Download PDF
With gene therapy plus visual stimulation, scientists have coaxed retinal ganglion cells (RGCs) to grow long-distance, target-specific axons that restored partial functional vision in mice1—an unprecedented feat that the researchers hope eventually might pave the way to fruitful clinical studies.
Methods and results in mice. The scientists injected a recombinant adeno-associated virus, modified to promote production of mTOR, a peptide that regulates RGC growth, into the eyes of mice with crush injuries to the optic nerve.
The peptide molecules alone stimulated a small amount of axon growth, the investigators found. But when the treated mice also spent 21 days exposed to high-contrast vertical lines on TV monitors, the axons grew past the optic chiasm and found their correct subcortical targets. In monocular tests of tasks related to visual function, these mice regained some ability to track a moving grating and to respond to a simulated predator overhead.1
“We were absolutely surprised but delighted that the retinal ganglion cells in adult eyes are capable of regenerating long distances back into the brain given the appropriate stimuli, and that without any additional help they are able to renavigate these tortuous paths back to their correct location in the brain, form synapses, and reestablish some degree of visual perception behavior,” said coauthor Andrew D. Huberman, PhD, associate professor of neurobiology at Stanford University.
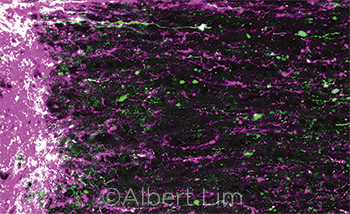 |
AXON ACTION. Retinal axons regenerating in the optic nerve (green and magenta), after damage and treatment with gene therapy and visual stimulation.
|
Hopes tempered by “unknowns.” “This is really encouraging for future clinical prospects, because it suggests that if we can get human neurons to regenerate, they’re going to know where to go, as opposed to wiring up willy-nilly,” Dr. Huberman said.
Over the next several years, the many unknowns about this apparently synergistic combination of gene therapy and RGC “training” must be addressed before the approach can proceed to clinical trials, Dr. Huberman said. “We know now that regenerating neurons is not entirely science fiction, but there are important steps that need to take place first,” he said. Among the outstanding issues:
- Axons did not regenerate completely in all of the eyes’ RGCs. Perhaps this could be improved by adding recombinant genes to block molecules that otherwise inhibit RGC growth, Dr. Huberman said.
- The study involved only 2 of the approximately 30 types of RGCs in mammalian eyes, and the remaining cell types must be studied, he said.
- What types of visual stimuli should be used, first in mice, then in humans? “You need the RGCs to be firing a lot, and for that they need high-contrast and high-frequency stimulation. But we don’t know what patterns and forms are best,” he said.
Seeking to learn more. Since publication of the results, Dr. Huberman said, glaucoma patients have swamped him with requests for guidance about using high-contrast, high-frequency visual stimulation to possibly boost their RGC health—and he plans to begin studies of this soon.
“This is a completely noninvasive approach; and while it’s true that in our study the visual stimulation by itself was not sufficient to get the axons to grow all the way back to the brain, we were using pretty severe, complete lesions of the optic nerve,” he said. “So we are embarking on some clinical studies, setting up virtual reality environments to stimulate RGCs in patients, as a way to try and preserve their health through electrical activity and presumably maintenance or regeneration of fibers.”
He said he initially envisions monitoring effectiveness with visual field testing. But he is working with the Glaucoma Research Foundation to find ways to track the outcomes by developing advanced imaging technology to count RGCs in the living eye.
—Linda Roach
___________________________
1 Lim JA et al. Nat Neurosci. Published online July 11, 2016.
___________________________
Relevant financial disclosures—Dr. Huberman: None.
For full disclosures and disclosure key, see below.

More from this month’s News in Review