Download PDF
Two simple changes in office routines can significantly reduce the incidence of keratopathy after complex retinal detachment (RD) surgery with pars plana vitrectomy and silicone oil tamponade, a study at the University of California, San Francisco (UCSF) has found.1
The changes involved:
- keeping a registry of patients who received silicone oil, and
- calling to remind them to return for follow-up appointments.
After UCSF vitreoretinal surgeons adopted these procedures, the proportion of patients with complex RD who developed keratopathy fell from 33.3% to 12.8%. “We were surprised that it was so simple to achieve such a difference,” said coauthor Jay M. Stewart, MD, at UCSF and Zuckerberg San Francisco General Hospital.
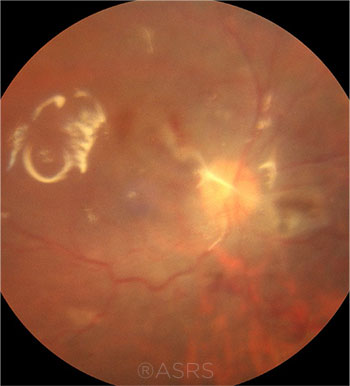 |
TAMPONADE. Silicone oil was used in this patient with an RD (note retinal surface reflex). A lengthy period of tamponade has been shown to be the greatest risk factor for silicone oil emulsification, which can lead to keratopathy, glaucoma, and cataracts. This image was originally published in the ASRS Retina Image Bank. Ronald C. Gentile, MD. PVR Retinal Detachment With Subretinal Bands Slide 2. Retina Image Bank. 2012; Image Number 1744. © The American Society of Retina Specialists.
|
Instigation. Dr. Stewart said the vitreoretinal division in the hospital’s ophthalmology department implemented this registry and reminder system in 2014, after physicians and staff noticed that patients were experiencing complications in the months after complex RD surgery.
These adverse outcomes appeared to be related to a lack of follow-up and the duration of the silicone oil in their eyes, Dr. Stewart said. “We encountered complications that could have been avoided if the oil had been removed sooner.”
Improved outcomes. After early indications that the reminders were working, the practice did a retrospective records review to compare outcomes in 48 eyes treated before the system was in place (control group), and 39 eyes treated afterward. The review showed statistically significant reductions not only in keratopathy cases but also in two other key areas:
- The number of patients lost tofollow-up was 23 (47.9%) in the control group, versus six (15.4%) in the treatment group (p = 0.0015).
- The mean duration before silicone oil removal was 79.6 ± 91.7 weeks (mean ± standard deviation) in the control group and 36.3 ± 31.5 weeks in the treatment group (p = 0.015).
With regard to other outcomes, intraocular pressure measurements did not vary significantly between the two groups. Finally, cataract formation was not analyzed as an outcome measure in this study.
Standard practice. Dr. Stewart said the registry and reminder system is now standard practice in the hospital’s clinic. “If patients don’t show up, then they start getting chased down by the staff” until they do return, he said. “Establishing a line of communication directly between the clinic and the patient gets it back on their radar. It reinforces to them that we consider the appointment to be important—and that there’s value in keeping it.”
—Linda Roach
___________________________
1 Ma D et al. Ophthalmol Retina. 2019;3(7):543-547.
___________________________
Relevant financial disclosures—Dr. Stewart: None.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Annabi NIH: S; UCLA: S; U.S. Department of Defense: S.
Dr. Baker Alcon: S; Genentech: S; Novartis: S; Regeneron: S.
Dr. Solomon Alcon: C,S; AqueSys: C; ClarVista Medical: C; Glaukos: C; Icon Bioscience: C; Integrity Digital Solutions: C; Mati Therapeutics: C; Octane Visionary VC Fund: C; OcuHub: C; Omeros: C; PogoTec: C; PRN: C; Versant Ventures: C.
Dr. Stewart Carl Zeiss Meditec: C; Genentech: C; Merck: C.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|
More from this month’s News in Review