Download PDF
Unplanned return to the OR following trabeculectomy surgery was more common than expected, according to researchers at the Wilmer Eye Institute in Baltimore.1 In a retrospective case-control study, the researchers found that nearly one in 10 treated eyes required an unplanned reoperation within 180 days, while one in five underwent reoperation at any time up to three years.
This higher than expected rate of return “highlights the importance of reporting such data so that clinicians and patients have a better understanding of the risks and postoperative course after trabeculectomy,” said coauthor Michael V. Boland, MD, PhD.
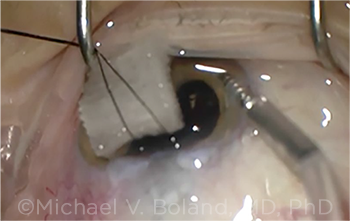 |
BLEB NEEDLING. Most reoperations following unplanned return to the OR required bleb needling.
|
Two calculations. The findings are based on clinical data from 881 eyes that had undergone trabeculectomy from January 2014 to December 2016. Each eye was randomly matched to a control patient who had surgery near the same time. Reoperation and control eyes did not differ with regard to a number of factors, including mean follow-up, age at surgery, type of glaucoma, history of either prior incisional or glaucoma surgery, mean baseline intraocular pressure (IOP), and mean number of pre-op glaucoma eyedrops. The reoperation rate was 9.5% (84 eyes) up to 180 days post-trabeculectomy and 23.3% (205 eyes) up to a mean of three years.
A second analysis excluded bleb needling to account for significant differences in surgeons’ preferences for where to perform this procedure. One surgeon performed a large number of these procedures in the OR, while others performed bleb needling in the clinic. Given that the latter group was not identified by billing records, this biased the results. The return to OR rate in this second analysis fell to 6.5% (57 eyes) at 180 days and 13.6% (120 eyes) at any time.
Reasons for reoperation. In the earlier post-op period, the most common reasons for return to the OR were bleb leak, choroidal effusion, and persistently elevated IOP despite medical therapy. These cases typically resulted in bleb revision, choroidal drainage, and intraoperative bleb needling procedures.
At any time, the most common reason for return to the OR by far was persistently elevated IOP despite medical therapy. In these cases, reoperation typically involved bleb needling or a new glaucoma surgery, such as a tube shunt. Factors that were not associated with return to the OR included maximum IOP prior to trabeculectomy, preoperative use of oral glaucoma medications, and combined cataract-trabeculectomy surgery.
Similar outcomes. Although those returning to the OR used more topical medications and underwent more surgeries than controls (sometimes multiple surgeries), outcomes between the two groups were similar. For instance, mean IOP, proportion of eyes meeting target IOP, and change in visual acuity following the original trabeculectomy were comparable.
Still, a return to the OR is far from optimal, Dr. Boland said. “Accordingly, these analyses of reoperation after trabeculectomy are important in helping to set appropriate expectations for patients and providers alike.”
—Miriam Karmel
___________________________
1 Cardakli N et al. Am J Ophthalmol. 2020;219:132-140.
___________________________
Relevant financial disclosures—Dr. Boland: Carl Zeiss Meditec: C.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Boland Carl Zeiss Meditec: C.
Dr. Ciolino TherOptix: C,O,P.
Dr. Delcourt Allergan: C; Bausch + Lomb: C; Laboratoires Théa: C; Novartis: C.
Dr. Subramanian None.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|
More from this month’s News in Review