Download PDF
Rates of diagnostic error of neuro-ophthalmologic conditions prior to evaluation by a neuro-ophthalmology specialist may be as high as 60% to 70%, according to Valérie Biousse, MD, at Emory Eye Center in Atlanta. “This results in mismanagement, delayed diagnosis, worse outcomes, and increased costs.”
In a recent referral pattern study of 300 patients, Dr. Biousse and coauthors found that the median time from symptom onset to neuro-ophthalmology consultation was 210 days.1 About 40% of referred patients had been misdiagnosed, 49% had been at least partially misdiagnosed, and—reflecting the complexity of this subspecialty—7% had unknown diagnoses after neuro-ophthalmologic evaluation.
What are the main reasons for these diagnostic errors? These range from time constraints to a lack of appropriate training to the expected inability to systematically review brain imaging studies, said Dr. Biousse. “Facilitating rapid access to a neuro-ophthalmologist has the potential to protect patients from harm, improve patient outcomes, and decrease the financial burden of inappropriate utilization of diagnostic tests and treatments triggered by these misdiagnoses.”
With neuro-ophthalmologists limited in numbers, however, that’s easier said than done. Yet, ophthalmologists can take steps to facilitate more timely—and accurate—diagnoses.
 |
|
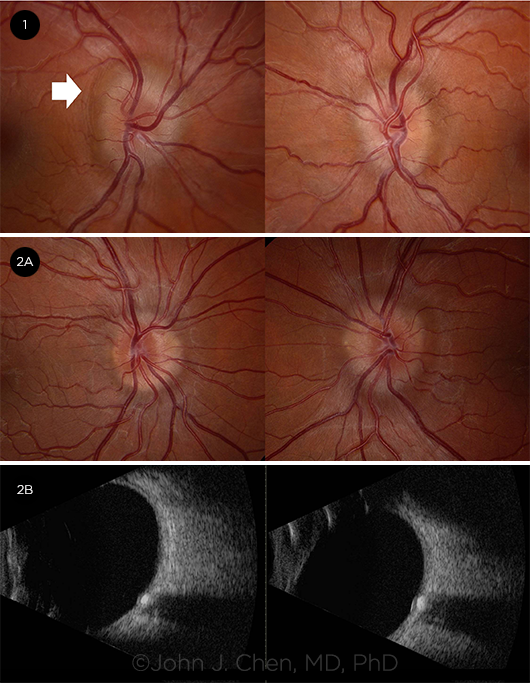
PAPILLEDEMA VS. PSEUDOPAPILLEDEMA. (1) Fundus photos of a 24-year-old woman with grade 2 papilledema from IIH. There are subtle Paton folds in the right eye indicating this is true papilledema (white arrow). (2A) Fundus photos of a 19-year-old man with pseudopapilledema from optic disc drusen. (2B) Ultrasound shows echogenic lesion within the optic nerve head in both eyes of the 19-year-old confirming optic disc drusen.
|
Addressing Barriers to Correct Diagnosis
Ophthalmologists should be able to diagnose and appropriately manage or expediently refer most simple or emergent neuro-ophthalmologic disorders, said Dr. Biousse.
That said, many ophthalmologists don’t have the opportunity to see patients with neuro-ophthalmologic complaints daily, so, of course, their comfort level isn’t as high as that of a neuro-ophthalmologist, said Courtney E. Francis, MD, at the University of Washington School of Medicine in Seattle.
A resulting issue for ophthalmologists is not recognizing what’s dangerous—and what’s not, said John J. Chen, MD, PhD, at Mayo Clinic in Rochester, Minnesota. “Some patients get referred urgently for nondangerous things, but others are referred as ‘next available’ when it could be life-threatening.” A few tips can help the ophthalmologist make this distinction.
Improve pattern recognition. First steps toward accurate diagnosis are the ability to recognize patterns, ask the right questions, and tailor the exam and testing to the patient’s complaint, said Larry P. Frohman, MD, of Rutgers-New Jersey Medical School in Newark. “We hope all ophthalmologists will develop the expertise to tell the radiologist, for example, ‘By my exam, I think this is a pituitary tumor, so please look carefully in that area.’”
Take time when you need to. Time pressure may push ophthalmologists to take shortcuts and skip essential parts of the clinical exam, said Dr. Biousse. “If a patient has red flags such as acute vision loss, optic disc edema, pain with eye movements, or accompanying neurological symptoms,” said Dr. Chen, it’s important to take more time to tease out how urgently that patient needs to be evaluated. “However, even if a patient’s case seems routine at first glance, I always ask myself, ‘What is the most likely diagnosis and more importantly, what is a diagnosis that can’t be missed?’”
Avoid false paths. Prem S. Subramanian, MD, PhD, is at the University of Colorado School of Medicine in Aurora. He noted that if you don’t get a thorough history and detailed exam, you may anchor on one element of the history, guiding you down a false path. It’s easy to pay attention to the findings that confirm the initial incorrect suspicion, Dr. Chen agreed, adding that neuro-ophthalmologists are also not immune to this pitfall.
Learn from pupils. A key part of the neuro-ophthalmologist’s exam is a check of the pupils, especially when evaluating vision loss or a potential third cranial nerve (CN III) palsy, said Dr. Francis. Because office protocol for many ophthalmologists often includes dilation before the doctor sees the patient, their technicians also must be trained to recognize concerning symptoms for which the pupil exam may be crucial, and dilation withheld.
Be prepared. “Ophthalmologists need to have a preestablished pathway including a network with a neuro-ophthalmologist, neurologist, and dedicated emergency department where brain imaging and specialists are readily available,” said Dr. Biousse. They also should develop a working relationship with a neuro-radiologist, if possible.
Pinpoint the problem and seek guidance. “Use your exam and history to localize the likely problem and tell the radiologist to scan and focus on that area of interest,” said Dr. Frohman.
What if you’re not sure which imaging modality to order and you don’t have quick access to a neuro-ophthalmologist? “You can at least touch base with the radiology department and describe your concern,” said Dr. Francis. “Then ask, ‘What is the best imaging study to help answer this question?’”
Consider imaging options. Optical coherence tomography (OCT) and visual fields are important elements of the workup of vision loss, said Dr. Chen. “Sometimes patients are referred to a neuro-ophthalmologist for an optic neuropathy without having had an OCT. I do the OCT, and it shows a retinal problem.”
In addition to the type of test, consider the technique, said Dr. Frohman. “Ophthalmologists often forget about needing a contrasted study. I could retire if I had a dollar for every time an MRI without contrast was read as normal, but was abnormal with contrast.”
Other imaging tips. In addition, Dr. Subramanian advises:
- Only order tests if you have a good understanding of what you will do with the information.
- Beware of making a diagnosis based on a single testing abnormality, especially if it contradicts other data.
- Order the correct imaging study. For example, computed tomography (CT) is best when looking for a bony abnormality; MRI is best when looking for an optic nerve abnormality.
- Be alert to artifacts.
- Keep an open mind and be willing to reconsider your diagnosis. Revisit the information and make sure what you thought at the last visit is still true.
 |
|
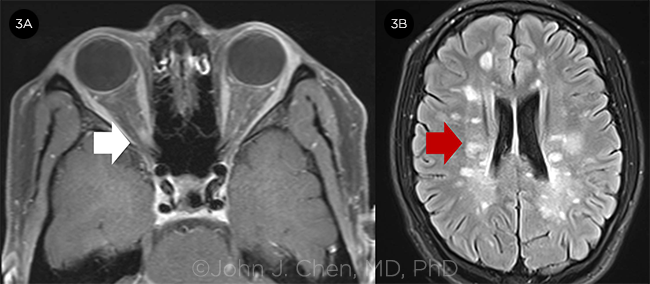
OPTIC NEURITIS. A 17-year-old girl with optic neuritis in the right eye. (3A) MRI orbits with contrast shows enhancement of the right optic nerve (white arrow). (3B) MRI FLAIR shows prominent periventricular white matter lesions consistent with multiple sclerosis.
|
 |
|
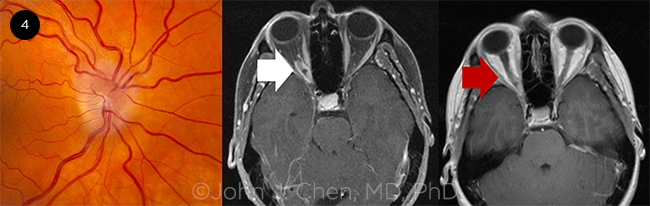
OPTIC NERVE SHEATH MENINGIOMA. A 59-year-old woman with an optic nerve sheath meningioma along the right optic nerve causing optic disc edema in the right eye. MRI orbits with fat saturation and contrast shows enhancement of the optic nerve sheath on the right in the classic “tram track appearance” of an optic nerve sheath meningioma (white arrow). The optic nerve sheath meningioma is not visible on the MRI without fat saturation (red arrow).
|
Four Common Misdiagnoses
Don’t make the mistake of misdiagnosing—or missing—the conditions below. A few insights and tips from the experts can guide you toward the correct diagnosis.
Idiopathic intracranial hypertension (IIH). IIH is due to high pressure in the brain without a known cause. A recent review coauthored by Dr. Biousse and colleagues found that nearly 40% of patients referred to neuro-ophthalmologists for IIH had been misdiagnosed, with pseudopapilledema being the most common correct diagnosis.2 Several factors may contribute to erroneous diagnosis, including the following:
Examination errors. Errors can originate with performance of the ophthalmoscopic exam or the interpretation of its findings. Why? “A patient’s optic nerves may look fuller, but not be truly swollen,” explained Dr. Chen. “The patient might have optic disc drusen or a congested optic nerve, or just have been born with anomalous-looking nerves.”
Demographic delusion. “Demographics, combined with headache and fundus findings, can mistakenly lead to a diagnosis of IIH,” said Dr. Subramanian. A common mistake, he said, is that the ophthalmologist anchors on the fact that the patient is a young, obese woman and forgets that two-thirds of Americans are overweight or obese—but a very small minority has IIH. This is an example of aberrant pattern recognition, he noted.
Hypervigilance. IIH is drilled into residents during training, said Dr. Francis. “The message is: You really don’t want to miss this one.” But misdirected conscientiousness has a clear downside. Before receiving a neuro-ophthalmologic consultation, many patients undergo unnecessary testing or treatment: Eight in 10 patients misdiagnosed with IIH receive spinal taps, about one-third have brain MRIs, and 76% get medical treatment.2 “Some even have surgery, which is dangerous,” said Dr. Chen.
Optic neuritis. Another retrospective review found even more errors with optic neuritis.3 The study found a misdiagnosis rate of nearly 60%, and the most common correct diagnosis was migraine. Dr. Frohman has found that other common diagnoses mislabeled by referring ophthalmologists as optic neuritis are nonarteritic anterior ischemic optic neuropathy (NAION) and retinal causes of visual loss. As with IIH, incorrect diagnoses of optic neuritis can lead to unnecessary imaging and treatments and even the inappropriate diagnosis of a disorder such as multiple sclerosis or neuromyelitis optica with resultant patient anxiety and distress.2
The experts provide a few tips to get to a correct diagnosis more quickly.
Avoid a single focus. A combination of blurry vision and pain with eye movement is characteristic of optic neuritis but is by no means specific, said Dr. Subramanian. “However, if you think you know that the diagnosis is optic neuritis, you may ignore other aspects of the exam.” As a result, you may overlook other potential causes, said Dr. Chen, such as dry eye or even functional vision loss.
RAPD is key. Often the only objective finding for optic neuritis on the eye exam is a relative afferent pupillary defect (RAPD), said Dr. Francis. “If the patient does not have a RAPD,” said Dr. Subramanian, “think long and hard why they don’t.” Of note, said Dr. Francis, if the patient’s pupils have already been dilated, then the ophthalmologist is at a diagnostic disadvantage.
Time course is a clue. The patient with optic neuritis tends to have a clear history of fairly sudden vision loss and pain, which can progress over a week, said Dr. Frohman. “This is not a situation where the patient suddenly wakes up with no vision in one eye and has blindness that remains consistent.” In addition, said Dr. Subramanian, if the pain lasts for one to three months, you can be more comfortable saying, “Maybe it’s not optic neuritis because the history doesn’t fit.”
Note a loss of color perception. This is another factor that can help pinpoint the diagnosis. “People with optic neuritis lose their color perception out of proportion to their visual acuity, meaning they could have 20/20 vision, but their color vision may be wiped out,” said Dr. Frohman. “This is a nuance that the comprehensive ophthalmologist knows but often forgets.”
Unilateral optic nerve sheath meningioma (ONSM). ONSMs are rare benign tumors that originate from the meninges surrounding the optic nerve. Compression of the optic nerve will typically cause progressive vision loss over time, said Dr. Chen. “Although most patients undergoing radiation therapy for this condition tolerate it well and are able to preserve or even improve their vision, early treatment is better,” said Dr. Subramanian. Unfortunately, about 70% of these patients experience a delayed diagnosis for years.2 Some reasons for the delay include the following.
Missing subtle signs, symptoms. Patients with ONSM are usually in their 40s and 50s and are more often women than men, said Dr. Subramanian. If they have a normal-looking nerve with decreased vision, this can be misdiagnosed as optic neuritis, or if a patient has a swollen optic nerve with intact vision, they may be misdiagnosed as having papilledema from IIH, said Dr. Chen. In either case, the patient is usually referred to a neuro-ophthalmologist. But when diagnosis is delayed, said Dr. Subramanian, it’s more often because the patient has insidious vision loss and a pale optic nerve.
Wrong test. Ophthalmologists may send these patients for an MRI, but it is often the wrong kind: a brain MRI, said Dr. Subramanian. “It’s hard to see an optic nerve sheath meningioma on a brain MRI unless you know what you are looking for and you look carefully,” he said.
“But if you get an orbit MRI done properly, it should be a slam dunk.”
Order thin section views of the orbit MRI, with and without contrast, in all the right projections to ensure your imaging is adequate to capture what you need, added Dr. Frohman. “If you’ve identified a clear pattern from the history and exam, you’ll be able to communicate exactly what you are looking for to the radiologist.”
Posterior communicating artery aneurysm. All ophthalmologists know the signs and symptoms of a possible third cranial nerve palsy: diplopia, ptosis, and severe headache, said Dr. Subramanian. A third cranial nerve palsy may be the first sign of a posterior communicating (PCOM) artery aneurysm rupture, which comes with a 50% mortality rate, said Dr. Chen. Taking the actions below can be life-saving.
Don’t wait for pupil sign. “Comprehensive ophthalmologists are attuned to the fact that a pupil-involving third cranial nerve palsy requires emergency action,” said Dr. Frohman. “However, the abnormal pupil doesn’t invariably send a ‘blinking light’ telling you that you need to get this patient to the ER. For that reason, neuro-ophthalmologists no longer wait to see what the pupil does.”
Get an angiogram immediately. Today, any patient with a suspected third cranial nerve palsy needs immediate imaging with either a CT angiogram or an MR angiogram, said Dr. Chen. “The angiogram portion of these scans is key to ruling out the aneurysm.” It can detect almost all aneurysms large enough to cause third nerve palsy with minimal risk to the patient, added Dr. Frohman.
Share information. “When you order a scan, provide as much information as you can to the radiologist,” said Dr. Francis. “Rather than just saying ‘double vision,’ say ‘left third nerve palsy.’ There’s a lot of real estate in the brain, and this will help the radiologist know where to look.”
Imaging expertise. Not only do you have to get the right imaging sequences, but you also need them reviewed by a trained person who knows what they are looking for, said Dr. Subramanian. Finding these aneurysms relies upon the acumen of a neuro-radiologist, who can read the scans and do the postprocessing 3-D–reconstruction needed to best spot an aneurysm, said Dr. Frohman. Ophthalmologists should know if their radiologist has this expertise.
 |
|
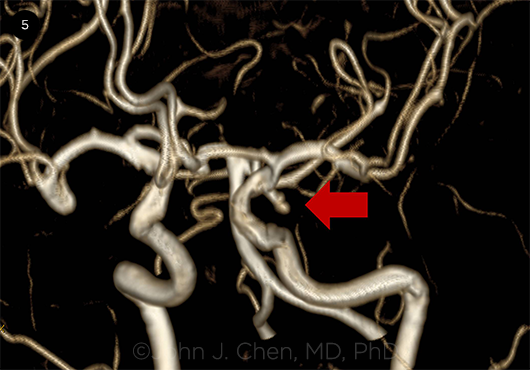
POSTERIOR COMMUNICATING ARTERY ANEURYSM. MR angiogram demonstrating a left posterior communicating artery aneurysm that presented with a left third nerve palsy (red arrow).
|
 |
|
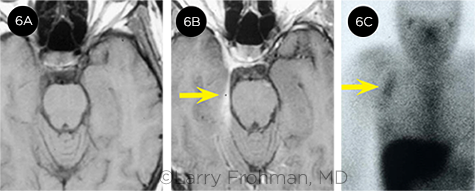
CORRECT IMAGING IS KEY. The patient is a 32-year-old woman who presented with headache and a sixth nerve palsy. (6A) She initially had a negative CT scan and nonenhanced MRI of the brain, and she was diagnosed by neurology with ophthalmoplegic migraine. Neuro-ophthalmology was consulted, thought that was an unlikely diagnosis, and asked for an enhanced MRI. (6B) This revealed enhancement of the dura in a pattern called a “dural tail” (arrow). This image suggested infiltrative or inflammatory disease of the meninges. (6C) This led to a gallium scan, which showed only an unrecognized axillary lymph node with significant uptake. A biopsy confirmed the diagnosis of sarcoidosis, and the symptoms resolved with corticosteroids.
|
A Shortage of Neuro-Ophthalmologists
Although referral is often the ideal solution for managing these conditions, the shortage of neuro-ophthalmologists is getting worse, said Dr. Chen. “A recent survey among practicing neuro-ophthalmologists found that one-third are over the age of 60.4 When they retire, there will be a huge void in a subspecialty that already has too few providers.” (For more on this topic, see this month’s Opinion.)
Shortfalls and mounting pressures. Today, the average wait time to see a neuro-ophthalmologist is about seven weeks, said Dr. Frohman. “That’s because there is one full-time neuro-ophthalmologist for every 1.7 million Americans—and this number should be closer to 1 per million.”
Adding insult to injury, most ophthalmologists are increasingly expected to follow productivity guidelines, said Dr. Frohman. In the past, they might have previously worked up the patient on their own. Now they’re passing patients on with less workup or none at all, he said. Of course, this leads to increased demand for neuro-ophthalmologic consultations.
Why so few subspecialists? “The current reimbursement model in the United States is heavily weighted toward procedures and patient volume, incentivizing speed and devaluing complex diagnostic reasoning skills,” said Dr. Biousse. “This makes our specialty less attractive to ophthalmology trainees.”
And a survey of graduating residents5 that probed the reasons for not picking this subspecialty showed that respondents were concerned that neuro-ophthalmology did not offer the opportunity to do surgery. This is a bit of a misconception, said Dr. Chen, because 50% of neuro-ophthalmologists do perform surgical procedures—the types of surgery are dependent upon fellowship and comfort levels.
Dr. Francis agreed, “Many of us in neuro-ophthalmology continue to do surgeries that are typically done by comprehensive ophthalmologists, including cataract surgery. Others do strabismus surgery, temporal artery biopsies, optic nerve sheath fenestrations, or orbital surgery, which all fit nicely with the types of patients we see in neuro-ophthalmology.”
Other barriers to choosing neuro-ophthalmology? Salaries and the perception that this is a purely academic field. “In reality,” said Dr. Chen, “one-third of neuro-ophthalmologists are in nonacademic private practice settings. And, there is a wide range of salaries within neuro-ophthalmology, higher if surgery is a part of your practice.”
Perks aplenty. Of course, finding ways to adequately compensate neuro-ophthalmologists might lure more into the field. But the job brings plenty of perks right now, said Dr. Francis. “It’s a perfect fit for me in terms of the pace and ability to spend time with patients, the variety of complex diagnostic challenges, the collaborative coordination of care with other specialists, and the medical and surgical interventions that I am able to offer,” she said. “It’s very satisfying. You won’t find anyone who regrets going into this field.”
Dr. Chen agreed. “Both neuro-ophthalmology practice and research are very stimulating. Every patient and every day is different—and sometimes we make life-saving diagnoses.”
Lightening the load. A few changes to consults and other processes may help patients get the proper care more quickly.
Build confidence in ophthalmologists. One way to relieve backlogs in neuro-ophthalmology is to help other ophthalmologists gain more confidence in handling straightforward cases, said Dr. Subramanian. “We should make ourselves available for quick phone consultations to help other ophthalmologists know that a little extra time with patients may be all they need for diagnosis and management,” he said.
Capitalize on telemedicine. Telemedicine has a clear role to play and can be really useful, said Dr. Subramanian. That’s particularly true when records are available and the patient has already had a lot of the testing done—and the expertise of a neuro-ophthalmologist is needed for an evaluation.
“We’re not there yet,” said Dr. Frohman, “but in the future, the ophthalmologist may call us while the patient is still present and ask, ‘What tests should I get?’ Then we’ll recommend test parameters and schedule a telemedicine consult for soon after the patient has completed the requisite testing.”
Artificial intelligence (AI). In neuro-ophthalmology, said Dr. Subramanian, AI might help, one day, with pattern recognition on visual field tests and reduce the number of patients who need to undergo further testing. “It will never replace our problem-solving in history gathering and differential diagnosis,” said Dr. Chen, “but it could certainly aid in the interpretation of photographs, which can help with triaging.” In collaboration with a Singapore group, Dr. Biousse, Dr. Chen, and colleagues were able to train AI to diagnose papilledema with 90% accuracy—as good as a practicing neuro-ophthalmologist.6
___________________________
1 Stunkel L et al. J Neuroophthalmol. 2019. Published online Oct. 11, 2019.
2 Stunkel L et al. Curr Opin Neurol. 2019;32(1):62-67.
3 Stunkel L et al. JAMA Ophthalmol. 2018;136(1):76-81.
4 DeBusk AA et al. J Neuroophthalmol. In press.
5 Solomon AM et al. Factors affecting ophthalmology resident choice to pursue neuro-ophthalmology fellowship training. Accepted by J Neuroophthalmol.
6 Milea et al. New Engl J Med. 2020;382(18):1687-1695.
|
(Click to enlarge)
|
 |
IRIS Registry Snapshot: Cataract Surgery and NAION
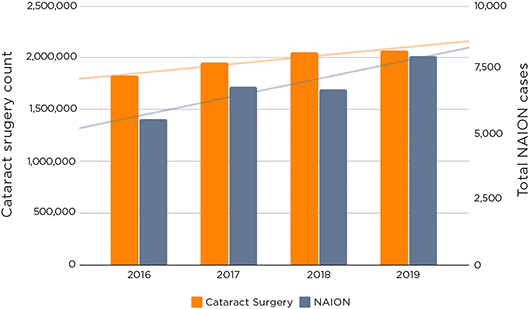
About 70 years ago, cataract surgery was identified as a possible contributor to nonarteritic anterior ischemic optic neuropathy (NAION), but to this day debate about the strength of the connection continues.
Using electronic health record data from the IRIS Registry, Verana Health assessed the number of new NAION cases and number of cataract surgeries over a four-year period. To ensure that eyes were not included twice, only eyes with right/left eye attribution were included in this analysis.
The increasing number of new NAION cases over time may be correlated with the increased yearly prevalence of NAION risk factors such as diabetes, hypertension, heart disease, and tobacco use in the general population (bar graph below).
Verana also used the registry to assess the number of new NAION cases within three years of cataract surgery (table below). This analysis limited the count of NAION cases to practices that submitted a CPT code for cataract surgery at least three years prior to the NAION date. With the number of annual cataract surgeries increasing over the study period, there was a correlating increase in the number of NAION cases.
Cases Within Three Years of Cataract Surgery
| Year |
2016 |
2017 |
2018 |
2019 |
| NAION Count |
261 |
249 |
298 |
321 |
| Note: The Academy has partnered with Verana Health to curate and analyze IRIS Registry data. |
Economic Impacts of Incorrect Diagnoses
“It’s hard to put a dollar amount on the economic loss of a patient who’s had a fatal aneurysm that ruptured but could have been detected, or of a patient with bilateral blindness from giant cell arteritis, where at least one eye could have been saved,” said Dr. Chen.
But some data do exist to give a sense of the losses from incorrect or delayed diagnoses. “Twenty-three percent of MRI scans show incidentally abnormal findings, which often lead to unnecessary follow-up tests,” said Dr. Subramanian. “For example, a majority of patients given a diagnosis of optic neuritis outside of a neuro-ophthalmologist’s office probably didn’t need an MRI scan. This is a $3,000-5,000 expense, multiplied by tens of thousands of patients.”
Dr. Frohman echoed this concern. “Years ago, another neuro-ophthalmologist and I put together a series of double vision cases that were sent to us, in which we ultimately diagnosed keratoconus as the cause of the double vision. We found that a huge amount of money was spent on unnecessary testing before we saw the patients.”
Dr. Frohman also pointed to a retrospective review of records for 588 patients referred for a neuro-ophthalmologic evaluation.1 The authors wanted to compare the frequency and cost of unnecessary diagnostic testing ordered by “gatekeeper physicians” with neuro-ophthalmologist consultations. Between 16% and 26% of patients with optic neuropathy, diplopia, or ptosis were subjected to overtesting. This resulted in $57,900 of excessive costs, a 724% overcharge.
___________________________
1 Dillon EC et al. Ophthalmology. 1994;101(9):1627-1630.
|
Meet the Experts
Valérie Biousse, MD Professor of ophthalmology and neurology at the Emory University School of Medicine in Atlanta. She is also neuro-ophthalmology fellowship director and president of the North American Neuro-Ophthalmology Society (NANOS). Relevant financial disclosures: None.
John J. Chen, MD, PhD Associate professor of ophthalmology and neurology at the Mayo Clinic College of Medicine in Rochester, Minn. He is also associate residency program director and neuro-ophthalmology fellowship director at the Mayo Clinic, and cochair of the Upper Midwest Neuro-Ophthalmology Group. Relevant financial disclosures: None.
Courtney E. Francis, MD Associate professor of ophthalmology and neurological surgery, and director of neuro-ophthalmology and medical student education in ophthalmology at University of Washington School of Medicine in Seattle. Relevant financial disclosures: None.
Larry P. Frohman, MD Professor of ophthalmology and neurology and vice chair of ophthalmology, at the Rutgers–New Jersey Medical School in Newark. He is also executive vice president of NANOS. Relevant financial disclosures: None.
Prem S. Subramanian, MD, PhD Professor of ophthalmology, neurology, and neurosurgery, division head of neuro-ophthalmology and vice chair for academic affairs, University of Colorado School of Medicine in Aurora, Colo. He also is president-elect of NANOS. Relevant financial disclosures: None.
Full Financial Disclosures
Dr. Biousse GenSight Biologics: C; Neurophoenix: C.
Dr. Chen None.
Dr. Francis None.
Dr. Frohman None.
Dr. Subramanian GenSight Biologics: C,S; Horizon Therapeutics: C; Invex Therapeutics: C; Quark Pharmaceuticals: S; Santhera Pharmaceuticals: S.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|