By Aaron M. Hein, BS, and Preeya K. Gupta, MD
Edited by Sharon Fekrat, MD, and Ingrid U. Scott, MD, MPH
Download PDF
Epidemic keratoconjunctivitis (EKC), an ocular infection caused by various serotypes of adenovirus, is the most common cause of infectious conjunctivitis in the world. As the name suggests, cases of EKC generally occur in clusters. The rugged capsule of the adenovirus confers resistance to pH extremes and enables long-term viability on contaminated surfaces. This results in high infectivity of adenovirus and frequent outbreaks of EKC among populations in close quarters, such as schools, camps, or nursing homes. It is particularly problematic in health care and ophthalmology clinics.
Currently, no antiviral therapies have been shown to significantly impact length or progression of EKC. Thus, preventive strategies undertaken by clinicians are paramount in reducing infections and their public health consequences. This article will describe barriers to EKC prevention, as well as clinical and public health strategies to decrease the risk of epidemics of adenoviral ocular infection, with particular emphasis on rapid diagnosis and patient management.
Presentation
Often, EKC initially manifests as a flu-like syndrome consisting of fever, malaise, and myalgias followed by the appearance of ocular signs and symptoms, including a red eye, eyelid edema, excessive tearing, irritation, foreign body sensation, and photophobia. EKC frequently begins as a unilateral condition but, in 70% of cases, will become bilateral within the first week of symptoms as a result of hand-to-eye transmission.1 EKC is typically a self-limiting infection lasting 2 to 3 weeks, and patients are typically contagious for 10 to 14 days.
The initial conjunctival phase of infection can be complicated by the formation of pseudomembranes and involvement of the cornea. Corneal subepithelial infiltrates composed of leukocytes and collagen deposition can appear 7 to 10 days after symptoms present in up to 50% of cases and can persist for months to years after infection. These subepithelial infiltrates can cause significant visual impairment and irregular astigmatism that may require further evaluation and treatment.1,2 Thus, prompt diagnosis and infection control are essential to reduce potentially harmful complications.
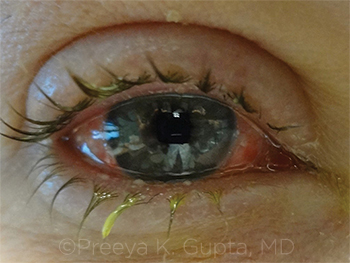 |
|
EKC PRESENTATION. Typical clinical presentation of patient with EKC. Note crusting, lid swelling, and conjunctival injection.
|
Diagnosis
Diagnosis of EKC is typically made on clinical presentation. However, because up to 62% to 75% of cases of infectious conjunctivitis are caused by adenovirus, rapid confirmation of the diagnosis can be key to preventing further spread.3 The diagnosis can be confirmed with the commercially available adenovirus detection device, AdenoPlus (Rapid Pathogen Screening), an immunoassay technique with 88% sensitivity and 91% specificity for adenoviral infection.4 However, adoption of this device has not been widespread, in part due to reliance on the clinical diagnosis.
Furthermore, there is insufficient awareness of the socioeconomic and public health implications of EKC, which can include absenteeism, decreased productivity, lost wages, and long-lasting ocular morbidities. Delay in recognition of an outbreak has promoted spread of adenovirus at some institutions, prompting research into better preventive strategies and early diagnosis.
Prevention
Adenovirus is highly infectious and often spreads via contact with hands or surfaces contaminated with ocular secretions; consequently, most outbreaks are traced to exposure at public venues or from medical equipment at health and ophthalmology clinics.
Many clinics have instituted programs that have focused primarily on prevention via hand-washing, medical equipment sanitation with alcohol wipes, single-use equipment, and triage of EKC patients with universal isolation precautions.5
One such program, at Johns Hopkins Hospital, focuses on triaging employees who have “red-eye” complaints, based on symptom duration, to isolation in designated patient rooms; further, it instituted a 14-day furlough for employee-patients who have positive results on polymerase chain reaction testing for adenovirus.6
Disinfecting surfaces and equipment. CDC guidelines recommend 60% to 80% ethyl alcohol as an effective virucidal agent for adenovirus for the disinfection of surfaces.2,7 With regard to disinfection of tonometers, a recent systematic review found that alcohol swabs had equal utility compared with immersion in peroxide or use of disposable tonometer tips (Tonosafe, Haag-Streit) in reducing adenoviral transmission, while being much more cost-effective.8
Strategies to prevent adenovirus spread in the clinic. The following measures can help to reduce the transmission of EKC in a clinical setting.
- Identify patients with complaints that suggest adenoviral infection and see them in designated isolation rooms.
- Utilize rapid diagnostic methods to identify, quantify, and isolate adenoviral cases in the community.
- Use proper hand-washing technique between patients, especially when a case of adenovirus is suspected or confirmed.
- Employ single-use devices when available; if not, alcohol swabs are effective in limiting spread of EKC.
- Use povidone-iodine eyedrops and avoid corticosteroids.
- Educate patients about the infectivity window and the risk to family, friends, and contacts.
What About Corticosteroids?
In the past, topical corticosteroids such as prednisolone were frequently prescribed to alleviate severe symptomatic inflammation in EKC. Animal studies have shown that steroids are effective in reducing inflammation during the acute phase of EKC and decreasing the likelihood of development of corneal subepithelial infiltrates. However, the studies also showed that their use increased viral replication and titers and prolonged the mean duration of viral shedding from 10 days to 14 days.9
A longer infectious period has significant public health implications, resulting in a wider transmission to contacts at home, work, and school. For these reasons, routine corticosteroid use is not indicated for EKC, and ophthalmologists managing severe EKC inflammation should carefully weigh the risks and benefits of steroids.
Povidone-Iodine Eyedrops
One novel strategy to decrease spread of EKC has been the off-label use of the antiseptic povidone-iodine ophthalmic solution (Betadine, Alcon) administered as eyedrops. In vitro studies have shown that povidone-iodine is able to rapidly inactivate adenovirus within 1 minute of exposure.10 Small clinical studies have suggested that 2% or greater solutions of povidone-iodine can reduce duration of infectivity and limit spread of ocular adenovirus.11,12
The most common ocular side effect of povidone-iodine administration is stinging. Thus, we recommend application of topical anesthetic before instilling povidone-iodine drops. The eye should be gently irrigated with sterile saline after administration to reduce ocular irritation and possible toxicity.
Povidone-iodine use in EKC. Following is a suggested sequence:
- Anesthetize affected eye(s) with 0.5% proparacaine ophthalmic solution. Allow 30 seconds to take effect.
- Place a few drops of 5% povidone-iodine ophthalmic solution into the inferior fornix.
- Have the patient close his or her eyes and blink to spread solution throughout affected surface.
- Wipe away excess solution and consider gentle irrigation with sterile saline after 1 to 2 minutes.
Conclusion
EKC is a common viral ocular infection. Although it is self-limiting, it is highly infectious with potentially serious complications, posing important public health concerns. Focusing on preventive strategies and limiting the window of infectivity can help reduce outbreaks and associated morbidities. We recommend rapid diagnosis, isolation precautions, and prompt use of antiseptic povidone-iodine drops to promote identification and prevent spread of adenovirus in the clinic.
___________________________
1 Jhanji V et al. Surv Ophthalmol. 2015;60(5):435-443.
2 Pihos AM. J Optom. 2013;6(2):69-74.
3 Sambursky RP et al. Optometry. 2007;78(5):236-239.
4 Sambursky RP et al. Ophthalmology. 2006;113(10):1758-1764.
5 Gottsch JD et al. Ophthal Epidemiol. 1999;6(1):29-39.
6 Kuo IC et al. Infect Control Hosp Epidemiol. 2014;35(6):728-731.
7 Rutala WA, Weber DJ. Guideline for Disinfection and Sterilization in Healthcare Facilities. Centers for Disease Control, 2008. Accessed Dec. 5, 2016.
8 Omar Akhtar A et al. Can J Ophthalmol. 2014;49(4):345-350.
9 Romanowski EG et al. Arch Ophthalmol. 1996;114(5):581-585.
10 Kawana R. Dermatology. 1997;195(suppl 2):29-35.
11 Trinavarat A, Atchaneeyasakul LO. J Ocul Pharmacol Ther. 2012;28(1):53-58.
12 Ozen Tunay Z et al. Cutan Ocul Toxicol. 2015;34(1):12-15.
___________________________
Mr. Hein is a medical student, and Dr. Gupta is assistant professor of ophthalmology, at Duke University School of Medicine, Durham, N.C. Relevant financial disclosures: None.
For full disclosures and the disclosure key, see below.
