB. EyeNet Magazine. 2011;15(9):47-49.
6 Salim S, Walton DS. Goniotomy and Trabeculotomy. In: Yanoff M, Duker JS. Ophthalmology. 3rd ed. New York: Elsevier;2008:1241-1245.
7 Edward DP et al. Ophthalmology. 2012;119(7):1485-1194.
8 Morales J et al. Ophthalmology. 2009;116(9):1725-1730.
___________________________
Dr. Prenshaw is a recent graduate of the University of Tennessee at Memphis, where Dr. Salim is associate professor of ophthalmology and director of the glaucoma service. The authors report no related financial interests.