Download PDF
To date, clinicians have had trouble distinguishing polypoidal choroidal vasculopathy (PCV) from age-related macular degeneration (AMD). But researchers at the University of Hawaii have found that OCT B-scans can detect a characteristic structural feature of PCV.1
Tell-tale sign. Gregg T. Kokame MD, MMM, at the University of Hawaii in Honolulu, and his coauthors conducted a retrospective study of case records on 112 eyes with AMD (106 patients). Sixty-nine of the eyes had been diagnosed with PCV using indocyanine green angiography (ICGA). Comparison to the patients’ pre- and post-treatment OCT B-scans showed that there was a characteristic sign of PCV: an inverted U-shaped elevation of the retinal pigment epithelium (RPE) that was usually located between the RPE and Bruch membrane. The location of this feature on OCT B-scans correlated to the sites of polypoidal lesions on ICGA.
Critical timing. However, the U-shaped elevations were visible primarily before beginning treatment, Dr. Kokame said. “We showed that if you only looked at the after-treatment OCT B-scan, only a quarter of the eyes with PCV were diagnosed. But if you looked at the before-treatment B-scan, the correct diagnosis was reached in 56% of the known PCV cases,” he said.
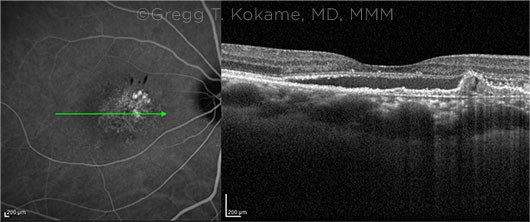 |
CRITICAL CLUE. PCV is characterized by subretinal neovascularization between the RPE and Bruch membrane, dilated polypoidal lesions, and a branching vascular network. In these images, an inverted U-shape elevation of the RPE on the OCT B-scan (right) corresponds to the polypoidal lesion on the ICG angiogram (left). The green arrow on the angiogram identifies the location of the image on the OCT B-scan.
|
A cause of anti-VEGF resistance? PCV is a subtype of exudative AMD that predominates among Asian populations, although it occurs, to a lesser degree, in other ethnic groups. Awareness of the clinical importance of this subtype has risen in the last decade as researchers sought to explain why some AMD eyes were resistant to anti-VEGF therapy. It now is thought that unrecognized PCV might be a factor, Dr. Kokame said.
Need to tailor treatment. According to a recent report from the EVEREST II clinical trial, PCV responds better to combined photodynamic therapy plus ranibizumab than it does to ranibizumab alone.2
Consequently, it is important for ophthalmologists who treat AMD to be able to identify possible PCV cases early, even though the traditional technology for doing so often is not available in many clinical settings, Dr. Kokame said. “Usually to get the best diagnosis of PCV requires specialized equipment, such as ICGA with the scanning laser ophthalmoscope, plus the ability to read an ICG angiogram, which most practitioners don’t have access to,” he said. “But almost every practice in ophthalmology has access to OCT, and the B-scan is one thing that just about every ophthalmologist knows how to look at.”
As ICGA is often not available, not ordered, or not comfortably read, OCT could help practices identify many patients who otherwise might not be considered for combination therapy, Dr. Kokame said. He added, “We want all ophthalmologists treating exudative macular degeneration to understand that polypoidal choroidal vasculopathy is the most important subtype of AMD.”
Specifically, PCV “acts differently from typical exudative macular degeneration, with higher resistance to current medications; it might be susceptible to alternative therapy; and it is predictive of response to different medications,” Dr. Kokame said. “We want them to learn to diagnose PCV in the majority of cases with the equipment that they do have available, the OCT B-scan.”
—Linda Roach
___________________________
1 Kokame GT et al. Ophthalmol Retina. Published online May 19, 2021.
2 Lim TH et al. JAMA Ophthalmol. 2020;138(9):935-942.
___________________________
Relevant financial disclosures—Dr. Kokame: None.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Ensign Co-inventor on patent applications describing the suture technology; Robert H. Smith Family Foundation: S; Research to Prevent Blindness: S; National Institutes of Health: S; National Science Foundation: S.
Dr. Hedges None.
Dr. Kokame Allergan: C; Bayer: C,L; Bausch + Lomb: C,L; Carl Zeiss: C,L; Genentech: C,S; Novartis: S; Regeneron: C,L,S; RegenxBio: S; Salutaris: S.
Dr. Reddy Alimera: C; Clearside: C; Eyepoint: C; Heidelberg: C.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|
More from this month’s News in Review