By Kathryn Lewis, BS, Bea Palileo, MS, Charles Pophal, BS, Joseph Yasmeh, BS, and Ray Glendrange, MD
Edited By: Ingrid U. Scott, MD, MPH, and Bennie H. Jeng, MD
Download PDF
Herpes zoster (HZ), or shingles, results from reactivation of latent infection with varicella- zoster virus, which also causes chickenpox. Anyone who has had chickenpox, even in subclinical form, is at risk for developing HZ. It is estimated that the lifetime risk of HZ is 30%, and 1 million new cases are reported annually in the United States.1 An effective shingles vaccine has been approved in the United States, Europe, and Japan; however, HZ remains common worldwide, in part because of lack of knowledge of the vaccine, as well as limited access to and shortages of the vaccine.
Although the majority of HZ cases present along truncal dermatomes, viral reactivation along the trigeminal nerve is possible, resulting in herpes zoster ophthalmicus (HZO). The estimated rate of HZO is approximately 10% of all HZ cases.1,2 In recent years, the incidence of both HZ and HZO has almost tripled, possibly related to the larger aging population.2,3 HZO is a serious and vision-threatening disease. However, with proper treatment, it is estimated that ocular complication rates decrease from 29% to 2% in those diagnosed with HZO.1
Clinical Presentation
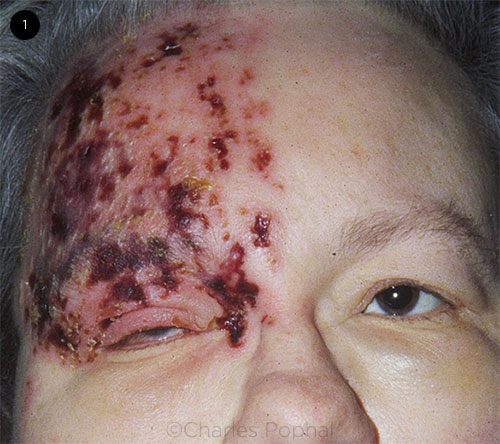
Patients with HZO may present to the emergency department, primary care, or specialty care clinics. They typically report unilateral burning pain, allodynia, and headache along the ophthalmic (V1) branch of the trigeminal nerve. After the onset of these prodromal symptoms, an erythematous, vesicular rash subsequently erupts, lasting up to 10 days (Fig. 1).
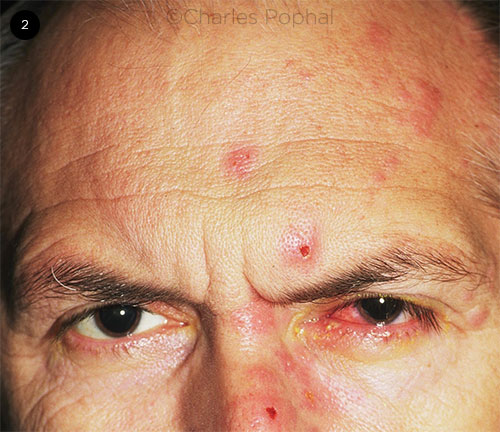
The rash may involve the tip of the nose or nasal ridge, known as Hutchinson sign (Fig. 2). This region is innervated by the nasociliary fibers, which also serve the conjunctiva, sclera, cornea, iris, and choroid. Hutchinson sign is associated with an increased risk of HZO. The absence of this sign does not rule out disease, however, as up to 30% of patients with HZ but without Hutchinson sign will develop HZO.
In addition to the characteristic dermatologic findings, HZO can present with a variety of ocular findings, which occur in up to 50% of patients who do not receive antiviral treatment.4 Depending on the severity of infection, patients may develop significant ptosis secondary to edema, resulting in inability to close the eyelid and dry eye. Red eye, excessive tearing, eye pain, blurred vision, photophobia, and decreased visual acuity are common complaints. The most common ocular presentations include mucopurulent conjunctivitis, anterior uveitis, and keratitis (nummular and disciform).1 Episcleritis and chemosis are often seen in acute disease, as well.
 |
|
CLINICAL PRESENTATION. A woman with V1 dermatomal rash, ptosis, and periorbital edema.
|
 |
|
EARLY RASH. Patient with HZO and early rash outbreak. Note the presence of Hutchinson sign on the nose.
|
Differential Diagnosis
The differential diagnosis of HZO can be divided into the following categories, depending on presentation.
Pain. When patients present with pain, consider tension headache, migraine, cluster headache, and giant cell arteritis.
Rash. Although the vesicular rash is characteristic of HZO, other causes include contact dermatitis, impetigo, and herpes simplex virus (HSV) infection. Distinguishing HSV from HZO can be difficult. HSV can present with a vesicular rash, but it is not typically confined to one dermatome. Further, classic corneal dendrites occur in HSV, compared with the pseudodendrites in HZO.5
Ocular findings. In cases of periorbital swelling and ptosis, consider preseptal cellulitis, orbital tumor, and cavernous sinus thrombosis. Other possibilities include infectious keratitis, interstitial keratitis, and uveitis.
 |
|
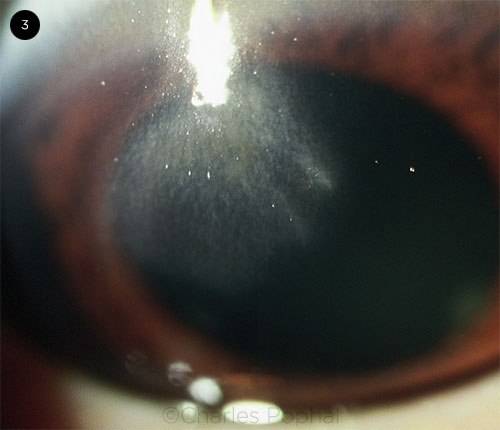
CORNEAL COMPLICATIONS. Central opacification and scarring of the cornea.
|
Complications
Complications of HZO have been documented in the eye and central nervous system. Some studies suggest that up to 50% of untreated patients with HZO will go on to develop ocular complications.1 These commonly include corneal involvement (Fig. 3), often in the form of neurotrophic keratitis. Intraocular pressure (IOP) should be measured during treatment because of the risk of IOP spikes, which may be related to the ocular disease or to steroid use. Complications can extend to the posterior segment, resulting in ocular apex syndrome, optic neuritis, and acute retinal necrosis.4
Notably, HZO has been associated with central nervous system pathology, including cerebral stroke.6 Within one year of a patient’s initial episode, those with HZO have a risk of stroke 4.3 times higher than that of all HZ patients (whose risk is 1.3 times that of controls). It is believed that the virus can cause vascular inflammation and occlusion, as the trigeminal nerve fibers extend along the middle, anterior, and inferior cerebellar arteries. Currently, no data have shown that treatment of HZO reduces the risk of stroke. Although rare, additional neurologic sequelae have been documented, including cerebellar ataxia and meningoencephalitis.6
 |
|
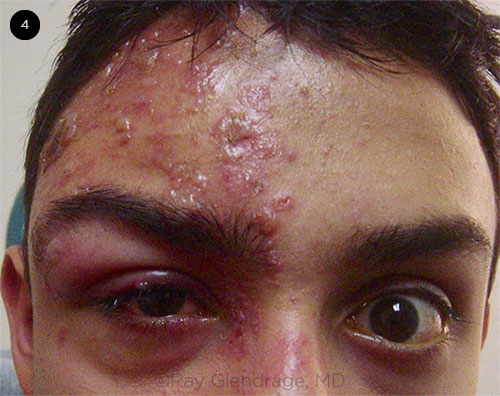
HZO IN YOUNG PATIENT. Although HZO is most common in people 50 years or older, it can occur at younger ages, as seen in this teenage boy. Note the rash in the V1 dermatome, periorbital edema, and ptosis.
|
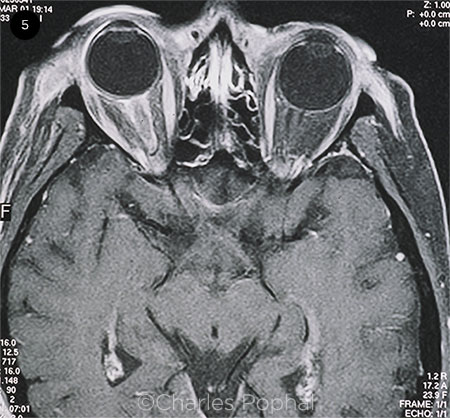 |
|
OPTIC NERVE INVOLVEMENT. MRI shows enhancement of the right optic nerve sheath consistent with optic neuritis in the setting of HZO.
|
Risk Factors
Multiple studies have evaluated the risk factors associated with the likelihood of acute, chronic, and recurrent HZO (Table 1). Of all risk factors, reactivation along the trigeminal nerve has been implicated in developing HZO. Of the branching fibers (nasociliary, frontal, and lacrimal), frontal nerve involvement is the most frequently documented. Risk factors for both acute and recurrent HZO include immunocompromised status, female sex, autoimmune disorders, and an age of 50 years or older.1 Additional risk factors for recurrence of disease include 30 or more days of zoster-associated pain during acute infection, ocular hypertension, and uveitis following acute infection.1
As expected, the risks for postherpetic neuralgia (PHN) following HZO are similar to those of truncal zoster, such as an age of 50 years or more, greater extent and severity of rash, and presence of early neuralgia.2 Ocular findings have also been associated with the likelihood of developing PHN, including decreased corneal sensation, conjunctivitis, keratitis, and uveitis.2
Table 1: Risk Factors for HZO and PHN
| Acute HZO |
Chronic HZO |
Recurrent HZO |
PHN |
|
Female sex
≥50 years old
Immunocompromised
Involvement of frontal nerve fibers of V1
Autoimmune conditions
|
Ocular HTN
Uveitis
|
Female sex
≥50 years old
Immunocompromised
≥30 days of pain
Autoimmune conditions
Ocular HTN
Uveitis
|
≥50 years old
Extent and severity of rash
Reduced corneal and cutaneous sensation
Conjunctivitis
Keratitis
Uveitis
|
| Abbreviations: HTN, hypertension; HZO, herpes zoster ophthalmicus; PHN, postherpetic neuralgia. |
Management
Vaccination. The first line of defense against HZO is prevention through vaccination. Compared to the live attenuated vaccine (Zostavax), the newer subunit vaccine (Shingrix) is more effective at reducing the incidence of HZ (51% vs. 97%, respectively, over three years).4 The CDC7 and an Academy Clinical Statement8 recommend that all immunocompetent individuals over age 50 receive Shingrix (two doses two to six months apart). Patients should be monitored for four to six weeks after vaccine administration for viral reactivation, although this occurrence is rare.
Medications. The standard treatment for HZ is antiviral medication, preferably initiated within 72 hours of rash onset.1 Antivirals reduce pain, speed rash-healing time, and decrease ocular involvement; however, they do not reduce the incidence or severity of PHN.1 A seven- to 10-day course of famciclovir (500 mg orally three times daily) or valacyclovir (1,000 mg orally three times daily) is more effective at resolving pain than acyclovir (800 mg five times daily).6 In disseminated HZ or in immunocompromised patients, IV acyclovir is indicated; if the disease is resistant to acyclovir, IV foscarnet may be considered.9
For HZO in particular, adjuvant therapy is used. Analgesics or steroids are given to reduce pain. If blinking is impaired, lubricating and antibiotic ophthalmic ointments may be used. Nonophthalmic topical antibiotics and dressings with adhesive are typically avoided, as they can exacerbate the rash and delay healing.9
For HZO with ocular involvement, therapy depends on the specific complication. For stromal keratitis and uveitis, topical corticosteroids are used judiciously with close monitoring. Be aware that tapering prior to resolution of inflammation can prolong healing time. For optic neuropathy or orbitopathy, IV acyclovir is used at a dosage of 10 to 15 mg/kg three times daily for two to three weeks. The treatment for retinitis is similar, with the addition of several months of oral antiviral therapy.9
There is not yet an evidence-based treatment regimen for chronic HZO. The Zoster Eye Disease Study (ZEDS) is assessing whether one year of oral valacyclovir 1,000 mg daily reduces ocular complications from HZO (i.e., keratitis and iritis). In a survey of ZEDS investigators, over half of respondents have reported using prolonged oral antivirals for treatment of HZO.10
___________________________
1 Tran KD et al. Ophthalmology. 2016;123(7):1469-1475.
2 Borkar DS et al. Ophthalmology. 2013;120(3):451-456.
3 Voelker R. JAMA. 2019;322(8):712-714.
4 Liesegang TJ. Ophthalmology. 2008;115(2):S3-S12.
5 Grose C. J Med Virol. 2018;90(8):1283-1284.
6 Kang JH et al. Stroke. 2009;40(11):3443-3448.
7 Shingles Vaccination. www.cdc.gov/vaccines/vpd/shingles/public/shingrix/index.html.
8 Recommendations for Herpes Zoster Vaccine for Patients 50 Years of Age and Older, 2018. aao.org/clinical-statement/recommendations-herpes-zoster-vaccine-patients-50-.
9 Le P, Rothberg M. BMJ. 2019;364:k5095.
10 Lo DM et al. Cornea. 2019;38(1):13-17.
___________________________
Ms. Lewis and Ms. Palileo are fourth-year medical students, Mr. Yasmeh is a second-year medical student, and Dr. Glendrange is an assistant professor of ophthalmology; all are at the University of California, Riverside, School of Medicine. Mr. Pophal is a fourth-year medical student at Northeast Ohio Medical University, in Rootstown, Ohio. Financial disclosures: None.