By Megan A. Rowlands, MD, MPH, Bing Chiu, MD, and Joel S. Schuman, MD
Edited By: Ingrid U. Scott, MD, MPH, and Sharon Fekrat, MD
Download PDF
Terson syndrome (TS) is the presence of any intraocular hemorrhage, including vitreous, subhyaloid, intraretinal, or subretinal bleeding, in patients with intracranial hemorrhage or traumatic brain injury. The term originally referred only to vitreous hemorrhage in the setting of a subarachnoid hemorrhage (SAH), but the definition has since been expanded.
Incidence. In part due to this change in diagnostic criteria, estimates of the incidence of TS vary. In a systematic review of SAH patients, McCarron et al. reported a 13% incidence of TS among patients evaluated prospectively.1 Czorlich et al. reported an incidence of 19% in patients with SAH, 9% in those with intracerebral hemorrhages, and 3% in patients with traumatic brain injury.2
The incidence of TS is significantly higher in patients with greater impairment in consciousness (as indicated by a low Glasgow Coma Scale) or more severe subarachnoid hemorrhage (based on either a high Hunt and Hess grade or a high Fisher grade).2 TS is more likely to occur in patients who have had prolonged episodes of unconsciousness or elevated intracranial pressure3; and some, but not all, studies have observed a stronger association with anteriorly located aneurysms.4 Although TS usually develops within hours of the neurological event, it can occur days or weeks later.2
Delayed diagnoses. Despite the relatively high incidence of TS in patients with SAH, the syndrome remains underdiagnosed. One reason may be that the patients who are most likely to have TS are also more likely to be neurologically impaired and, therefore, limited in their ability to verbalize their ocular complaints.2 In addition, given the neurological acuity and severity of these patients’ conditions, an ocular examination may not be performed until other, more emergent, interventions have been undertaken.
Thus, referral of patients with TS to ophthalmology is often delayed. In a review of TS patients who later underwent vitrectomy, Gnanaraj et al. reported an average of 5.2 months between the time that a TS patient first complained of ocular symptoms and when an ophthalmology consultation occurred.5 This delay in diagnosis can lead to permanent visual impairment and impede neurorehabilitation efforts.
 |
|
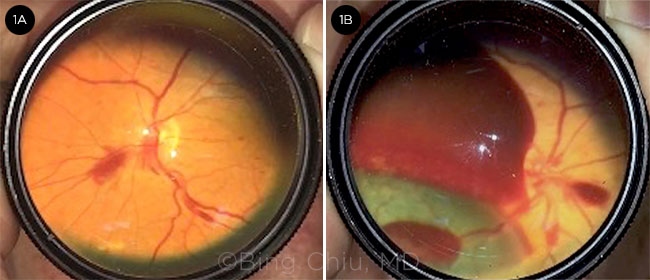
FUNDUS FINDINGS. Fundi of a 48-year-old woman who had an anterior communicating cerebral artery aneurysm, diffuse subarachnoid hemorrhage, and left inferior frontal cerebral hematoma. (1A) Right eye, showing intraretinal hemorrhages nasal and superior to the disc. (1B) Left eye, showing a large area of preretinal hemorrhage overlying the macula, subretinal hemorrhage along the superior arcade, and intraretinal hemorrhage nasal to the disc. (Images, taken with a 20 D lens and iPhone 7 camera with flash, are inverted vertically and horizontally.)
|
Pathophysiology
The pathophysiology of TS is debatable, and multiple mechanisms have been proposed. The leading theory suggests that an increase in intracranial pressure causes a rapid efflux of cerebrospinal fluid or hemorrhage via the optic nerve sheath into the orbit. This, in turn, compresses the central retinal vein, obstructing venous outflow and subsequently rupturing the smaller retinal venules.6
Another theory proposes that an acute elevation in intracranial pressure can increase orbital venous pressure, leading to backflow of blood through the retinal veins.7 A third theory suggests that the intraocular hemorrhages may be the result of direct extension of blood from the subarachnoid space itself via the optic nerve sheath.8
Although the exact etiology remains unknown, an association with intracranial pressure has been supported by Czorlich et al., who found that patients with TS were more likely to have had periods of elevated intracranial pressure greater than 25 mm Hg.3
 |
|
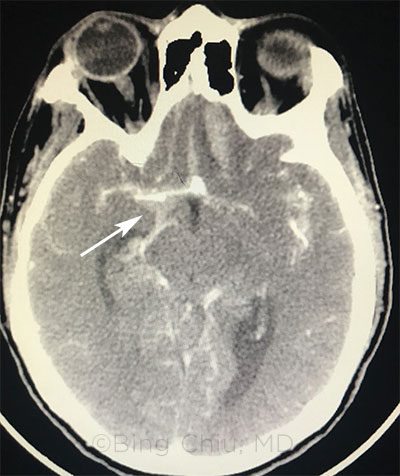
ASSESS FOR TS. CT of a 48-year-old woman admitted to our emergency department shows a 6 × 5 mm anterior communicating cerebral artery aneurysm, diffuse subarachnoid hemorrhage, and left inferior frontal cerebral hematoma. She later underwent a left craniotomy for clipping of the anterior communicating aneurysm.
|
Symptoms
The symptoms reported by TS patients can vary widely, depending on the degree and location of the hemorrhage as well as the individual’s neurological status. Many patients, especially those who are most neurologically compromised, may not be able to perceive or communicate ocular complaints. In such cases, prompt diagnosis depends on the primary physician’s first being aware of the possibility of Terson syndrome and then arranging for evaluation by an ophthalmologist. Patients who are able to describe their symptoms typically report an acute decrease in vision in 1 or both eyes in the setting of a recent severe headache or head trauma.
Diagnosis
Given the notable incidence of TS in patients with intracranial hemorrhage, SAH, or traumatic brain injury—especially in patients with a loss of consciousness or low initial Glasgow Coma Scale—screening for TS is important once the patient is medically stable.2,3
Fundus examination. Funduscopy is the gold standard for detecting and diagnosing TS. Hemorrhages involving multiple intraocular layers may be seen; they can manifest as a “double ring” sign, in which blood is present below the internal limiting membrane and posterior hyaloid.9 Patients may also have a loss of the red reflex.
TS has been linked with development of macular holes, epiretinal membranes, retinal folds, proliferative vitreoretinopathy, retinal detachment, and optic nerve sheath hemorrhage as early as 1 week after onset.9
Imaging. It can be challenging to identify this pathology by funduscopy if the view is obscured by vitreous hemorrhage; in such cases, other modalities, such as B-scan ultrasonography, should be considered to aid in the diagnosis. In a study of patients with SAH, B-scan ultrasonography was 100% sensitive and specific for identifying vitreous or preretinal hemorrhages and 44% sensitive for identifying intraretinal hemorrhages. Head CT scans, in contrast, were 60% sensitive for detecting preretinal hemorrhages and 32% sensitive for any intraocular hemorrhage.4 Thus, these modalities may be viable screening options.
Management and Outcomes
Ophthalmologic. The ophthalmic prognosis for TS patients is quite good. Many intraocular hemorrhages resolve spontaneously over several months. For those that do not, vitrectomy has been successful in improving visual outcomes.5,10
In a series of 25 TS eyes undergoing vitrectomy, 88% of eyes achieved 20/30 vision or better.5 A study of 44 vitrectomized TS eyes found that patients who underwent early vitrectomy (within 90 days of vitreous hemorrhage) achieved better visual outcomes than those who were operated on after 3 months.11 Thus, many experts advocate for early vitrectomy, especially in cases of bilateral vitreous hemorrhages, dense unilateral hemorrhage, or hemorrhages in young children.5,7,9,10
The other types of retinal pathology associated with TS, including retinal detachment and macular holes, can also have profound effects on vision if not promptly recognized and treated.9
Neurological and systemic. In terms of neurological and survival outcomes, the prognosis for these patients is notably poor. In a systematic review of outcomes in SAH, patients with TS had a risk of mortality almost 5 times higher than that of patients with SAH alone (50% vs. 11%, respectively, among patients studied prospectively).1 Furthermore, TS patients who do survive have significantly lower Glasgow scales at 3 months than SAH patients without TS.2 Thus, it is important for TS be recognized early, not only for its prognostic significance and impact on patients’ neurorehabilitation efforts, but also because its complications may lead to permanent vision loss if left untreated.
Key Points
- When medically stable, patients with intracerebral and subarachnoid hemorrhages should receive a prompt funduscopic exam to evaluate for TS.
- B-scan ultrasonography or CT head scan can be used as to screen for TS.
- Early vitrectomy should be considered in patients with severe vision loss or bilateral hemorrhages and in young children at risk for amblyopia.
- The presence of TS is associated with a worse neurological prognosis and higher risk of mortality.
___________________________
1 McCarron MO et al. J Neurol Neurosurg Psychiatry. 2004;75(3):491-493.
2 Czorlich P et al. Neurosurg Rev. 2015;38(1):129-136.
3 Czorlich P et al. J Clin Neurosci. 2016;33:182-186.
4 Bauerle J et al. J Neuroimaging. 2016;26(2):247-252.
5 Gnanaraj L et al. Retina. 2000;20(4):374-377.
6 Manschot WA. Am J Ophthalmol. 1954;38(4):501-505.
7 Weingeist TA et al. Ophthalmology. 1986;93(11):1435-1442.
8 Gress DR et al. J Neuroradiol. 2013;40(4):312-314.
9 Ko F, Knox DL. Ophthalmology. 2010;117(7):1423-1429.e1422.
10 Schultz PN et al. Ophthalmology. 1991;98(12):1814-1819.
11 Garweg JG, Koerner F. Acta Ophthalmol. 2009;87(2):222-226.
___________________________
Dr. Rowlands and Dr. Chiu are residents in ophthalmology at NYU Langone Health, in New York. Dr. Schuman is professor and chairman of ophthalmology, and professor of neuroscience and physiology at NYU Langone Health, and professor of electrical and computer engineering at NYU Tandon School of Engineering. Relevant financial disclosures: None.
Acknowledgment: The authors thank Yasha Modi, MD, for his helpful comments.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Chiu None
Dr. Rowlands None
Dr. Schuman Aerie: C; Alcon: C; Annexon: C; Elsevier: P; IOP Medical: C;O; Massachusetts Eye and Ear Infirmary: P; Ocugenix: C,O,P; Opticent Health: C,O; Pfizer: C; Shire: C; Slack/Vindico: C; Zeiss Optical: P. Additionally, rights in intellectual property are assigned to the University of Pittsburgh.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|