Download PDF
In an ideal world, every patient who is undergoing intravitreal treatment for neovascular age-related macular degeneration (AMD) would return faithfully, 12 times a year, for a lengthy visit that includes a clinical examination, retinal imaging, and, possibly, an intravitreal injection of an anti-VEGF drug. In the real world, however, patients frequently have trouble meeting this monthly schedule—and that places their vision at risk.
“This comes up every day in my practice,” said K. Bailey Freund, MD, in private practice in New York City. “Some of these patients are over 100 years old, and just to get them into the office can be a difficult ordeal for themselves and their family members.”
One solution being adopted by retinal physicians is “treat and extend,” a dosing strategy that can enable patients with wet AMD to go as long as 12 weeks between office visits and injections.
Gaining Popularity
Despite limited evidence, positive clinical experiences with the treat-and-extend approach have fueled the spread of the protocol in retinal practices around the world, Dr. Freund noted.
A 2015 survey of retinal subspecial ists found that 66.7% of the 586 U.S. respondents preferred a treat-and-extend regimen for their wet AMD patients. Internationally, about a third of the retinal physicians who responded in Europe, Asia, and Latin America expressed a preference for this approach to wet AMD therapy.1
An incremental approach. The specifics vary among practitioners, but in general terms, the regimen follows this course, according to Dr. Freund: The patient’s retina is first cleared of macular fluid and retinal hemorrhages with a series of monthly injections of the chosen drug. Then the physician begins extending the interval between treatments, 2 weeks at a time, as long as the retina remains dry and stable.
Who benefits? Treat and extend “applies mainly to neovascular AMD. You can also use it for treating patients with diabetic macular edema or retinal vein occlusions,” said Dr. Freund, who led a group that coined the term “treat and extend” in a 2010 paper.2
Coming to a Consensus
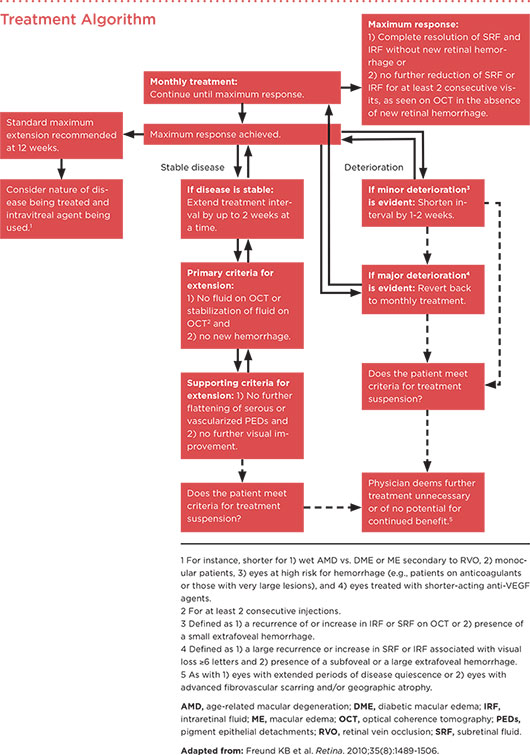
More recently, Dr. Freund was part of an international panel of retinal physicians that developed an algorithm for using treat and extend against retinal diseases, based on their clinical experience and information from published studies.3 (For the algorithm, see below.)
A key tool: OCT. The panel recommended monitoring for subretinal and intraretinal fluid with optical coherence tomography (OCT), in order to assure the retina’s readiness for a longer treatment interval. In most cases, the experts deemed fluorescein angiography unnecessary as therapy proceeds.
Timing the extensions. “Let’s say that after 3 monthly injections, the patient achieved what you determine is the maximum response, based on OCT,” Dr. Freund said. “Then you might have the patient come back in 6 weeks; and if they’re still stable, then you would inject and have them return in 7 or 8 weeks. So you could be seeing patients half as often, but they [would] get a treatment every time they come.”
When to reconsider. The algorithm recommends a maximum interval between injections of 12 weeks and notes that the injection interval sometimes must be shortened, Dr. Freund said. “If there are minor changes between visits, such as a small increase in fluid, then all you do is go back to the interval that had kept them completely dry. Only in the situation of a major event, such as a large hemorrhage, would you revert back to monthly treatment.”
Different from PRN. Under treat and extend, the patient receives an anti-VEGF injection at every visit. That is in contrast to OCT-guided pro re nata (PRN), which has been shown to be effective in clinical trials. With PRN, OCT imaging and a clinical exam continue to be performed monthly. However, the patient receives an injection only if the OCT shows a recurrence of fluid or hemorrhage.
(click to expand)
Practical Benefits
“I use treat and extend quite frequently,” said Adrienne Williams Scott, MD, at Johns Hopkins University in Baltimore. “I find it to be a practical regimen in which you can maintain the vision gains from anti-VEGF therapy yet minimize the injections and the burden on patients.”
Pravin U. Dugel, MD, agreed. “In every survey that I know of, treat and extend is the regimen that is most used by retina specialists.” Dr. Dugel is at Retinal Consultants of Arizona in Phoenix and the University of Southern California’s Eye Institute in Los Angeles. In addition to lessening the burden on patients, treat-and-extend regimens help clinicians cope with the unknowns of retinal neovascularization, Dr. Dugel said. “There’s a great deal of variability between patients, and we have no way to predict who will respond and how. So treat and extend seems to be logical.”
Parsing the Evidence
Literature review. The consensus panel on which Dr. Freund served conducted a literature review and found 11 published studies reporting positive outcomes with treat and extend in eyes with neovascular AMD.3 Most of the studies were small or retrospective, and only one—the LUCAS trial—met Level 1 criteria for demonstrating efficacy.4
LUCAS results. The LUCAS researchers found that treat and extend with bevacizumab or ranibizumab gave wet AMD patients mean increases in best-corrected visual acuity (VA) of 7.9 and 8.2 letters, respectively, after 1 year of treatment.4 This was comparable to the VA gains in the CATT study, which employed monthly injections of these drugs; CATT gains were 8.0 and 8.5 letters, respectively, at 1 year.4 (For a second report of LUCAS results, see “Treat and Extend at 2 Years,” below.)
Treat and Extend at 2 Years
The LUCAS researchers compared the efficacy and safety of bevacizumab and ranibizumab for neovascular AMD when the drugs are delivered via a strict treat-and-extend protocol. They found equivalent outcomes between the 2 agents with regard to VA and central retinal thickness (CRT).1
A total of 339 patients were available for analysis. At the 2-year mark, bevacizumab was equivalent to ranibizumab, with 7.4 and 6.6 letters gained, respectively. Mean CRT decreased by –113 µm in those who received bevacizumab and by –122 µm in those who received ranibizumab. All told, those in the bevacizumab group received 18.2 injections, versus 16.0 injections in the ranibizumab group. When serious adverse events were evaluated, no significant differences emerged between the treatment groups.
___________________________
1 Berg K et al. Ophthalmology. Published online Oct. 15, 2015. doi: 10.1016/j.ophtha.2015.09.018.
|
A Practical Compromise?
Dr. Scott said that she views treat and extend as a therapeutic option for patients who can’t—or won’t—comply with a monthly dosing regimen. “Treat and extend is really more of a compromise,” she said. “Because when you look at Level 1 evidence from large pivotal clinical trials, the best results have come with monthly treatment. The patients who are injected monthly tend to do the best in terms of less vision lost, more vision gained, and drier OCTs.”
Dr. Dugel agreed. “We know in real life that none of us are getting visual outcomes as good as the published trials, because the treatment burden would be far too arduous.” He said he moves patients who have had trouble coming in for monthly visits to a treat-and-extend approach. “I tell them that I think the results we get will probably be as good—or almost as good—as they would be with monthly surveillance. But that is based on my clinical experience, not on Level 1 data.”
While Dr. Freund agrees that data supporting treat and extend are not as robust as those for monthly treatment, he emphasized the chronic nature of neovascular AMD. “The reality is that some patients will require treatment for 10 years or longer. This fact necessitates a viable management strategy, such as treat and extend, that can be maintained for this duration.”
___________________________
1 American Society of Retina Specialists. 2015 Global Trends in Retina Survey Results. Accessed Oct. 23, 2015.
2 Engelbert M et al. Retina. 2010;30(9):1368-1375.
3 Freund KB et al. Retina. 2015;35(8):1489-1506.
4 Berg K et al. Ophthalmology. 2015;122(1):146
___________________________
Dr. Dugel is managing partner at Retinal Consultants of Arizona and the Retinal Research Institute in Phoenix and clinical professor of ophthalmology at the University of Southern California’s USC Eye Institute and Keck School of Medicine in Los Angeles. Relevant financial disclosures: Abbott Medical Optics: C; Acucela: C; Alcon: C; Alimera Sciences: C,O; Allergan: C; Digisight: O; Genentech: C; Novartis: C; Ophthotech: C,O; Ora: C; Regeneron: C; ThromboGenics: C.
Dr. Freund practices at Vitreous-Retina-Macula Consultants of New York and is clinical professor of ophthalmology at New York University Medical Center. Relevant financial disclosures: Bayer Healthcare: C; Genentech: C; Heidelberg Engineering: C; Ohr: C; Optos: C; Optovue: C; ThromboGenics: C.
Dr. Scott is assistant professor of ophthalmology at Johns Hopkins University in Baltimore. Relevant financial disclosures: ThromboGenics: S.
See the disclosure key at www.aao.org/eyenet/disclosures.