Download PDF
Want to learn more from Dr. Zinkernagel?
Check out his poster, “The Impact of the Vitreomacular Interface in Neovascular AMD in a Treat-and-Extend Regimen with Exit Strategy,” at AAO 2017!
Register now
Fluorescence lifetime imaging ophthalmoscopy (FLIO), a novel imaging modality that reveals specific patterns in almost any fundus disorder, may one day serve as a tool for visualizing early retinal changes in myriad retinal disorders.
While the role of FLIO is still evolving, researchers expect FLIO to be “a cornerstone for retinal imaging in the future,” said Martin S. Zinkernagel, MD, PhD, at University Hospital Bern in Bern, Switzerland, and coauthor of a review of FLIO.1 In proof-of-concept studies, he said, “We and other groups have provided [evidence] that FLIO can provide unique information to complement that [which is] obtained through other imaging modalities.”
What is FLIO? Like conventional fundus autofluorescence (FAF), FLIO is mainly a qualitative imaging procedure. (It relies on a modified Heidelberg Spectralis platform, which is not yet a standard product.) In contrast, unlike FAF, FLIO can provide quantitative analysis by measuring the fluorescence lifetime of fluorophores.
In other words, FLIO is a tool that tells how long any given retinal fluorophore glows after excitation with a laser pulse. This span of time is known as fluorescence lifetimes.
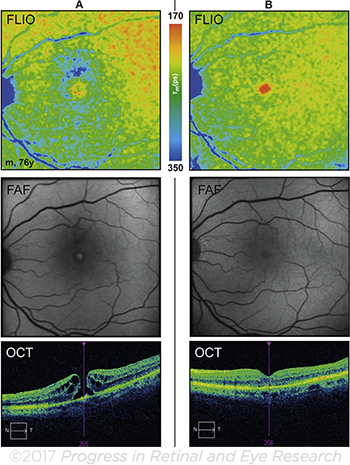 |
FLUORESCENT LIFETIMES. Macular hole, before surgery (A) and 3 months after successful closure of the hole (B).
|
Pilot studies. Comparisons of mean lifetimes of diseased eyes to healthy controls have revealed FLIO’s potential for monitoring disease progression and therapeutic outcomes, Dr. Zinkernagel said. He added that FLIO has provided information on potential markers for disease progression in retinal hered-itary disorders and macular degeneration, suggesting it may be used in clinical trials on gene therapy for these diseases.
In addition, earlier this year, researchers reported using FLIO to better understand the pathogenesis of drusen in AMD. They found that soft drusen associated with AMD progression showed longer fluorescence decays than did hard drusen. Thus, they hypothesized, FLIO might give information about the individual risk for the development of late AMD.2
Looking forward. Although Dr. Zinkernagel predicted that FLIO will become an important diagnostic technology, its precise use is yet to be revealed. “Whether it will be used in an academic setting or by ophthalmologists in practice is difficult to predict based on the current knowledge.”
—Miriam Karmel
___________________________
1 Dysli C et al. Prog Retin Eye Res. Published online June 30, 2017.
2 Sauer L et al. Time-resolved fundus autofluorescence in dry AMD. Presented at: ARVO; May 9, 2017; Baltimore.
___________________________
Relevant financial disclosures—Dr. Zinkernagel: National Cancer Institute/National Institutes of Health: S; Swiss National Science Foundation: S.
For full disclosures and disclosure key, see below.
More from this month’s News in Review