Download PDF
This month, News in Review highlights selected papers from the original papers sessions at AAO 2019. Each was chosen by the session chairs because it presents important news or illustrates a trend in the field. Only four subspecialties are included here; papers sessions will also be held in six other fields. For more information, see the Mobile Meeting Guide.
Researchers have used reporting measures developed for the Academy’s Intelligent Research in Sight (IRIS) Registry to assess treatment outcomes at Boston Children’s Hospital (BCH) for children with amblyopia.
The relevant IRIS Registry measure—Amblyopia Interocular Acuity—was published as IRIS7 in 2015 and relabelled as IRIS50 in 2019, after undergoing some changes to its specifications. The researchers found that their amblyopia treatment was successful in 71% of eligible patients by IRIS7 and in 81% of eligible patients by IRIS50, for a statistical significance of p = .006, said lead author Talia Shoshany, BS.
“We are the first to report our success by IRIS50 and compare with IRIS7 outcomes,” said Ms. Shoshany, at Harvard Medical School in Boston. “To date, only one other group has evaluated its success by IRIS7,” finding a 46% success rate.1
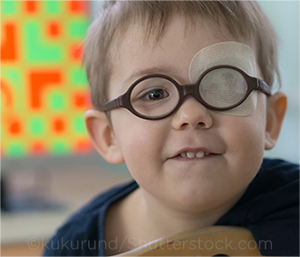 |
TRACKING OUTCOMES. The newer IRIS50 measure captures patients with denser amblyopia at baseline and tracks their overall improvement.
|
Study specifics. For this study, the researchers drew from the amblyopia outcomes database at BCH, which currently includes information on more than 2,000 patients.
Question of inclusion. The researchers evaluated all patients who were treated at BCH for amblyopia from 2010 to 2015. However, only 12% of the children met the measure’s inclusion criteria for analysis, Ms. Shoshany noted. That is, they had newly been diagnosed with amblyopia, were between the ages of 3 and 7, had an interocular difference (IOD) that was greater than .29 logMAR (or 3 Snellen lines), and had no deprivation amblyopia.
In discussing the criteria, Ms. Shoshany noted that under IRIS7, a successful treatment is that which results in an IOD of less than .23 logMAR (approximately 2 lines) at 12-18 months. In comparison, she said, IRIS50 “added visual acuity (VA) of 20/30 or better and improvement in VA of 2 lines or more” at three to 12 months.
Predictors of success. “Multivariable logistic regression was used to evaluate whether specific variables were independently predictive of success,” Ms. Shoshany said. Using IRIS7, baseline IOD and insurance status were predictive of success, but the other variables—presenting age, type of amblyopia, family history of amblyopia, initial VA, stereopsis, and type of treatment (e.g., glasses, patching, atropine, or surgery)—were not.
In contrast, under IRIS50, none of these variables was predictive of success.
Bottom line. “We believe IRIS50 is a better tool for measuring treatment success, as it takes improvement into account rather than just final VA outcomes,” Ms. Shoshany said. “This captures patients with denser amblyopia at baseline” and tracks their improvement, “even if they didn’t reach perfection.”
With regard to the question of inclusion, she said, the IRIS criteria “exclude a large proportion of patients we treat for amblyopia at BCH,” such as those who are outside the age range, have previously been treated elsewhere, or do not meet IOD specifications (i.e., those with bilateral amblyopia or those with 2 lines of IOD who are still considered amblyopic). Additional criteria should be established to allow practices to evaluate outcomes in this broader range of patients that the BCH researchers captured in their database, she concluded.
—Jean Shaw
|
Amblyopia Treatment Outcomes Using AAO IRIS7 and IRIS50 Criteria. Presented during the pediatric ophthalmology and strabisumus original papers session. When: Tuesday, Oct. 15, 8:30-10:00 a.m. Where: South 152. Access: Free.
|
___________________________
1 West CE et al. J AAPOS. 2015;20:e10.
___________________________
Relevant financial disclosures: Ms. Shoshany—None.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Hamada None.
Dr. Lin None.
Ms. Shoshany None.
Dr. Swaminathan None.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|
More from this month’s News in Review