Download PDF
Lens-induced glaucoma is a secondary glaucoma in which the crystalline lens is involved in the mechanism of intraocular pressure (IOP) increase. The glaucoma may occur in open-angle or angle-closure forms, and there are 4 distinct variants: phacolytic, lens-particle, phacoantigenic, and phacomorphic.
Phacolytic Glaucoma
Pathogenesis. Phacolytic glaucoma,as first described by Flocks and colleagues,1 was originally thought to be caused by obstruction of the trabecular meshwork by macrophages distended by engulfed lens material and Morgagnian fluid that had escaped from an intact crystalline lens. Later, Epstein et al.2 provided evidence for the role of high-molecular-weight soluble lens protein in causing direct obstruction of aqueous outflow channels.
In one series, Yanoff and Scheie3 reported a lack of IOP elevation in children at the time of cataract surgery, despite the presence of engorged macrophages in aspirates of their aqueous fluid. It has been demonstrated that high-molecular-weight proteins are rare in pediatric lenses, which may account for the infrequent occurrence of phacolytic glaucoma in children.
Regardless of the exact pathogenesis of phacolytic glaucoma, the condition occurs chiefly in the setting of a senile hypermature, or Morgagnian, cataract with leakage of lenticular material through microscopic openings in an apparently intact lens capsule.4 In rare cases, the cataract may be immature, with liquefaction of the posterior cortex.
Clinical features. Patients typically present with severe pain, a red eye, and blurry vision, with a history of a gradual decrease in vision over the preceding months or years. Although poor vision would be expected secondary to the advanced cataract, an acute reduction in vision is usually the result of corneal edema associated with the glaucoma.
On examination, the IOP is very high, accounting for the pain at presentation. The drainage angle is open, with no visible abnormality. Microcystic edema may be present in the cornea, and there may be scattered cells on the endothelium or endothelial precipitates. Often, an inflammatory reaction is present throughout the eye. Cellular reaction in the anterior chamber can vary from mild cells and flare to an intense reaction with pseudohypopyon.5 In phacolytic glaucoma, cells in the aqueous may be larger than the lymphocytes seen in other uveitic processes. These larger cells are thought to be swollen macrophages with engulfed lenticular material. Aggregates of macrophages may also be seen along the surface of the lens capsule.4
Diagnosis. While phacolytic glaucoma is a clinical diagnosis, microscopic examination of aspirated anterior chamber fluid can aid in suspected cases. Biochemical studies can help identify high-molecular-weight lens proteins that have leaked out of the cataract. Engorged macrophages may be seen as well.5
Treatment. Phacolytic glaucoma is typically handled as an emergency. Every effort is made to reduce the inflammation and IOP medically with topical steroids and topical aqueous suppressants (beta-blockers, alpha-2 agonists, and carbonic anhydrase inhibitors [CAIs]). Systemic CAIs and osmotic agents are sometimes needed as well. Despite these efforts, IOP elevation can remain recalcitrant or rebound on medical therapy; definitive treatment for patients with presumed phacolytic glaucoma is cataract extraction.
 |
|
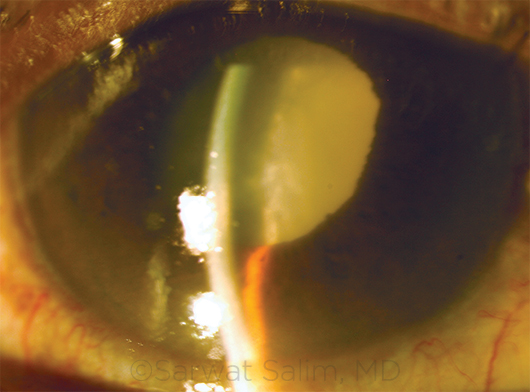
PHACOMORPHIC GLAUCOMA. This patient with a long history of reduced vision in the right eye presented with severe pain. Examination revealed a mature cataract, edematous cornea, shallow anterior chamber, and IOP of 60 mm Hg.
|
Lens-Particle Glaucoma
Pathogenesis. In lens-particle glaucoma, IOP elevation is caused by obstruction of aqueous outflow by lens particles. Like phacolytic glaucoma, this is a secondary open-angle glaucoma; however, in contrast to the phacolytic type, lens-particle glaucoma is associated with a grossly disrupted lens capsule and liberated fragments of lens material in the anterior chamber. It may occur after cataract surgery, trauma to the lens, or YAG posterior capsulotomy.4
Clinical features. Clinical findings of lens-particle glaucoma are similar to those of phacolytic glaucoma: conjunctival injection, corneal edema, elevated IOP, and anterior chamber reaction. Often, lens-particle glaucoma has a greater inflammatory component, associated with anterior and posterior synechiae and pupillary membranes.
Diagnosis. The diagnosis of lens-particle glaucoma can be made based on a history of recent intraocular surgery or trauma, along with the presence of gross lens material in the anterior chamber.
Treatment. Prompt treatment is crucial to avoid serious consequences, including intractable glaucoma due to peripheral anterior synechiae caused by continued inflammation.
Further, uncontrolled inflammation may lead to the development of a pupillary membrane and subsequent pupillary block. Cystoid macular edema and even tractional retinal detachments may also occur. Permanent damage to the aqueous outflow channels may result if the retained lens material is not removed from the eye.5
The severity of disease upon presentation dictates the course of treatment. If minimal cortical material is present, cycloplegics, corticosteroids, and aqueous suppressants may be employed. However, if inflammation is significant and the IOP cannot be controlled quickly, urgent removal of the residual lens cortex is necessary. Loose material can be removed with irrigation, but material adherent to ocular structures may require vitrectomy.
Phacoantigenic Glaucoma
Pathogenesis. Phacoantigenic, also known as phacoanaphylactic, glaucoma is the rarest type of lens-induced glaucoma and is often difficult to diagnose in vivo. The term phacoanaphylactic is a misnomer in that it does not involve an allergic or anaphylactic reaction; rather, the underlying mechanism may be an Arthus-type immune complex reaction, mediated by IgG and the complement system, against lens proteins.4 These proteins are normally immune-privileged antigens sequestered within the lens capsule. However, during a complicated cataract surgery with vitreous loss or trauma, a mixture of lens material and vitreous may occur, resulting in retention and subsequent slow release of sensitizing lens proteins.5 Phacoantigenic glaucoma typically occurs 1 to 14 days after cataract surgery, although there may be a longer latent period after sensitization to lens proteins.
Clinical features. The clinical signs of phacoantigenic glaucoma include eyelid edema, conjunctival injection, corneal edema, an intense anterior chamber reaction, posterior synechiae, and mutton-fat keratic precipitates. Anterior vitritis may also be present.4
Diagnosis. Definitive diagnosis requires the presence of polymorphonuclear leukocytes in the aqueous or vitreous specimen, as well as circulating lens proteins within the aqueous humor, the amount of which is usually insufficient to account for the severity of the glaucoma.5
Treatment. Treatment often begins with topical steroid therapy and antiglaucoma medications. However, as with the other types of lens-induced glaucoma, surgical intervention to remove the remaining lens material is often necessary.
Phacomorphic Glaucoma
Pathogenesis. A senile cataractous lens can become intumescent, increase in thickness, and cause pupillary block. This iridolenticular apposition disrupts the flow of aqueous humor from the posterior chamber to the anterior chamber. This results in the accumulation of aqueous in the posterior chamber, pushing the iris root forward, which may ultimately contact the trabecular meshwork and lead to angle closure. Risk factors predisposing to phacomorphic glaucoma include hyperopia, which is associated with a smaller anterior chamber.
Clinical features. The presentation of phacomorphic glaucoma is similar to acute angle-closure glaucoma. Patients may experience severe pain and headache secondary to elevated IOP, blurred vision, perception of halos around lights, nausea, vomiting, bradycardia, and sometimes diaphoresis.4 Clinical features may include corneal edema, conjunctival injection, and a mid-dilated pupil. The intumescent lens may be observed pushing the iris forward and reducing the anterior chamber depth. Anterior chamber cells and flare may also be present.
Diagnosis. Phacomorphic glaucoma is diagnosed clinically. Unlike the other types of lens-induced glaucoma, gonioscopy reveals a closed angle.
Treatment. Initial treatment is directed at lowering the IOP with aqueous suppressants or hyperosmotic agents. Depending on the degree of corneal edema, laser iridotomy is often recommended (also in the fellow eye, if it is anatomically predisposed to angle-closure glaucoma). Once IOP and inflammation are under control, definitive treatment with cataract extraction should proceed.
___________________________
1 Flocks M et al. Arch Ophthalmol. 1955;54(1):37-47.
2 Epstein DL et al. Invest Ophthalmic Vis Sci. 1978;17(3):272-277.
3 Yanoff M, Scheie HG. Arch Ophthalmol. 1968;80(2):166-170.
4 Papaconstantinou D et al. Clin Interv Aging. 2009;4:331-336.
5 Conner IP et al. Lens-induced glaucoma. In: Kahook M et al, eds. Chandler and Grant’s Glaucoma, 5th ed. Thorofare, N.J.; Slack; 2013:441-447.
___________________________
Dr. Laurenti is a senior resident in ophthalmology at the Medical College of Wisconsin. Dr. Salim is professor of ophthalmology and chief of the glaucoma service at Medical College of Wisconsin in Milwaukee. Relevant financial disclosures: None.