By Jennifer S. Griffin, MS, Contributing Writer, interviewing Amar Agarwal, MD, Sanjay Asrani, MD, and Alan Crandall, MD
Download PDF
Angle closure compounded by peripheral anterior synechiae (PAS) is one of the biggest challenges we face as glaucoma surgeons, said Sanjay Asrani, MD. PAS can irreversibly impair flow through the trabecular meshwork,1 resulting in angle-closure glaucoma that persists despite first-line treatment. If PAS have been present for longer than six months, the chances of reestablishing function of the trabecular meshwork are very low, even if you remove the adhesions and anatomically restore the angle, said Dr. Asrani, who is at Duke University in Durham, North Carolina.
After treatment, recurrence of adhesions is a constant concern, said Alan Crandall, MD. In chronic angle-closure glaucoma, the iris often is atrophic, and conventional measures to resolve PAS deteriorate during long-term follow-up, he said. Dr. Crandall is at the Moran Eye Center in Salt Lake City.
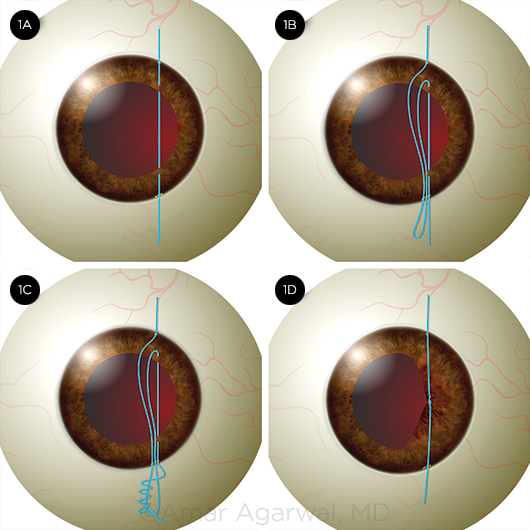 |
|
SFT PUPILLOPLASTY. (1A) A needle is passed through the proximal and distal portions of the iris tissue. (1B) The proximal and distal portions of the iris are approximated, and a loop of suture is withdrawn. (1C) The suture end is passed through the loop four times. (1D) When the suture ends are pulled, the loop slides inside the iris tissue, yielding a stable knot.
|
A New Approach
In 2017, Amar Agarwal, MD, tried something different to treat angle-closure glaucoma in phakic patients. He performed a new technique—single-pass four-throw pupilloplasty (SFT)—in combination with lens extraction.2 He found that the procedure opened the angle, released associated PAS, and secured the iris centrally to help prevent recurrence of the PAS. Dr. Agarwal is with Dr. Agarwal’s Eye Hospital and Eye Research Centre in India.
Other techniques, such as a Siepser slipknot, can also be used for pupilloplasty. However, “In the evolution of anything we do surgically, we’re trying to make procedures as elegant, as safe, and as cost-effective as we can,” said Dr. Crandall. “And this is the case with SFT because you’re placing one suture multiple times. It’s much less time-consuming than a Siepser knot and just as effective.”
No matter what treatment is used, angle closure inevitably causes damage to the trabecular meshwork, said Dr. Asrani. But SFT and medical therapy, if implemented in the early stage of the disease, might be enough to restore aqueous flow and stabilize IOP, obviating the need for more invasive interventions such as trabeculectomy or tube surgery, he said. “When I look at this technique, I think of the patients I could have helped with it.”
Traditional Treatments
First-line therapies for angle-closure glaucoma include pilocarpine eyedrops, laser surgery, goniosynechiolysis, and cataract extraction, depending on the severity and underlying cause. If initial treatment fails, surgeons usually turn to trabeculectomy or implantation of a drainage device, said Dr. Asrani.
Laser surgery. Laser peripheral iridotomy is the mainstay for pupillary-block angle closure. However, in angle closure caused by plateau iris, iridotomy will not resolve the narrow angle or prevent PAS. In these cases, argon laser peripheral iridoplasty and pilocarpine eyedrops are initial options, but over time, the iris usually migrates back to the periphery, and PAS recur, said Dr. Asrani.
Synechiolysis and lens removal. Dr. Asrani noted that even goniosynechiolysis coupled with lens removal is a temporary fix because the inflamed iris remains close to the trabecular meshwork and tends to readhere. “In contrast,” Dr. Asrani said, “SFT pupilloplasty along with synechiolysis prevents recurrence of PAS by keeping the pupil taut. For patients who have angle closure with a chronically dilated pupil, SFT also will improve the optics by reducing glare.”
Techniques and Outcomes
Dr. Agarwal performs SFT pupilloplasty under peribulbar anesthesia, with supplemental anesthesia given as needed.2 He prefers maintaining the anterior chamber with fluid, rather than viscoelastic, because “fluid will wash away hyphema, which may occur when PAS are broken.” He also recommends using an endoilluminator for good visualization, especially if the cornea is hazy.
Technique. An end-opening forceps is used to grasp the iris and pull it toward the center of the pupil at 60-degree intervals around the pupillary margin. Dr. Crandall pointed out, “This ‘pull and release’ technique partially detaches the PAS and informs the surgeon about the extent of the adhesions and the amount of iris tissue available for reconstruction.”
With the proximal iris held with forceps, a straight needle with a 10-0 or 9-0 nonabsorbable polypropylene suture is inserted. From the other end, a 26- or 30-gauge needle is passed through a clear-corneal incision into the distal iris. The straight needle is docked into the lumen of the 26- or 30-gauge needle, and the two are withdrawn together through the distal incision. A loop is created at the suture exit side with a Sinskey hook, and four throws of the distal end of the suture are made through this loop. The ends of the suture are pulled apart to yield a self-locking, helical knot that lies flat against the iris. The suture ends are trimmed with a microscissors, leaving 1-mm ends. (See video below.)
Dr. Agarwal explained his rule of thumb for SFT knot placement. “If PAS are observed around more than 270 degrees of the pupillary margin, carry out six-point traction (i.e., three SFT knots); if the PAS constitute less than 270 degrees, you only need four-point traction to sufficiently constrict the pupil.”
Recent findings. In a 2018 study coauthored by Dr. Agarwal, SFT pupilloplasty was performed following cataract surgery in five patients with angle-closure glaucoma and PAS.2 By six to eight months postoperatively, all patients had fewer PAS, an open angle, lower IOP, and better visual acuity. “As an adjunct to PAS lysis and to prevent further synechiae development, SFT makes sense theoretically, but it is early in its development,” said Dr. Crandall.
Benefits of SFT
Anatomy. “In SFT pupilloplasty, you are not introducing an artificial drainage pathway, as in trabeculectomy or valve placement,” said Dr. Agarwal. “Instead, you are enabling function of the existing trabecular meshwork. You are restoring, rather than changing, the anatomy.” Dr. Crandall agreed, “It makes physiologic sense as treatment for chronic angle-closure glaucoma.”
A simpler knot. Dr. Agarwal said that surgeons who prefer the Siepser slipknot or cerclage can adapt those pupilloplasty maneuvers to treat angle-closure glaucoma. However, Dr. Crandall noted, “SFT is technically easier, less time-consuming, and as effective as other pupilloplasty techniques.”
Safety. Dr. Agarwal considers SFT to be safer than multiple-pass pupilloplasty techniques. “When you go for a second or third pass, you are manipulating the anterior chamber, and you can damage the iris and cornea,” he said. Additionally, in SFT pupilloplasty, the knot is self-retaining and is not tied, thereby reducing bulk in the anterior chamber.3
Considerations
Despite its advantages, SFT pupilloplasty combined with lens removal is not a one-size-fits-all solution for angle-closure disease.
Phakic status. Narang et al. noted that SFT cannot be performed in phakic eyes and that lensectomy should be done in the same surgical session,3 regardless of whether a visually significant cataract is present. However, this apparent drawback may be counterbalanced by the reported benefits of lens removal in angle-closure glaucoma. Results of a randomized controlled trial demonstrated that clear lens extraction is more efficacious and more cost-effective than laser peripheral iridotomy plus topical medical treatment in patients with primary angle closure and high IOP with or without glaucoma.4 Nevertheless, some surgeons have questioned the validity of clear lens extraction for angle closure, given the surgical risks and loss of accommodation with lens removal.5,6
Inflammation. Because SFT pupilloplasty is an intraocular procedure, inflammation is a concern. “If the patient has fixed pupillary dilation with chronic angle closure in uveitis, SFT may chronically inflame the iris. Additionally, the inflamed eye structures could remain in apposition, so PAS might not be prevented,” Dr. Asrani cautioned. “However, benefits of preventing PAS and reducing glare using SFT may have to be balanced with the risk of persisting iritis in such cases.”
Tissue tears. “In general, the surgeon should be careful to avoid overtightening the helical knot, which could tear the iris tissue,”3 said Dr. Agarwal. He added that extreme care should be exercised when performing SFT in eyes with secondary angle-closure glaucoma involving atrophic patches of the iris, such as in Urrets-Zavalia syndrome (UZS).2 Nevertheless, Dr. Agarwal said that SFT pupilloplasty—performed carefully—does open the angles well in cases of UZS.
Fundus visualization. Dr. Agarwal and his colleagues reported that patients treated with SFT pupilloplasty still can undergo mydriasis, although the extent of pupillary dilation in SFT-treated eyes is less than that in untreated eyes.7 Dr. Asrani pointed out that decreasing the pupillary opening, by means of SFT pupilloplasty, can limit the examination and treatment of retinal conditions. However, he said, “If the patient needs retinal treatment, the retinal surgeon can snip the SFT suture and reopen the pupil.” Dr. Agarwal added that an Nd:YAG laser also could be used to undo the pupilloplasty.
Cosmesis. “In terms of aesthetic results,” Dr. Crandall said, “pupillary cerclage is probably better than SFT. However, cerclage is technically challenging and time-consuming. And although cerclage may appear cosmetically better, it is not functionally better, he said. Moreover, Dr. Agarwal pointed out, “Cerclage is especially difficult to perform in the setting of PAS.”
Too late? “If angle-closure glaucoma goes untreated,” said Dr. Agarwal, “fibrosis can occur,” and SFT pupilloplasty would not be sufficient to normalize aqueous outflow. He noted, “Such patients would need additional medical treatment or even a shunt procedure or trabeculectomy.”
1 Hamanaka T et al. Invest Ophthalmol Vis Sci. 2011;52(12):8849-8861.
2 Narang P et al. Indian J Ophthalmol. 2018;66(1):120-124.
3 Narang P, Agarwal A. Eur J Ophthalmol. 2017;27(4):506-508.
4 Azuara-Blanco A et al. Lancet. 2016;388(10052):1389-1397.
5 Díaz-Alemán VT et al. Arch Soc Esp Oftalmol. 2017;92(8):401-402.
6 Traverso CE. Lancet. 2016;388(10052):1352-1354.
7 Kumar DA et al. J Cataract Refract Surg. 2017;43(10):1307-1312.
___________________________
Dr. Agarwal is chair and managing director of Dr. Agarwal’s Eye Hospital and Eye Research Centre, Chennai, India. Financial disclosures: Bausch + Lomb: S; Jaypee: P; Mastel: P; Sanoculus: C; Slack: P; Staar: C; Thieme: P.
Dr. Asrani is professor of ophthalmology at Duke University in Durham, N.C. Financial disclosures: Aerie: C; Bausch + Lomb: C; Camras Vision: C; Noveome Biotherapeutics: C; Regenexbio: C.
Dr. Crandall is professor and senior vice-chair of ophthalmology and visual sciences and director of glaucoma and cataract at the Moran Eye Center, University of Utah, Salt Lake City. Financial disclosures: Alcon: C,L; AqueSys: C; ASICO: C; Excel-Lens: C; Glaucoma Research Foundation: C; Glaukos: C; Iantech: C; iSportGames: C; Ivantis: C; iVeena: C; Johnson & Johnson: C; Mastel: C; New World Medical: C; Omeros: C.
See the disclosure key at www.aao.org/eyenet/disclosures.