Download PDF
The modern practice of ophthalmology excels at myriad remarkable treatments and outcomes. It also excels at producing appalling amounts of discarded packaging and other garbage.
Is it possible to take the profession in a greener direction—to follow the dictum “First, do no harm” not only for patients but also for the environment?
A growing number of ophthalmologists argue that it’s time to tackle this challenge. “As ophthalmologists, the impact we have on our environment is becoming clearer and more significant than ever before,” said Alan L. Robin, MD, at the University of Michigan in Ann Arbor and Johns Hopkins University in Baltimore. But as Dr. Robin noted, “changing our habits is not a matter of fundamentally changing what we do—patient care is always our first priority. Practicing green medicine is about acknowledging the waste we produce, reducing our impact wherever possible, and spreading the word.”
A Rising Tide of Waste
“There is no doubt that modern ophthalmic practice creates an unbelievable amount of waste on a daily basis,” said Rengaraj Venkatesh, MD, at the Aravind Eye Hospital in Pondicherry, India. “Think about the disposables involved in a typical cataract surgery in the United States: Single-use eyedrops. Single-use phaco tips, tubing, sleeves, and other accessories. Single-use gowns and sheets. Single-use surgical markers. And don’t forget the cartons, the boxes, the plastic wrapping, and the paper inserts that come with IOLs and other packaging. Now multiply that by the millions. The generation of ophthalmic waste is unprecedented.”
What’s behind this avalanche of waste? Key factors include regulatory policy, economic factors, and a general frame of mind.
A wasteful mindset. “In many high-income countries, ophthalmology’s waste largely stems from a single-use disposable mindset that filters down to almost everything we do,” said Jeff H. Pettey, MD, at the University of Utah in Salt Lake City. “We’re not aware of the volume we throw away or where it ends up, nor are we conscious of the life cycles of our medical products—everything from how they are produced to how they are discarded. The lack of awareness drives our waste problem.”
The herd mentality plays a large role as well, Dr. Venkatesh added. “When I travel and meet other ophthalmologists to discuss the merits of the Aravind system [which has implemented measures to reduce waste], I get the sense that many physicians don’t want to antagonize the status quo or question what they’ve become accustomed to. Perhaps they’ve been taught that the cleaning and sterilization of reusable instruments is simply not efficient. Perhaps it’s simply a fear of ‘What if? What if something were to happen to my patient?’”
Safety constraints. Maintaining patient and consumer safety is the cornerstone of the U.S. regulatory system. For instance, the Federal Food, Drug, and Cosmetic Act requires that all drugs and devices labeled for single use be used as such, even if a surgeon believes they can be safely reused. But the notion that single-use instruments and devices are safer for patients is not always supported by actual evidence, said David F. Chang, MD, in private practice in Los Altos, California. “We should challenge industry to provide us with safe, reusable options that minimize waste as well as cost.”
Instructions for use. For example, consider the common international practice of reusing phacoemulsification tips. In the United States, surveyors that accredit or license ambulatory surgical centers (ASCs) do not allow for any deviation from the manufacturer’s instructions for use (IFU). Some manufacturers specify a single use, while others label for multiple uses, even though these are virtually the same titanium tips. Dr. Chang and his colleagues tested both reusable and single-use phaco tips in the lab on porcine nuclei with multiple simulated uses. They found no structural differences between the two types of tips—nor any changes that would pose a safety risk to patients.1
“IFUs are often worded in order to limit liability and may be overly cautious by addressing a worst-case scenario,” Dr. Chang said. “But surgeons should be allowed to use their judgment and discretion in certain situations—much as we are allowed to practice medicine off label. There is no way that emulsifying a soft cataract could damage a single-use tip. On the other hand, I might damage a multiuse tip against a chopper on its first case. As long as accreditation and licensing require strict adherence to these IFUs, it falls on industry to provide for rational reuse and [to allow for] surgeon discretion.”
Controlling infections. One of the strongest arguments in support of single-use devices is infection control. But endophthalmitis rate studies by Dr. Chang and clinicians at the Aravind hospitals raise the possibility that many of the costly, but unquestioned, infection control regulations for ORs in the United States could be unnecessary.2
“Approximately 60% of Aravind’s surgical volume is performed in charity patients, which necessitates maximizing surgical efficiency and eliminating wasteful, unnecessary practices,” Dr. Chang pointed out. “So Aravind routinely reuses gowns, gloves, irrigation/aspiration tubing, phaco tips, cannulae, irrigating solution, and medications. What’s remarkable is that their endophthalmitis rate was 0.01% in their last 335,000 consecutive phaco cases using routine intracameral moxifloxacin.” (See “Pushing the Boundaries of Green Surgery.”)
The profits of waste. Unfortunately, however, there’s money to be made in waste, Dr. Robin noted. “The waste of medication in the United States is almost criminal. Look at the example of anesthesia in a single cataract surgery,” he said. “During a single procedure, a nurse anesthetist will use 1 to 5 mL of a 20-mL bottle of propofol. Where does the rest go? At best, a hazardous materials container. At worst, [into] the garbage can and into our drinking water.”
The same wasteful patterns are also evident outside of surgery, said Dr. Robin, especially with regard to eyedrops. Microdrops could reduce pharmaceutical waste and provide cost savings to patients, he pointed out.
This is not a new concern: In 1992, Dr. Robin was the principal investigator of a research team that was tasked by a leading pharmaceutical manufacturer to assess a smaller drop for one of its glaucoma drugs. The company became aware that many patients were complaining of stinging and burning due to drop overflow. So the team recruited 29 patients to test a microdrop (an amount almost half the size of the standard 30-mL drop). The microdrop was just as effective at reducing IOP as the larger drop—and all of the patients preferred its use.3
A similar study in 2006 funded by another pharmaceutical company came to the same conclusion.4 But the manufacturers never took the products to market. Now, however, technology—in addition to heightened awareness and environmental advocacy—may be spurring change (see “A Green Mainstream”).
 |
|
WASTE NOT. What can be recycled or reused? How can ophthalmic practices and surgery centers rethink sterilization and disposal practices?
|
Call to Action
“We’re limited in the United States as to how we can practice green medicine,” said Dr. Pettey. “But if we maintain our focus on quality patient care and safety, we can find ample avenues and simple opportunities in our daily lives to decrease our footprint. The little things can really add up.”
Some steps to consider:
Cultivate a green culture. Whether you’re in private practice, academia, or anything in between, it begins with the habits and customs of your organization. “Establishing a green way of thinking is paramount,” said Dr. Pettey. “Putting a recycling can in the break room is a start, but sustainability is about trying to create a top-to-bottom approach where everyone is focused on minimizing environmental impact as a team. Mindfulness is the secret sauce that can bring about larger change.”
Communicate to your staff why reducing waste is important and encourage ideas from every level. Once you’ve created that green mindset, each person can take ownership of their role with a common goal, said Dr. Pettey. “If you’re in the OR, everyone involved can reflect on that culture when making decisions. ‘Do we need to open up that surgical pack for this specific procedure? Or is it just part of our routine? Is it something that we can reuse safely? Can it be recycled? How?’”
These new habits will need constant attention, nurturing, and revisiting, he added. “It’s a lot like a garden. It really requires you to be deliberate and intentional—otherwise things will die on the vine and go by the wayside.”
This intentional approach is key to the success of the Aravind system, said Dr. Venkatesh. New personnel hear the same story—the “Bedsheet Story”—upon walking in the door of an Aravind hospital their first day. “When your bedsheet becomes frayed and tattered after years of use, you don’t just throw it in the trash,” he said. “You cut out the damage and make a pillow cover out of it. When the pillow cover becomes frayed and tattered after years of use, you don’t just throw it in the trash then either. You cut out the damage and make a dust cloth. Only then, when you have gotten every use possible out of your original bedsheet, is it disposable.”
Reduce, reuse, recycle, repeat. Reuse underscores everything that happens at Aravind, said Dr. Venkatesh. “We don’t throw anything away. For example, when the teeth of our surgical forceps become blunted from overuse, we remove the teeth and make cotton swab tweezers.”
For those working with U.S. regulations, the challenge then becomes, how can you get the most out of your materials?
Switch to reusable if possible. “Entire single-use instrument sets are thrown away at the end of procedures,” said Dr. Pettey. “Consider multiuse alternatives. And if you don’t know of any, ask your colleagues what they are doing or reach out to manufacturer reps in your network for advice.”
If the surgical situation does dictate single-use supplies, minimize as many nonessentials as possible in your prepackaged surgical packs. “Get with the other ophthalmologists in your group and evaluate your standardized equipment list,” said Dr. Robin. “Ask yourselves, ‘How often do we use item A? Is it a majority of surgical cases or just on an as-needed basis? Is it necessary to bring it to the table and dispose of it unused every time?’”
Expand recycling efforts. “We should of course recycle everything [we possibly can],” said Dr. Robin. “And in my experience around the country, it’s not being done nearly as much as it could be. I’ve seen entire bags of garbage thrown into the trash before a surgery even begins.” But most of the waste materials that come out of a surgical center’s back end aren’t meant for the landfill; they’re recyclable. Examples include paper products, plastics, glassware, and other items that don’t come into contact with a patient, including toner cartridges, fluorescent lights, steel, aluminum, and several different types of batteries.
Rethink anesthesia. Dr. Robin’s wife recently underwent cataract surgery at Aravind Eye Hospital. She received no systemic anesthesia, no IV equipment or anesthesiologist was in the room, and she was entirely comfortable during the process. The experience helped Dr. Robin rethink the role of general anesthesiologists in eye care. “Can we reduce unnecessary sedation? It’s already happening around the world for cataracts, and most high-volume surgeons in the United States can perform without it,” he said. “By eliminating the presence of an anesthesiologist whenever possible, we can simplify our procedures and produce less waste—fewer unused drugs discarded, less discarded tubing in our landfills, and fewer disposables of every kind. It’s another small step we can take.”
Rethink sterilization. Last year, the Ophthalmic Instrument Cleaning and Sterilization Task Force—which is cochaired by Dr. Chang and comprised of representatives from the Academy, the American Society of Cataract and Refractive Surgery, and the Outpatient Ophthalmic Surgery Society—published updated specialty-specific guidelines for anterior segment surgery.5 “The guidelines include evidence-based support of short-cycle sterilization for sequential same-day cataract surgery,” said Dr. Chang.
Approved containment devices can be used without wrapping if allowed by the sterilizer IFU. “This also reduces the waste, energy burden, and expense associated with instrument processing and sterilization,” Dr. Chang noted.
Reduce patient travel. Being green isn’t only limited to what’s going on in your office or surgical center, Dr. Venkatesh noted. “A significant portion of our carbon footprint involves the extensive travel of patients. And that’s largely ignored by physicians. A patient will drive hundreds of miles, sometimes three or four times a week, to receive the care they need—one for an evaluation, another for a procedure, another for a follow-up, and so on.”
There isn’t always a simple solution to such a complex problem, Dr. Venkatesh said, but ophthalmologists should at least be cognizant of how much they are asking patients to travel. “Can we develop networks with other clinicians who are closer to patients in order to help alleviate travel time? Can we use telemedicine to help reduce the amount of pre- and postoperative visits? Can we use artificial intelligence to change the nature of the typical in-office medical encounter?”
 |
|
NO SEDATION. The majority of Aravind surgeries are conducted with topical anesthesia. A side benefit: less environmental waste.
|
Join Forces
Helping reverse wasteful trends in ophthalmology requires individual effort, but the support of organized medicine is critical for overcoming the larger legal-political challenges. How can you get involved?
Collect data. Clinical research is a critical first step, said Dr. Robin. “We need to show the powers that be as much medical waste data as possible. We need good data on cataract surgeries, glaucoma surgeries, high-volume surgeries of all sorts. Without it, we’re powerless.”
Cross-specialty collaboration is needed as well, he said. “We need ophthalmologists from all walks of life to come together with infectious disease specialists and epidemiologists to empirically evaluate the efficacy of today’s regulations that govern medical waste. The more data [that’s available], the more convincing our argument.”
Advocate. Will you take up the mantle and be the messenger? “If I were to give one piece of advice for overcoming the regulatory burdens affecting our environment, it would be to get involved in advocacy efforts,” said Dr. Pettey. He pointed out that national medical associations, such as the Academy, provide participants with “the loudest voice and the most robust resources for affecting large-scale change when it comes to regulatory burdens that aren’t based in valid or sound evidence.”
And the power of advocacy is real. Most recently, the Illinois State Medical Society heard the voices of ophthalmologists speaking out against the unnecessary disposal of medications. As a result, the society passed a resolution calling for certain patients in ER, OR, and ASC settings to be able to receive stock supplies of glaucoma drops and other antibiotic and anti-inflammatory medications upon discharge. The next step will involve working with the state department that oversees all Illinois pharmacies to achieve a regulatory change.
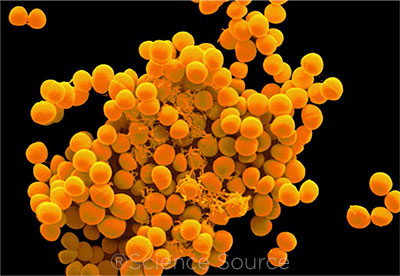 |
|
CONCERN. Staphylococcus epidermis (shown here) is a leading culprit in endophthalmitis. But are U.S. infection control regulations overly restrictive?
|
A Green Mainstream
It will take time to turn the minds of many, but as Dr. Pettey noted, there’s a sense of inevitability when it comes to sustainable health care. “As a member of the Academy’s Young Ophthalmologist Committee, I’m witnessing a refreshing [level of] awareness and concern among young physicians. As they become the leaders of tomorrow, I predict green medicine will become a core value for all of ophthalmology.”
And in a positive note of change on the biopharmaceutical front, one company, Eyenovia, recently received patents for an innovative drug delivery system that uses piezo-print technology to apply microdroplets of medication to the eye in less than 80 milliseconds, much like an inkjet applies a pixel-sharp amount of ink to paper.6
This type of disruptive technology is only going to become more common, said Dr. Robin. And when it does, physicians will really start to take notice of how practicing green ophthalmology and reducing waste is good for patients, the planet, and the bottom line. “As ophthalmologists, we want our patients and our profession to do well. But we have to ask ourselves, ‘If we could do better with less, why are we doing worse with more?’”
___________________________
1 Tsaousis KT et al. J Cataract Refract Surg. 2018;44(1):91-97.
2 Haripriya A et al. Ophthalmology. 2017;124(6):768-775.
3 Vocci MJ et al. Am J Ophthalmol. 1992;113(2):154-160.
4 Fiscella R et al. J Ocul Pharmacol Ther. 2006;22(6):477-482.
5 Chang DF et al. J Cataract Refract Surg. 2018;44(6):765-773.
6 Pasquale LR et al. Clin Ophthalmol. 2018;12:2451-2457.
Reducing Surgery’s Carbon Footprint
Destruction of the environment is one of the most significant challenges of our century—and the medical industry plays no small role in this global issue.
 Evidence indicates that the surgical systems in most developed countries produce a carbon footprint that is not environmentally sustainable.1 For instance, one recent study confirmed that the U.S. health care sector is responsible for 10% of the country’s total greenhouse gas emissions, 10% of its smog formation, and 9% of its ozone depletion.2 Evidence indicates that the surgical systems in most developed countries produce a carbon footprint that is not environmentally sustainable.1 For instance, one recent study confirmed that the U.S. health care sector is responsible for 10% of the country’s total greenhouse gas emissions, 10% of its smog formation, and 9% of its ozone depletion.2
How does ophthalmology contribute to harming the environment? Consider this: Cataract surgery is the most commonly performed surgical procedure in the United States and one of the most common surgeries performed worldwide. According to researchers in the United Kingdom, the carbon footprint of a single phacoemulsification procedure is equivalent to the energy consumed by the average U.K. person during any given week. Or in other words, performing the surgery is the equivalent of driving an automobile over 400 miles.3,4
Eye ASCs and ORs can take a number of steps to reduce their environmental emissions, starting with the basic mantra of “reduce, reuse, and recycle” and extending toward rethinking the supply chain and adopting green building practices. For more information, see Healthcare Without Harm (https://noharm.org) and Practice GreenHealth (https://practicegreenhealth.org).
___________________________
1 Thiel CL et al. J Cataract Refract Surg. 2017;43(11):1391-1398.
2 Eckelman MJ, Sherman J. PLoS One. 2016;11(6):e0157014.
3 Venkatesh R et al. Curr Opin Ophthalmol. 2016;27(1):82-88.
4 Morris DS et al. Eye (Lond). 2013;27(4):495-501.
|
Pushing the Boundaries of Green Surgery
The Aravind Eye Care System is challenging conventions about the delivery of health care and showing the world that cutting waste and maintaining high quality can go hand in hand.
Founded over 40 years ago as an 11-bed clinic in a small town in southern India, the network has grown to eight tertiary eye care hospitals and more than 70 primary and secondary eye care centers. Each year, Aravind teams see almost 4 million patients and perform nearly half a million ophthalmic surgeries and procedures—a large majority of which are cataract related.1
How do they do it? They employ a lean approach that focuses on eliminating as much waste as possible and streamlining the patient encounter. “We began with a mission to eradicate needless blindness,” said Dr. Venkatesh. “To meet that kind of surgical volume, we constantly focus on putting good systems in place.”
For example, no appointment system exists at Aravind. Anyone can walk in and receive treatment immediately. “We have open registration from 7:30 a.m. until 5 p.m. Monday through Saturday,” said Dr. Venkatesh. “If somebody is medically cleared for a cataract surgery today, we schedule them for the procedure tomorrow.”
Roughly 1,500 surgeries are performed each day throughout the Aravind network. “Our doctors operate on two tables,” Dr. Venkatesh said. “With the assistance of midlevel ophthalmic personnel, a cataract surgeon will operate on the first patient while the second is being prepped. When the first is complete, the surgeon simply rolls the phaco machine over to the second table, sterilizes with an antiseptic, and proceeds, all while another patient is prepped for the first table. It’s an assembly-line system that maximizes what ophthalmologists do best—surgery.”
It’s also a system designed for efficiency. Ten surgeons can comfortably perform 300 cataract surgeries before lunch. And nothing is casually discarded. Instruments, syringes, and phaco tools are washed, sterilized, and reused the same day. Surgical gloves are discarded after 10 cases, while plastic table drapes are disposed of once daily. Caps, gowns, and masks are laundered at the end of the operating day. Pharmaceuticals are used until either the bottle is empty or the day has ended. The Aravind team has even created their own wetlands to treat wastewater. (For a video overview of the Aravind system’s multifaceted approach, go to aao.org/annual-meeting-video/environmentally-sustainable-high-volume-eye-care-m.)
The reduction in waste is astounding. Aravind generates 250 grams of waste per phacoemulsification—roughly 5% of the waste generated by the same procedure in the United States.2 Moreover, an average trabeculectomy with phacoemulsification performed at Aravind produces half the waste of the same procedure performed in the United States—with similar surgical outcomes and complication rates.3
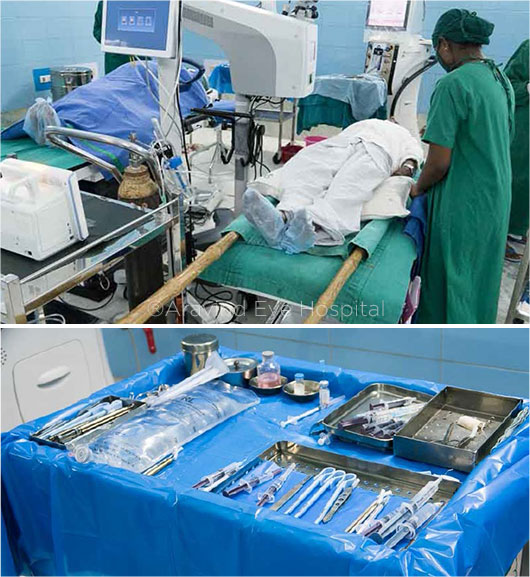
INNOVATION. The two-bed surgical system (top) is a key component of the lean approach in place at Aravind. Surgical instruments (bottom) undergo daily sterilization and reuse.
___________________________
1 Namburar S et al. Am J Ophthalmol Case Rep. 2018;12:87-90.
2 Thiel CL et al. J Cataract Refract Surg. 2017;43(11):1391-1398.
3 Le HG et al. Health Aff (Millwood). 2016;35(10):1783-1790.
|
Meet the Experts
David F. Chang, MD Clinical professor of ophthalmology at the University of California, San Francisco, and in private practice in Los Altos, Calif. Relevant financial disclosures: None.
Jeff H. Pettey, MD Associate professor of ophthalmology, vice chair of education, and medical director of global outreach at the University of Utah’s Moran Eye Center in Salt Lake City. Relevant financial disclosures: None.
Alan L. Robin, MD Professor of ophthalmology at the University of Michigan in Ann Arbor, associate professor of ophthalmology at the Wilmer Eye Institute in Baltimore, and associate professor of international health at the Johns Hopkins Bloomberg School of Public Health in Baltimore. Relevant financial disclosures: None.
Rengaraj Venkatesh, MD Chief medical officer of the Aravind Eye Hospital in Pondicherry, India. Relevant financial disclosures: None.
Full Financial Disclosures
Dr. Chang Carl Zeiss: C; Eyenovia: O; ianTech: C,O; Icon Bioscience: O; iDrops: C,O; Ivantis: C,O; Johnson & Johnson: C; Mynosys: C,O; PowerVision: C,O; Presbyopia Therapies: O; Omeros: C,L; PogoTec: O; Presbyopia Therapies: O; Rapid Pathogen Screening: O; Sun: C,L; Tissue Tech: O; TrueVision: O; uBeam: O.
Dr. Pettey None.
Dr. Robin Aerie: C; Novaliq: C; Superior Vision: C.
Dr. Venkatesh None.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|