By Justine Philteos, BA, Harleen Bedi, HBSC, MD, and Edsel Ing, MD, FRCSC, MPH, CPH
Edited By: Steven J. Gedde, MD
Download PDF
Ramon Silva,* a 41-year-old personal trainer, had developed a painless, yellowish lump on his right eyelid about 12 months earlier. The lesion was nonprogressive and had no associated discharge. Mr. Silva had visited several physicians for the lump, which was resistant to conservative medical treatment, including warm compresses and several trials of topical antibiotic ointment and steroid ointment. By the time he was referred to our oculoplastics clinic for an opinion, he was frustrated about the lack of improvement in this lesion.
We Get a Look
On examination, Mr. Silva’s visual acuity was 20/20, and his intraocular pressure was 16 mm Hg in both eyes. The pupils were equal, round, and reactive, with no afferent pupillary defect. Examination of the conjunctiva, cornea, anterior chamber, and fundus demonstrated no abnormalities.
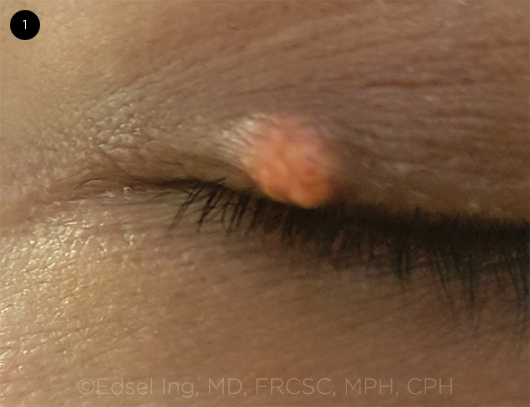
A firm, yellowish, elevated nodule, measuring approximately 3 × 4 mm, was noted on the temporal aspect of his right upper eyelid (Fig. 1). There was no inflammation, ulceration, bleeding, or discharge. The lid architecture was normal, and we saw no madarosis or telangiectatic vessels. Lid eversion revealed normal-appearing forniceal and bulbar conjunctiva, with no posterior extension of the mass.
His previous ophthalmic history was unremarkable. He had been taking indomethacin and oxycodone-acetaminophen (Oxycocet) since having knee surgery in 2016 for a sports-related injury. Review of systems was negative for skin cancer in our darkly pigmented patient, and Mr. Silva said he had no cutaneous lesions elsewhere.
 |
|
WE GET A LOOK. The patient presents with a lesion of the upper right eyelid.
|
Differential Diagnosis
The lack of erythema and inflammation was not typical of chalazion. The lesion did not have the malignant features or morphology of basal cell carcinoma, squamous carcinoma, or sebaceous carcinoma. The nodule lacked the violaceous hue that is commonly seen in a pilomatrixoma. Subepidermal calcification tends to be less yellow than this patient’s lesion. The mottled opacification was not characteristic of epidermal inclusion cyst. Xanthogranuloma was in the differential, but the patient had no other lesions elsewhere.
Further Testing and Biopsy
An incisional biopsy was performed. At the time of the biopsy, the lesion was remarkably dry, gritty, and whitish-yellow. The specimen was submitted to the lab in formalin.
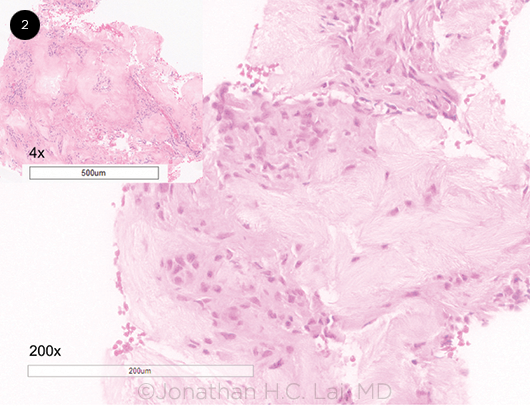
Low-power magnification (4×) of the hematoxylin-eosin (H&E)-stained specimen revealed pale, basophilic amorphous areas surrounded by inflammatory infiltrate composed of lymphocytes, histiocytes, and occasional giant cells. A high-power view (200×) exhibited pinkish aggregates of wispy acellular material surrounded by a palisade of histiocytes and foreign body giant cells (Fig. 2). The pale, basophilic granular material represents a proteinaceous matrix housing needlelike spaces in radial array. These spaces are left behind when urate crystals are dissolved by formalin fixation.
 |
|
HISTOLOGY. Fragments of fibrous tissue studded with basophilic, acellular wispy material associated with a brisk inflammatory infiltrate composed of lymphocytes, histiocytes, and occasional multinucleated giant cells, with an intact epithelium (hematoxylin- eosin stain, inset ×4, high-power section ×200). The pale, basophilic granular material represents the proteinaceous matrix housing needlelike spaces in a radial array. These spaces represent outlines of dissolved urate crystals after formalin fixation.
|
Our Diagnosis
From the histologic appearance of this eyelid lesion, we determined that it was a tophus, which is pathognomonic for gout. This case illustrates an uncommon ocular manifestation of a common systemic disease.
At the follow-up exam three weeks later, the eyelid was completely healed without scarring. Mr. Silva’s uric acid level measured at 654 mmol/L (upper limit of normal, 430 mmol/L), but he elected not to take uricosurics, preferring to control his condition with diet and exercise. On direct questioning, Mr. Silva said that he had been diagnosed with gout a few years earlier.
Discussion
Gout is the most common inflammatory arthritis, characterized by the abnormal deposition of monosodium urate crystals in and around joints such as the metatarsophalangeal joint of the hallux (podagra). The crystals may also be deposited in the skin as potentially disfiguring subcutaneous tophi.
Risk factors for gout include male sex, age, and obesity. Impaired renal excretion of uric acid, overproduction of uric acid, and excess dietary intake of purines contribute to the pathophysiology of this crystalline arthropathy.
Gout is associated with metabolic syndrome, cardiovascular disease, and renal disease. Although gout was traditionally considered a disease affecting only the affluent, that is no longer true because of the increasing trend in obesity.
Gout and the eye. Although gout is a very common disease with an estimated prevalence of up to 4% in North America,1 eyelid tophi are a rare manifestation. Our review of literature found only six other cases of tophaceous gout of the eyelid (Table 1).2-7 The average age at presentation was 50 years, with a median age of 44 years. There was a marked male predominance, which is consistent with the epidemiology of gout.
The patients with eyelid manifestations had preexisting gout ranging from three to 20 years. All cases developed yellowish or whitish tophi in the upper lid or canthi. Four of the six patients reported associated gouty arthropathy involving the first metatarsal, elbow, ankle, and finger joints.
Tophi are pathognomonic of gout. In addition to eyelid tophi, other ophthalmic manifestations of gout include orbital tophi, persistent subconjunctival hemorrhage, corneal crystals, and uveitis.8
Systemic associations with tophi. The development of tophi corresponds with early age of gout onset, longer duration of hyperuricemia, and higher serum urate levels. Tophaceous lesions usually develop in patients after 10 years of hyperuricemia. However, tophus formation may occur sooner in individuals with myeloproliferative disorders, chronic renal disease, and long-term diuretic use, as well as those with early-onset gout. An isolated normal uric acid level does not exclude the diagnosis of gout, just as an elevated uric acid level is not diagnostic of gout. Occasionally, gout may be triggered by a rapid drop in uric acid levels, and up to 30% of men with gout may have low or normal uric acid levels at the time gout symptoms present.9
Biopsy tips. Alcohol is the ideal fixative for preservation of gout crystals because they are soluble in formalin and water. Although formalin dissolves uric acid crystals, the needlelike spaces seen on H&E, as in our patient, are often diagnostic. Ancillary studies are required only if there is an inadequate amount of tissue, a paucity of crystals, or an unusual histologic appearance.
If gout is suspected and the characteristic needlelike spaces are not seen on conventional formalin-fixed H&E preparation, staining with nonaqueous alcoholic eosin or Carnoy fixative can be done to preserve the sodium urate monohydrate deposits. Birefringent crystals can be subsequently visualized under a polarizing microscope, revealing negative birefringence (yellow color) when the long axis of the crystal is oriented parallel to the polarized light. If the specimen has already been submitted in formalin for less than 12 hours, an unstained coverslip technique with 10-μm thick slides can sometimes reveal negative birefringence.
Classification. The 2015 American College of Rheumatology and European League Against Rheumatism (ACR-EULAR) gout classification criteria are the most widely used for diagnosis of gout. The 10 criteria include history, clinical examination, uric acid level, laboratory results, and radiologic findings to predict the likelihood of an acute gout flare.10
Although tophi are mentioned in the ACR-EULAR document, the ocular adnexal location is notably absent. Despite the rarity of a tophaceous lesion in the eyelid, it is important to be aware of this atypical manifestation of a common systemic disease to expedite diagnosis. When faced with a nonresolving eyelid lump, the clinician should consider a biopsy. A periocular tophus could be the precursor of a painful systemic disease and subclinical renal dysfunction.
Table 1. Case Reports of Eyelid Tophi
|
| Author |
Age (years) |
Sex |
Duration of gout (years) |
Duration of lesion |
Location of lid lesion |
Lesion size (mm) |
Other lesions |
| De Monteynard2 |
62 |
F |
- |
2 days |
Lateral canthus |
- |
- |
| Morris3 |
44 |
M |
- |
1 year |
Lateral canthus |
6 × 5 × 4 |
- |
| Yen4 |
27 |
M |
3 |
3 months |
Medial canthus |
11 × 5 × 5 |
1st metatarsal (MT) |
| Jordan5 |
68 |
M |
20 |
2 years |
Medial canthus |
5 × 6 × 4 |
Elbow |
| Yang6 |
64 |
M |
- |
9 years |
Middle upper lid |
14 × 10 × 8 |
Fingers |
| Nakatsuka7 |
41 |
M |
10 |
1 year |
Lateral canthus |
4 × 7 × 4 |
Ankle, 1st MT |
| Present case |
41 |
M |
3 |
1 year |
Temporal upper lid |
4 × 5 × 4 |
Knee? (no surgical specimens were sent to pathology for gout) |
Conclusion
Eyelid lesions can point to the diagnosis of systemic disease. Ophthalmologists should be aware of the ophthalmic manifestations of gout,8 which may be the initial presenting sign of poorly controlled hyperuricemia. Tophaceous gout should be in the differential diagnosis if a middle-aged patient, usually male, presents with a nonulcerated, yellowish-white lid lesion, especially if it is gritty and dry at the time of biopsy. Patients should be asked about a preceding history of gouty arthritis, particularly podagra. If gout is suspected, the specimen is ideally submitted in alcohol because the crystals are soluble in formalin.
___________________________
*Patient name is fictitious.
___________________________
1 Kuo CF et al. Nat Rev Rheumatol. 2015;11(11):649-662.
2 De Monteynard MS et al. Bull Soc Ophtalmol Fr. 1986;86(1):53-54.
3 Morris WR, Fleming JC. Arch Ophthalmol. 2003;121(8):1195-1197.
4 Yen CC et al. Am J Ophthalmol. 2005;140(3):542-544.
5 Jordan DR et al. Ophthalmic Plast Reconstr Surg. 2008;24(5):403-404.
6 Yang CC et al. Ophthalmic Plast Reconstr Surg. 2008;24(5):404-406.
7 Nakatsuka AS. Case Rep Ophthalmol Med. 2016. doi:10.1155/2016/9786810.
8 Ao J et al. Clin Exp Ophthalmol. 2017;45(1):73-80.
9 Bădulescu M et al. Rev Med Chir Soc Med Nat Iasi. 2014;118(4):942-945.
10 Neogi T et al. Arthritis Rheumatol. 2015;67(10):2557-2568.
___________________________
Ms. Philteos is a medical student, Dr. Bedi is an ophthalmology resident, and Dr. Ing is associate professor and specialist in oculoplastics, strabismus, and neuro-ophthalmology. All are at the University of Toronto in Toronto, Ontario, Canada. Financial disclosures: None.