By Samuel J. Reiter, MD, and Rona Z. Silkiss, MD, FACS
Edited by Steven J. Gedde, MD
Download PDF
Mandy Mendel* noticed that the vision in her left eye seemed blurry. The 43-year-old had always experienced perfect vision in both eyes and had no significant medical problems. She went to an optometrist to have her eyes checked and was told to schedule an appointment at a nearby top-notch retina clinic.
Initial Evaluation
Within the week, Ms. Mendel was evaluated by a retina specialist, who questioned her about her visual loss. She denied having any flashes or floaters. When the specialist examined her, he found that her visual acuity (VA) was 20/16 in her unaffected right eye and 20/40 in her symptomatic left eye. He also noted temporal fullness and proptosis of her left eye. The fundus examination demonstrated moderate temporal disc pallor in her left eye; examination of her macula and optical coherence tomography (OCT) confirmed no other pathology to explain her visual loss.
Given these findings, the specialist immediately phoned us. We suggested that Ms. Mendel undergo magnetic resonance imaging (MRI) en route to our office.
We Get a Look
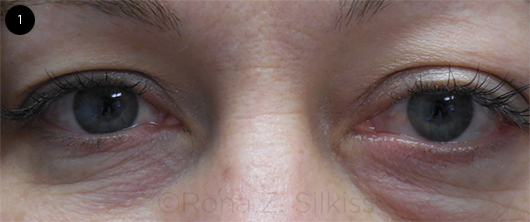
During the oculoplastic exam, Ms. Mendel corroborated having a history of blurred vision in her left eye for 2 weeks. Our examination confirmed the retina specialist’s findings of decreased VA of 20/40 in the left eye, and the VA was 20/20 in her right eye. An afferent pupillary defect was noted in the left eye. Her ocular movements were normal. The external exam was remarkable for fullness of the left temple and left-sided proptosis (Fig. 1). The slit-lamp exam was unremarkable, and the undilated fundus exam confirmed temporal pallor without edema on the left side.
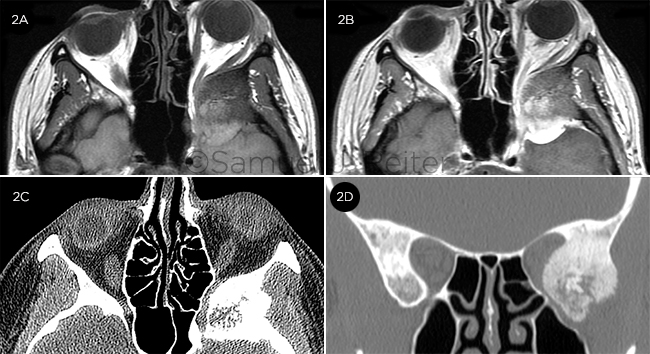
We reviewed her MRI images, which demonstrated a 3×3×3-cm mass arising from the left sphenoid wing, resulting in compression of the left optic nerve and orbital contents, producing 3 mm of proptosis (Fig. 2A). With intense enhancement, the posterior portion of the mass was evident (Fig. 2B). Confirmatory high-resolution computed tomography (CT) revealed sclerotic hyperostosis of the left sphenoid bone extending into the posterior lateral wall of the orbit (Figs. 2C, 2D).
 |
|
INITIAL VIEW. The patient had left-sided proptosis and decreased vision.
|
The Differential Diagnosis
The differential diagnosis for unilateral proptosis secondary to sphenoid wing bony changes includes orbital tumors/processes such as sphenoid wing meningioma, eosinophilic granuloma, ossifying fibroma, low-grade central osteosarcoma, Hand-Schuller-Christian disease, Paget’s disease of bone, and fibrous dysplasia.1
Pinning It Down
For our patient—a 43-year-old Caucasian woman with an enhancing intracranial mass resulting in optic nerve compression—we felt sphenoid wing meningioma to be the most likely diagnosis. The radiologist reported that fibrous dysplasia, a disease that is more common in childhood but may present in adulthood, also needed to be considered. Given that surgical management is indicated for optic nerve involvement in both entities, we referred Ms. Mendel for skull base surgery at a tertiary care facility, where a diagnosis of sphenoid wing meningioma was confirmed radiographically—and, subsequently, pathologically.
 |
|
IMAGING. (2A) Precontrast axial T1 MRI showed intracranial mass compression of the posterolateral left orbit. (2B) When intensely enhanced with contrast, the posterior portion demonstrated a dural tail. Axial (2C) and coronal (2D) high-resolution CT displayed volumetric contraction of the left posterior orbit by a heterogeneous bony lateral wall mass with feathery margins.
|
About the Disease
Sphenoid wing meningioma is the most common tumor of the intracranial space to extend into the orbit. This can occur via bone, the superior orbital fissure, or the optic canal. This slow-growing tumor arises from arachnoid villi epithelial cells. It is most common in middle-aged Caucasian women, and the average age of onset is 50 years old. Presenting features may include temporal fullness, proptosis, globe displacement, ptosis, and impaired ocular motility. If the optic nerve is involved, visual field defects, along with optic disc edema or atrophy, may be present.
CT of sphenoid wing meningioma reveals homogeneous hyperostosis of bone, often with adjacent soft tissue involvement. MRI with contrast is the imaging modality of choice, as it displays rapid and intense enhancement with gadolinium and better definition than CT of the adjacent soft tissue involvement. Classically, a “dural tail sign” of thickened dura surrounding the tumor can be identified on MRI in 52% to 78% of meningiomas. This has been demonstrated to be a reactive process that is not specific for meningioma; rather, it can be seen in any neoplastic, histiocytic, lymphocytic, or granulomatous etiology of focal meningeal irritation.2
Management typically consists of a multistage, multimodal approach that includes surgery and radiation. Approximately 90% of sphenoid wing meningiomas are benign and do not involve brain parenchyma (WHO Grade I), with 5-year recurrence-free survival approaching 90%.3
An Important Discussion
In our case, the radiologist initially favored a diagnosis of fibrous dysplasia, a benign process in which normal bone is replaced by fibro-osseous tissue and immature bone marrow. When it affects the craniofacial skeleton, fibrous dysplasia can present similarly to sphenoid wing meningioma, with proptosis and facial asymmetry in 80% of cases. Visual loss is present in 25% of patients at the time of diagnosis.1
Sphenoid wing meningioma has been known to mimic fibrous dysplasia radiographically, particularly if the tumor invades bone but not adjacent soft tissue.4 CT findings in fibrous dysplasia consist of a heterogeneous process with alternating radiodense and radiolucent areas and smooth margins. This is in contrast to the homogeneous hyperostosis and sclerotic appearance with “feathered borders” representing adjacent soft tissue involvement that is commonly seen in sphenoid wing meningioma.1
MRI is also quite helpful in differentiating between these 2 entities. In contrast to sphenoid wing meningioma, fibrous dysplasia has a variable appearance with low to isointense signal and mild to moderate contrast enhancement, no adjacent soft tissue involvement, and no dural tail sign. In our case, the dural tail was clearly evident.
Patient’s Progress
The skull base team successfully removed Ms. Mendel’s Grade I sphenoid wing meningioma. Three months later, her VA is now stable.
___________________________
* Patient name is fictitious.
___________________________
1 Katz BJ, Nerad JA. Ophthalmology. 1998;105(12):2207-2215.
2 Goli RS et al. Ophthal Plast Reconstr Surg. 1998;14(2):126-129.
3 Cornelius JF et al. Acta Neurochir (Wien). 2013;155(3):407-413.
4 Boon AP et al. J Neurol Neurosurg Psychiatry. 1990;53(9):818.
___________________________
Dr. Reiter is an ophthalmology resident, and Dr. Silkiss is chief of Ophthalmic Plastic, Reconstructive, and Orbital Surgery; both are at California Pacific Medical Center in San Francisco. Relevant financial disclosures: None.
Further Reading
Barresi V et al. Classification of human meningiomas: Lights, shadows, and future perspectives. J Neurosci Res. Published online July 4, 2016. doi:10.1002/jnr.23901
Goldberger R et al. EANO guidelines for the diagnosis and treatment of meningiomas. Lancet Oncol. 2016;17(9):e383-391. Published online Aug. 30, 2016. doi:10.1016/S1479-2045(16)30321-7
Leroy HA et al. Internal and external spheno-orbital meningioma varieties: Different outcomes and prognoses. Acta Neurochir (Wien). 2016;158(8):1587-1596.
|