By James Reidy, MD, FACS, with Kathryn Hatch, MD, and Erin Stahl, MD
Download PDF
When it presents in children, keratoconus often involves an aggressive clinical course, and disease progression is not slowed by conservative management with spectacles or contact lenses. Corneal cross-linking (CXL) can stave off further corneal thinning, but it can be challenging to carry out in pediatric patients. In this first installment of a two-part series, James Reidy, MD, FACS, at the University of Chicago, leads a roundtable discussion with Kathryn Hatch, MD, of Massachusetts Eye and Ear Waltham and Harvard Medical School, and Erin Stahl, MD, of Children’s Mercy Hospital at the University of Missouri, Kansas City School of Medicine in Kansas City. The experts share their CXL technique and discuss special considerations for treating keratoconus in children. Part 2 will appear in the September EyeNet.
Prevalence and Severity
Dr. Reidy: Results of a long-term epidemiologic study from 1986 indicated that the prevalence of keratoconus in the general population was approximately one in 2,000.1 However, recent epidemiologic findings suggest a much higher prevalence: one in 375.2 Keratoconus usually is identified during a patient’s late teens to early twenties, but this condition increasingly is being diagnosed in the pediatric age group. Practitioners have noted that children often present with worse keratoconus that progresses more rapidly than in adults.3 What has been your experience?
Dr. Hatch: I see keratoconus in patients of all ages. My experience has been that young patients are at the highest risk of rapid progression, occurring sometimes within a few weeks. In children, the corneal tissues are more susceptible to ectasia, making CXL imperative for pediatric patients with keratoconus.
Dr. Stahl: I work in an exclusively pediatric practice and don’t have a comparison population of adult patients. I’ve seen rapid, significant progression of keratoconus in children, either while we’re waiting for insurance approval or between treatment of the first and second eye. This is why I often recommend managing keratoconus bilaterally, even if the better-seeing, less affected eye does not show much corneal thinning initially.
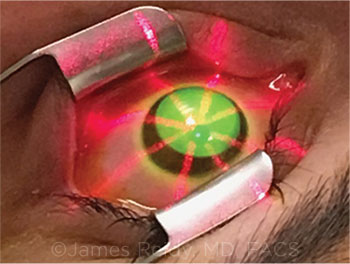 |
|
DRESDEN PROTOCOL. Application of UVA irradiation from the Avedro KXL machine.
|
CXL Protocol
Dr. Reidy: Which CXL protocol do you use to treat pediatric patients?
Dr. Stahl: I perform CXL by the standard Dresden protocol.4 I remove the central 9 mm of the corneal epithelium with alcohol assistance, and I apply one drop of Photrexa Viscous (Avedro) every two minutes for 30 minutes.
I assess the thickness of the cornea before epithelial removal, after epithelial removal, and at 15 and 25 minutes into the procedure. If I find significant thinning of the cornea at any of these time points, I switch from Photrexa Viscous to standard Photrexa (Avedro) for the remainder of the operation.
At 30 minutes, I verify that the corneal thickness is greater than 400 μm, and then I deliver ultraviolet A (UVA) light while continuing to apply Photrexa (standard or viscous) every two minutes for another 30 minutes. At completion of the surgery, I rinse the eye and apply a bandage contact lens, a drop of Vigamox (moxifloxacin; Alcon), and a drop of prednisolone.
Dr. Hatch: I don’t routinely use the same technique for all patients. I often perform the standard Dresden protocol. But, in some cases, I use an off-label, less-invasive partial epithelial removal technique, which I try to customize to the patient’s age, corneal thickness, and level of disease. Given the potential complications associated with epithelial removal in CXL, I try to minimize risk with a faster recovery by performing a less invasive transepithelial approach for some patients, especially in the pediatric population.
For my off-label epi-on technique, I load the cornea with Photrexa. I roughen the epithelium prior to loading the cornea. After about 40 minutes, I check the riboflavin load at the slit lamp and look for uniform saturation, check pachymetry, and follow with the UVA treatment. Sometimes I debride a small region over the cone—typically the central 3 to 4 mm of the cornea—to try to achieve deeper cross-linking in that area. For this, I have used either the Epi Clear epikeratome (Orca) or a 15-degree blade. After the debridement, I apply UVA light. At the end of the treatment, I administer antibiotic and steroid to the eye. In addition, if I had removed any epithelium, I place a bandage contact lens.
Indications to Treat
Dr. Reidy: Which signs do you consider to be indications to treat, and which are indications just to monitor?
Dr. Stahl: In my pediatric practice, retinoscopy is performed routinely. For me, the first signs of corneal warping that are visible on retinoscopy—including scissoring of the red reflex and the early cone—are indications to treat. When keratoconus is suspected, we also perform Scheimpflug tomography (Pentacam, Oculus) and anterior-segment optical coherence tomography. In most cases, results of these tests substantiate our retinoscopy findings.
Usually, pediatric patients present with keratoconus that is more severe in one eye than the other, and we treat the worse eye at diagnosis. If the best-corrected, or even uncorrected, visual acuity of the other eye is 20/20, I observe that eye while we visually rehabilitate the treated eye. As soon as I detect progression in the better-seeing eye, we go ahead and treat. Alternatively, I have had patients elect to receive treatment in the forme fruste eye, even if visual acuity was unaffected at the time. If the eye has topographic signs of keratoconus, patients sometimes want to undergo treatment proactively, andI think that’s reasonable.
Dr. Hatch: By the time a patient presents to me, I often know either through their records or medical history that the keratoconus is unstable.
I don’t believe keratoconus is a medical emergency, but there are cases, especially in pediatric and young patients, where it should be considered urgent. When the medical record indicates that corneal ectasia has progressed, I don’t wait to treat in pediatric, preteen, or teenaged patients. In general, in the past 15 months, I’ve found that insurance companies have become increasingly cooperative in covering CXL.
We know that keratoconus is a bilateral condition. I’m proactive about performing cross-linking in the affected eye as well as the forme fruste eye or less involved better-seeing eye. In most cases, patients and caregivers agree with this recommendation as they understand the goal of preserving vision. I routinely treat one eye at a time at least a day apart.
Treatment Challenges
Dr. Reidy: What are some obstacles associated with treating pediatric patients, and how do you manage these difficulties?
Dr. Stahl: At Children’s Mercy Hospital, I provide specialized care for children who need general anesthesia in order to undergo CXL, such as those with Down syndrome or anxiety, and those who were unable to receive CXL elsewhere. For pediatric patients who are older and able to handle the treatment without general anesthesia, I recommend undergoing CXL in the community setting, similar to the procedure an adult would receive.
Dr. Hatch: I primarily treat adult patients, but I do care for some pediatric patients and special needs patients with keratoconus. I evaluate the patient and perform a slit-lamp exam, which acclimates the child to the office and helps me determine whether I’ll be able to carry out CXL. I don’t perform CXL with general anesthesia. Instead, in our office-based setting, I give low doses of lorazepam, titrated by weight, and we employ “verbal” anesthesia. For instance, we encourage patients to choose music to play during the procedure, and, in certain situations, we invite parents or caregivers into the operating room to provide distraction. We take a team approach, and we have champion CXL technicians who are accustomed to caring for these children.
Infobox
According to Avedro, a total of 65 commercial plans now cover the KXL system and its associated Photrexa drug formulations. As of Jan. 1, 2019, the unique HCPCS J code J2787 was approved. Tip: Many treatments use 6 mL; in such cases, submit 2 in the unit field with a notation of any residual medication wasted.
|
Postoperative Care
Dr. Reidy: What is your post-op regimen?
Dr. Hatch: When I perform transepithelial CXL without epithelial removal—which is how I commonly treat the forme fruste eye—I prescribe a topical antibiotic ointment starting three days before the procedure, which is continued through five days postoperatively. I also treat with prednisolone four times a day for five days. In cases of CXL with epithelial removal, I prescribe a three-week tapered course of steroid, and I continue antibiotic treatment for two days after removal of the bandage contact lens, while re-epithelialization continues.
Dr. Stahl: I provide similar post-op care. The only difference is that I remove the epithelium in every case, so I routinely give antibiotic for five days postoperatively, and then I taper the steroid over a month.
With respect to follow-up, I monitor patients every day until the epithelium has healed and the bandage contact lensis removed. I began these frequent checks because I had some patients with sterile infiltrates or delayed healing after CXL. Daily post-op visits can be difficult to schedule, but they’re helpful for several reasons: 1) to reassure patients that their level of discomfort is normal and help them along, 2) to make sure the contact lens is in place and the surgical site is healing properly, and 3) to know if I need to titrate the steroid, based on the healing I observe.
Managing Pain
Dr. Reidy: What do you give for pain control in the first few postop days?
Dr. Hatch: Pain during the first postoperative day is a concern with the CXL procedure. I recommend high-dose ibuprofen and prescribe narcotics that patients typically take in the first 24-48 hours after surgery. I also suggest chilled preservative-free artificial tears and ice packs as needed.
Dr. Stahl: CXL is the only pediatric procedure for which I give narcotic pain medication. I consult with patients and caregivers about this. I tell them, “This is only to be used in case of emergency,” but I’d estimate that 50% of my patients take at least one of the oxycodone pills for pain management. I determine dosage based on the patient’s weight.
I also talk with patients before surgery to help them plan screen-free activities for their postoperative downtime. Often, these patients are very tied to their devices, and the screens may be too bright for them to use during the healing process.
In addition, we give patients little ice packs on the first postoperative day, and we encourage caregivers to dispense refrigerated artificial tears and do what they can to keep the patient as comfortable as possible without narcotic medication.
___________________________
1 Kennedy RH et al. Am J Ophthalmol. 1986;101(3):267-273.
2 Godefrooij DA et al. Am J Ophthalmol. 2017;175:169-172.
3 Naderan M et al. Int Ophthalmol. 2017;37(5):1169-1173.
4 Wollensak G et al. Am J Ophthalmol. 2003;135(5):620-627.
___________________________
Dr. Hatch is the director of refractive surgery service at Massachusetts Eye and Ear, site director of Mass Eye and Ear Waltham, and assistant professor of ophthalmology at Harvard Medical School in Boston. Relevant financial disclosures: Avedro: C.
Dr. Reidy is associate professor of ophthalmology and visual science and vice chair for clinical operations at the University of Chicago. Relevant financial disclosures: None.
Dr. Stahl is associate professor of ophthalmology at the University of Missouri, Kansas City School of Medicine. Relevant financial disclosures: Avedro: C.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Hatch Avedro: C,O; Carl Zeiss: C; EyePoint: C; Eyevance: C; Johnson & Johnson: C; Shire: C.
Dr. Reidy None.
Dr. Stahl Avedro: C; Retrophin: C,S; Treehouse Health: C,O.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|