By Gary N. Holland, MD, with Debra A. Goldstein, MD, James T. Rosenbaum, MD, and Russell N. Van Gelder, MD, PhD
Download PDF
Patients with uveitis and other inflammatory eye diseases may be treated initially, or even managed long-term, by nonuveitis specialists. For these practitioners, the selection and interpretation of diagnostic tests can be challenging. Here, in Part 1 of a 3-part series, Gary N. Holland, MD, of the UCLA Stein Eye Institute, hosts a roundtable with Debra A. Goldstein, MD, of Northwestern University’s Feinberg School of Medicine; James T. Rosenbaum, MD, of Oregon Health & Science University’s Casey Eye Institute and the Legacy Devers Eye Institute; and Russell N. Van Gelder, MD, PhD, of the University of Washington. These uveitis experts discuss their approach to the initial workup and share recommendations for ordering ancillary or invasive testing. Part 2 will cover interpreting exam findings, and Part 3 will provide treatment recommendations.
Standard or Tailored Workup?
Dr. Holland: Should a standard workup be performed for all patients who present with uveitis?
Dr. Goldstein: No. Because uveitis comprises many different conditions, I feel strongly that there should not be a standard workup. Other than evaluating all patients with uveitis for syphilis, my workup is guided by the patient’s history, a review of systems, and a clinical exam. For example, if a patient has acute anterior uveitis, I would consider ordering HLA-B27 testing, particularly if the patient is experiencing lower back pain and stiffness. For a patient with granulomatous disease, I would test for sarcoidosis and tuberculosis (TB)—the latter especially if I am planning systemic immunosuppression.
Dr. Rosenbaum: As a rheumatologist who sees patients with uveitis, I have a similar perspective. If uveitis can be classified from the patient’s medical history and exam, I don’t order any laboratory tests routinely. For patients with idiopathic uveitis, my standard workup is a chest x-ray—to look for sarcoidosis primarily and TB secondarily—and a syphilis serologic test.
Dr. Van Gelder: For a patient with uveitis, there are 3 reasons for a laboratory workup: 1) to ensure we don’t miss a treatable condition, such as an infection; 2) to rule out a concurrent systemic disease that requires treatment beyond the eye; and 3) to provide the patient with prognostic information about the disease and its anticipated course and outcome. I don’t perform any single standard workup for patients with uveitis. The patient’s history, demographic information, and disease presentation guide my choice of diagnostic tests.
Dr. Rosenbaum: Once treatment has begun (such as immunosuppressive medication), additional tests are ordered for monitoring. Because the decision-making process is shared between practitioner and patient, a lab study may be performed mainly to reassure the patient. Occasionally, a test may be ordered for research purposes.
 |
|
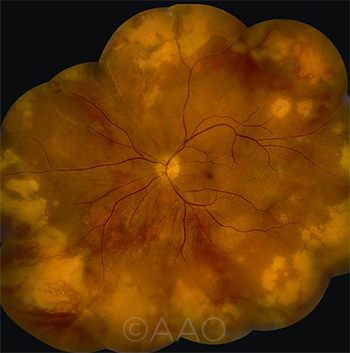
TIME FOR INVASIVE TESTING? If a diagnosis of necrotizing herpetic retinopathy is in doubt, anterior chamber paracentesis, with analysis of aqueous humor by PCR techniques, may provide confirmation of a causal organism.
|
Tests to Include
Dr. Holland: Are there any tests that you perform for every patient with uveitis?
Dr. Goldstein: I always order syphilis testing for patients with uveitis because syphilis is essentially the only easily curable cause of uveitis. I order specific treponemal tests and nontreponemal tests. Nowadays, I think that most U.S. labs conduct enzyme immunoassay for syphilis, and if the results are positive, a nontreponemal test is performed, such as an RPR (rapid plasma reagin) or VDRL (Venereal Disease Research Laboratory).
I treat many patients from TB-endemic areas. I always evaluate these patients for TB with Quantiferon Gold (Quest Diagnostics), an interferon gamma release assay (IGRA). I also test all patients for TB, regardless of possible TB exposure, prior to starting TNF inhibitor therapy. If testing is positive, therapy for latent TB should be started prior to initiation of TNF inhibitor therapy.
Dr. Holland: Syphilis is a disease that you don’t want to miss, especially because there is substantial morbidity associated with untreated syphilis, and there are public health implications for undiagnosed disease; however, I don’t order syphilis serologic tests for a patient if I already have an established diagnosis for the uveitis.
Tests to Omit
Dr. Holland: Are there laboratory tests that are not useful in the evaluation of patients with nonspecific intraocular inflammation?
Dr. Van Gelder: For adult patients, an antinuclear antibody (ANA) test has limited utility in the uveitis workup. Systemic lupus erythematosis is a clinical diagnosis, and an ANA can be supportive of its diagnosis. However, anterior uveitis, intermediate uveitis, or even panuveitis are uncommon presentations of lupus. Lupus may be associated with scleritis and rarely retinal vasculitis—and in those cases, there may be accompanying uveitis—but perhaps with the exception of those cases, I see many more ANAs ordered than I think are useful.
The one exception is in children, where the ANA carries significant prognostic information for uveitis associated with juvenile idiopathic arthritis.
Some other rheumatologic tests also have limited utility for uveitis. I rarely find erythrocyte sedimentation rate (ESR) to be helpful in terms of systemic disease, prognosis, or treatment; tests for C-reactive protein and complement levels typically are unnecessary for most cases of uveitis.
Dr. Rosenbaum: Screening for Lyme disease is rarely applicable to uveitis. Although Lyme disease is a cause of uveitis, it’s an extremely rare cause, and approximately 10% of the population has a positive test. If you screen everyone for Lyme disease, you’ll have many patients with positive test results who do not have uveitis secondary to the disease. Gallium scanning and analysis of angiotensin-converting enzyme (ACE) and lysozyme are rarely helpful because of the low sensitivity and specificity of these modalities.
ESR is an inexpensive test that can be useful for evaluating bowel symptoms when it is unclear if the patient has irritable bowel syndrome or an inflammatory bowel condition that could be associated with uveitis or scleritis. Although not definitive, the ESR may suggest that bowel symptoms have an inflammatory origin. Overall, tests with low general utility should be ordered very selectively.
Dr. Goldstein: Occasionally, I order an ANA for an adult with scleritis, but never for adults with uveitis. We can get stuck chasing borderline elevated test results, ordering unnecessary testing, and causing the patient to worry. In the absence of scleritis, I would avoid rheumatoid factor testing because rheumatoid arthritis does not cause anterior uveitis.
I think that ACE and lysozyme levels can be useful indicators of inflammation in the body, and ESR testing can be helpful for patients with vasculitis, for example. Although nonspecific for sarcoidosis, I think that results of ACE and lysozyme testing can suggest granulomatous disease, and these tests are inexpensive. For a patient with suspected sarcoidosis, I would order ACE testing but acknowledge that the results would not allow for definitive diagnosis.
Ancillary Testing
Dr. Holland: What are your thoughts about ancillary tests such as fluorescein angiography and optical coherence tomography (OCT) of the macula? Do you order them frequently, and what are your indications for doing so?
Dr. Van Gelder: As with serologic and radiologic testing modalities, the patient’s presentation guides my choice of ophthalmic imaging modalities. There is no tool equivalent to OCT for diagnosing and monitoring cystoid macular edema, a major cause of visual morbidity in patients with uveitis. OCT allows observation of vitreomacular traction and other noninflammatory causes of edema and yields quantitative information regarding the patient’s response to therapy.
Indications for fluorescein angiography include suspicion of retinal vasculitis, phlebitis, or white dot syndromes. Fluorescein angiography also is appropriate for differential diagnosis of acute posterior multifocal placoid pigment epitheliopathy and serpiginous choroiditis. Fundus autofluorescence imaging is very helpful for diagnosing white dot syndromes. Occasionally, indocyanine green (ICG) angiography may be indicated in subtle cases of birdshot chorioretinopathy.
Dr. Goldstein: Like Dr. Van Gelder, I perform OCT frequently. Cystoid macular edema is the most common cause of decreased vision in most forms of uveitis, and its presentation can be subtle. I use fluorescein angiography often in patients who have retinal vasculitis or a white dot syndrome.
Invasive Testing
Dr. Holland: Do you perform invasive tests, such as aqueous paracentesis or retinal biopsies, for diagnostic purposes?
Dr. Goldstein: Aqueous paracentesis is safe and easy to perform in the clinic. I use aqueous paracentesis in many cases of suspected herpetic disease (anterior uveitis or retinitis), especially when the results of this test would influence my choice of treatment. In cases of necrotizing herpetic retinitis, it’s easy to make the diagnosis from an anterior chamber paracentesis without having to go into the vitreous cavity. If a patient has clinical features of herpetic anterior uveitis (which may be secondary to herpes zoster or infection with herpes simplex virus), I usually treat empirically without paracentesis. However, if I suspect anterior uveitis secondary to cytomegalovirus (CMV) infection, I would perform aqueous paracentesis for subsequent PCR (polymerase chain reaction) analysis. Whereas valaciclovir, which we use to treat herpes simplex and zoster, is well tolerated, valgancyclovir, the preferred systemic therapy for CMV, is expensive and has significant side effects, including neutropenia. Therefore, I would not treat empirically with valganciclovir for CMV anterior uveitis without PCR confirmation.
Recently, I saw an immunosuppressed patient with necrotizing retinitis. The retinitis looked typical for herpetic retinitis; however, there was an atypical finding for herpes of posterior vitreous precipitates. This feature is much more common in toxoplasmic retinochoroiditis. I performed aqueous paracentesis for PCR, which was positive for Toxoplasma gondii and negative for all herpes viruses. In this atypical case, the PCR helped guide treatment.
Dr. Van Gelder: I advise retinal biopsy very rarely and mainly for cases in which a tissue diagnosis would impact the patient’s treatment course. For instance, I saw a patient who presented with multifocal choroiditis. The results of her chest x-ray were equivocal: probable granulomatous disease. Her IGRA (Quantiferon) result was positive, but she was a former nurse in her 60s, and we had to determine whether we should treat her for TB or sarcoidosis. She received 4-drug tuberculosis therapy but did not tolerate it, and she had vision-threatening disease. This was a very challenging scenario: Do we immunosuppress a patient who may have TB, or do we assume this is TB-associated uveitis? In this case, we felt that a tissue diagnosis was essential. The patient underwent chorioretinal biopsy with subsequent PCR, and the results were negative for Mycobacterium tuberculosis. Consequently, we suspected sarcoidosis, and the patient received immunomodulation therapy. Her disease responded to this treatment without evidence of systemic activation of TB. However, cases like this are extremely rare.
Because morbidity is common with retinal biopsy, we reserve this procedure only for cases of choroidal masquerade syndrome or similar diagnostic dilemmas in which the stakes are high.
Dr. Holland: How do you evaluate a patient with a suspected masquerade syndrome if you think the person may have intraocular B-cell lymphoma?
Dr. Van Gelder: There is no widely accepted testing protocol. However, we always begin with a patient history. If I observe any neurologic signs, I would order magnetic resonance imaging (MRI). I have not found lumbar puncture (LP) to be a high-yield procedure [for isolating lymphoma cells]. When my neurology colleagues have a patient with negative MRI results, they generally do not rush to perform an LP to check for central nervous system (CNS) lymphoma.
For a patient in their 50s or older who presents with unilateral or bilateral vitritis, with or without a chorioretinal process occurring, and whose workup is otherwise negative, we typically go straight for a tissue diagnosis. Specifically, we’d perform a core vitrectomy, transfer the material to RPMI media, and conduct cytology and flow cytometry analyses. Other tests also are effective in these cases. For instance, a laser capture microdissection candetect IgH gene rearrangements with high sensitivity and specificity. And there is evidence that the interleukin-10 level in the aqueous is a relatively sensitive indicator of lymphoma. These tests may be excellent for initial screening. However, in my experience, when it comes time to coordinate care with our oncology colleagues, the tumor board wants to see a tissue diagnosis.
Dr. Rosenbaum: I agree. Brain MRI is useful because most patients with intraocular lymphoma will have associated CNS lymphoma. Even if results of an initial screening were negative but I was still concerned about an intraocular lymphoma, I would feel reassured that there would be some time before the disease progressed to a fatal outcome. As Dr. Van Gelder noted, analysis of the cerebrospinal fluid may allow you to avoid vitrectomy, but the yield of this procedure frequently is low.
Dr. Goldstein: I would be cautious about assuming that a normal MRI mitigates against the diagnosis of primary vitreoretinal lymphoma. We very often see patients with tissue diagnosis of lymphoma in the eye, who present without identifiable CNS lesions. I would add that if the patient is receiving systemic steroid therapy, it is important that we try to taper the steroid treatment before vitrectomy to increase the yield of the biopsy. Moreover, for vitreous biopsy, submitting an undiluted vitreous specimen as well as the vitreous cassette can help improve the yield.
___________________________
Listen to the roundtable below:
Download Audio
___________________________
Dr. Holland is Professor of Ophthalmology and Jack H. Skirball Chair in Ocular Inflammatory Diseases, David Geffen School of Medicine at University of California, Los Angeles, UCLA Stein Eye Institute. Relevant financial disclosures: None.
Dr. Goldstein is Magerstadt Professor of Ophthalmology at Northwestern University Feinberg School of Medicine in Chicago. Relevant financial disclosures: None.
Dr. Rosenbaum is Professor of Ophthalmology, Medicine, and Cell Biology; Head of the division of Arthritis and Rheumatic Diseases; and Edward E Rosenbaum Professor of Inflammation Research at Oregon Health & Science University in Portland. He is also the Richard Chenoweth Chair of Ophthalmology at Legacy Devers Eye Institute in Portland. Relevant financial disclosures: Up-to-Date: P.
Dr. Van Gelder is Boyd K. Bucey Professor and Chair of Ophthalmology at the University of Washington in Seattle. Relevant financial disclosures: None.
For full disclosures and the disclosure key, see below.