Download PDF
Is it glaucoma? Or is it a condition that looks like glaucoma? Ophthalmologists assessing patients with high myopia often face this diagnostic dilemma. “High myopes may not have glaucoma, but the presentation looks like glaucoma. Or they may have glaucoma, and we are not sure,” said Simon K. Law, MD, at the Jules Stein Eye Institute.
These questions take on special urgency in light of what appears to be an almost epidemic explosion of myopia, particularly in Chinese populations (see “The Chinese Connection”).
The link between myopia and glaucoma has been explored for nearly a century.1 A number of studies, including the Blue Mountains Eye Study and the Beijing Eye Study, have found that the risk of glaucoma appears to increase in persons with high myopia (6 D or more).2,3 Recently, Kuldev Singh, MD, MPH, at Stanford University, and Shan C. Lin, MD, at the University of California, San Francisco, reported a similar association in a U.S. population.4 Dr. Law added that patients with mild myopia appear to follow a course similar to those without myopia.
While they acknowledge the link between myopia and glaucoma, Drs. Law, Singh, and Lin are among a group of glaucoma experts who ask: How do you distinguish patients with myopia and glaucoma from those who have myopia-related visual field defects that may mimic glaucoma—but are not destined to show progression characteristic of glaucoma?
| Myopia ± Glaucoma |
 |
|
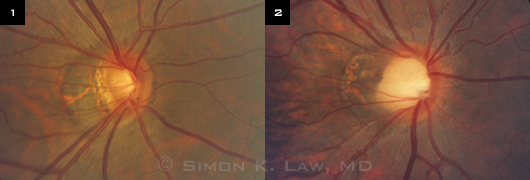
Optic nerve appearance of two highly myopic patients: (1) diagnosed with glaucoma; (2) without glaucoma.
|
How Myopes Are Different
Optic nerve findings. The presentation in these challenging cases is anything but classic. For starters, the optic nerve may look different from the characteristic glaucomatous optic nerves that clinicians have been trained to identify. “We call them ‘funny-looking’ optic nerves,” Dr. Lin said (Figs. 1 and 2).
Dr. Law put it this way: “The difficulty with diagnosis is because the optic nerve looks abnormal, and we base diagnosis on the optic nerve. But the nerves [in myopes] sometimes look different from nonmyopic patients. And the pressure may not be too high. They may even have visual field defects, but [these] don’t progress or progress differently. So we have a diagnosis problem and then a following problem.”
Visual field defects and progression. “A focus of our research is to try and determine whether or not those who are diagnosed with myopia and glaucoma follow a course similar to the classic presentation of primary open-angle glaucoma [POAG],” said Dr. Singh.
He explained that classic untreated POAG often presents with an arcuate visual field defect in one hemifield, followed by similar loss in the other hemifield, split fixation, and ultimately—in those with severe disease—loss of all vision. Dr. Singh hypothesized that myopia may be associated with regional damage to an optic nerve, sometimes the superior or inferior rim, with relative sparing of the other half of the nerve. “While such asymmetry between the superior and inferior nerve bundles is characteristic of most patients with glaucoma, those with myopia may perhaps be less likely to follow the same progressive POAG pattern, where the other half of the nerve and corresponding visual field ultimately become damaged.”
Dr. Singh reported observing this phenomenon in young Chinese myopes who presented with steep-edged visual field defects and commonly had “normal” intraocular pressure (IOP). This presentation resulted in a diagnosis of POAG or glaucoma suspect.5 Although these eyes may show extension of existing defects, they appear to less commonly exhibit new field defects in the opposite hemifield in later years. Still, he cautioned that this observation needs further study. In the meantime, Dr. Singh said that not all progression should be considered equally significant. “There is greater concern when patients show new field defects corresponding to regions of the optic nerve previously considered healthy.”
 |
|
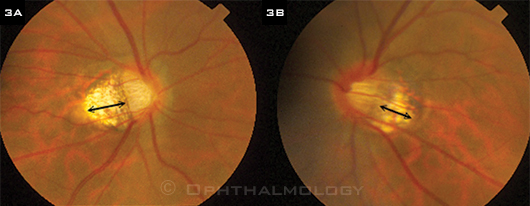
TILTED DISCS. (3A) Right and (3B) left optic nerves of highly myopic Chinese man with bilateral tilted discs and peripapillary atrophy (double arrows). At presentation, he had a dense superior field defect on the right and an early arcuate defect on the right and was being treated with antihypertensive eyedrops. During five years of follow-up and continuing topical treatment, the field defects remained stable.
|
Pearls From the Experts
Dr. Singh warned against “lumping everyone into one POAG category and treating them exactly the same.” For example, a 36-year-old myope with a tilted optic nerve and a visual field defect who is labeled as having POAG may have a very different disease than the 80-year-old nonmyope who carries the same diagnosis, he said.
Here is advice from the experts on how to assess and manage patients with high myopia and “funny-looking” optic nerves.
Dr. Law offered his recommendations for following myopic patients who are suspected of having, or are diagnosed with, glaucoma.
- Follow the nerve; you still have to rely on it. Take pictures of the optic nerve to document its appearance over time. If you see changes, then you know for sure that the glaucoma is progressing.
- Remember that visual field change isn’t always the result of glaucoma but may represent other types of optic nerve damage. For example, nerves that tilt inferiorly (Fig. 3) may have visual field defects at the top hemifield. Other visual field defects may be caused by myopic retinal degeneration, not glaucoma.
- Be mindful of the pressure. Eyes with high myopia may be less able to tolerate IOP fluctuations than other eyes: They have a longer globe, thinner lamina cribrosa, and thinner scleral wall with different elasticity. An IOP increase may be sufficient to cause damage in the posterior pole without causing abnormal corneal IOP readings.
In addition, because of the myopic eye’s elongated or ellipsoid shape, its posterior wall may be stretched more than the nonmyopic eye’s as IOP increases, making the optic nerve more susceptible to damage.
- Be cautious about doing glaucoma surgery. People with high myopia have a greater chance of hypotony maculopathy after surgery, particularly young high myopes, who have a thinner, less rigid scleral wall. In such patients, the scleral wall tends to collapse easily when the pressure is lowered after glaucoma surgery.
- Watch out for former high myopes, that is, patients whose myopia was reduced or eliminated by refractive surgery. Even though their refractive status has been improved by a keratorefractive procedure, their elongated eye structure and optic nerve status remain unchanged.
Ask these patients if they have had LASIK. Because that procedure thins the cornea, IOP measurements may be artifactually low. There are no good nomograms to tell how much the pressure will change, and the change is not linear. These patients should be followed carefully.
Code these post-LASIK patients as “high-risk glaucoma suspects.”
Dr. Lin noted several points to consider in deciding whether to initiate treatment.
- Treat each case differently, taking into account family history, degree of visual field defect, and your own comfort level with following versus treating.
- A large proportion of high myopes truly have glaucoma. If you’re not sure, you may want to treat it as if it’s glaucoma if the visual field defect is severe or very close to fixation.
- Drugs for treating glaucoma have a relatively safe systemic side effect profile, so it’s reasonable to err on the side of caution and treat.
- Recognize when the visual field pattern does not follow the typical pattern of glaucoma. For example, in myopic atrophy of the retina, the visual field usually doesn’t respect the horizontal meridian. In contrast, in glaucoma, when there is focal damage to the optic disc, it usually does not cross the horizonal meridian. If it crosses that midline, or is isolated in the periphery, it may not be glaucoma. But the physician must bear in mind that there are no hard-and-fast rules for these difficult diagnoses.
Dr. Singh cautioned against always treating these patients aggressively.
- Avoid filtration surgery, if possible. While I’m happy to treat with topical glaucoma medications, which are relatively safe, one should be cautious in setting a very low IOP goal at presentation, particularly in those with mild disease without a documented rate of prior progression. In these patients, early filtration surgery to achieve such goals not attained by medications may not be warranted.
- Don’t assume that moderate field loss in a young patient confirms rapid recent progression unless you have old data to confirm such a course.
- Progression of disease while under treatment with medications and laser trabeculoplasty does not always justify filtration surgery. Given the inherent risks involved, surgery is most appropriate in patients in whom the location and rate of progression suggest a significant risk of the patient becoming symptomatic from the disease.
The Chinese Connection
The growing interest in the role of myopia as a risk factor for glaucoma has been fueled by a startling increase in myopia, particularly in Chinese populations. For example, a study of 5,060 Chinese university students in Shanghai found that 95.5 percent of the students were nearsighted (defined in this study as –0.5 or worse), and 19.5 percent had high myopia.1
City life. The phenomenon, said Dr. Lin, appears related to rapid urbanization. “Environment appears to play a role,” he said. Children who once spent much time outdoors playing or doing farm chores are now spending time indoors studying and reading. Yet they need bright sunlight for normal development.2 “Your mother was right. If you read too much, you’ll become nearsighted. Except it now appears that the confounding factor is spending time outside.”
Dr. Singh noted that the sudden increase is occurring in certain populations, particularly Chinese, where myopia has been historically uncommon. “There has been a refractive shift in Chinese populations from hyperopia and short eyes to myopia and longer eyes. This is most evident in urban areas, where this shift has occurred with a concomitant increase in open-angle versus angle-closure glaucoma.”
Consider the overall clinical picture. This phenomenon is more than a matter of statistical interest. While some Chinese glaucoma patients are destined for poor outcomes that require glaucoma filtration surgery, Dr. Singh believes that there is a subset at risk of overtreatment. Perhaps these patients are destined to follow a very different course than young African-Americans presenting with equally severe visual field loss, though often in the setting of high IOP.
Further studies, said Dr. Singh, may determine whether it is appropriate to conceptualize a diagnostic subcategory under “glaucoma suspect.” One possibility: “Chinese myopia-related optic neuropathy suspicious for glaucoma.”
___________________________
1 Sun J et al. Invest Ophthalmol Vis Sci. 2012;53(12):7405-7509.
2 Wu PC et al. Ophthalmology. 2013;120(5):1074.
|
More Data Needed
Dr. Law wants to compare the progression of glaucoma in high myopes with that in patients who don’t have high myopia. He is seeking answers to the following questions: Are there different risk factors for progression in these groups? What is the influence of patient characteristics—higher or lower pressure, more myopia, Asian versus Caucasian?
Dr. Lin agreed that there are many unanswered questions, boiling down to who is most likely to progress—and, thus, who really needs treatment. “In the past, there wasn’t quite the need, but the recent explosion of myopia breeds necessity,” he said. “We need prospective data to say how likely person X is to get glaucoma. There may be racial differences. We haven’t done the study targeting this specific risk factor.”
In the meantime, as Dr. Singh noted, “We don’t have a crystal ball on these patients. We don’t know if they behave as classic glaucoma patients, so unless you think vision is immediately threatened, it’s best to avoid early filtration surgery. It’s best to initially follow these patients more conservatively, using safe medications whenever necessary. One often has the luxury of time.”
1 Knapp A. Arch Ophthalmol. 1926;55(1):35-37.
2 Mitchell P et al. Ophthalmology. 1999;106(10):2010-2015.
3 Xu L et al. Ophthalmology. 2007;114(2):216-220.
4 Qui M et al. Invest Ophthalmol Vis Sci. 2013;54(1):830-835.
5 Doshi A et al. Ophthalmology. 2007;114(3):472-479.
___________________________
Simon K. Law, MD, is clinical professor of health sciences at Jules Stein Eye Institute, University of California, Los Angeles. Financial disclosure: Has received speaker’s fees from Alcon, Allergan, and Tissue Bank International.
Shan C. Lin, MD, is professor of clinical ophthalmology and codirector of the glaucoma service at University of California, San Francisco. Financial disclosure: Is on the advisory boards of Alcon, Allergan, Merck, and Sucampo.
Kuldev Singh, MD, MPH, is professor of ophthalmology and director of the glaucoma service at Stanford Medical School, Stanford, Calif. Financial disclosure: Is a consultant to Alcon, Allergan, Ivantis, Santen, Sucampo, and Transcend.
More at the Meeting
Learn more about solving the diagnostic challenges presented by high myopes during Section 1, “Diagnostic Tests in Glaucoma,” at Glaucoma Subspecialty Day, Saturday, Nov. 16. From 8:38 to 8:54 a.m., join Felipe A. Medeiros, MD, Ki Ho Park, MD, PhD, and Nathan M. Radcliffe, MD, as they provide their perspectives on the case of a highly myopic patient, including considerations for appropriate monitoring of such patients and the role of corneal biomechanics.
|