Download PDF
Back in 2004, the original intravitreal (IVT) injection guidelines on injection technique and monitoring were published.1 Ten years and many studies later, an expert panel convened to review the literature, discuss various approaches to the procedure, and update the original guidelines. The full 2014 report is available in the journal Retina.2 “It should be read by all ophthalmologists who perform this valuable procedure,” said panelist George A. Williams, MD, at the Beaumont Eye Institute in Royal Oak, Mich.
In 2004—prior to the introduction of anti-VEGF therapy for retinal vascular disease—the number of IVT injections administered annually was less than 100,000, said Harry W. Flynn Jr., MD, at Bascom Palmer Eye Institute in Miami. “There are now an estimated 5 to 6 million IVT injections a year of anti-VEGF agents alone, and that number is growing,” he said. IVT injection is the most common ophthalmic procedure performed in the United States today.2
“As IVT injection has become part of our routine care, there has been an evidence-based evolution in our approach to the procedure,” said Julia A. Haller, MD, at the Wills Eye Hospital in Philadelphia. “We now have ways to maximize safety and minimize cost.”
The 2014 report identified aspects of IVT injection that have advanced over the past decade. It cites the evidence on which each guideline is based and summarizes the points of discussion among the expert panelists. “At the heart of the report are the tables that highlight the areas of general agreement among committee members and that summarize areas with no clear consensus,” said corresponding author Emmett T. Cunningham Jr., MD, PhD, MPH, of San Francisco.
Below, a brief discussion with the panelists focuses on several key guidelines in the new report.
Stop Using Antibiotics
Since the original guidelines were published, a preponderance of evidence has shown that the use of topical antibiotics to minimize endophthalmitis risk is not necessary for IVT procedures. The 2014 guidelines now specifically state that antibiotics are unnecessary pre-, peri-, or postinjection. Dr. Flynn noted that the elimination of antibiotics represents a huge cost savings and reduction in patient burden before and after the procedure.
“There are still some ophthalmologists who use antibiotics, but it’s gradually fading away,” said Dr. Haller. “It goes back to our treating IVT injection as a big procedure rather than the daily high-volume, standard practice it has become for so many retina specialists.” With careful povidone-iodine prep, antibiotics are not only superfluous but also potentially worse than superfluous, according to Dr. Haller. Specifically, she said, “We don’t want to encourage the emergence of resistant organisms in the flora of the eye, which could then lead to more serious infections should endophthalmitis eventuate.”
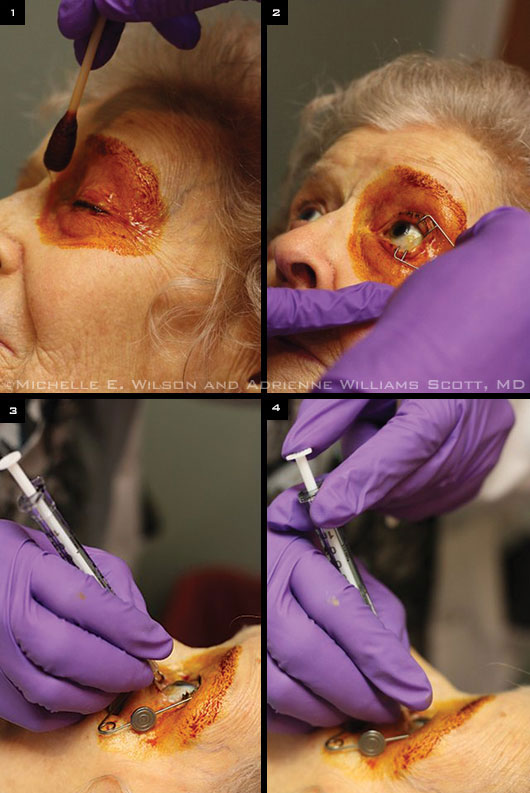 |
|
(1) Application of periocular povidone-iodine eyelid prep. (2) Optional insertion of sterile speculum to separate the lids. (3) Scleral penetration 3.5 mm posterior to the limbus in this pseudophakic patient. (4.0 mm recommended for phakic patients.) (4) Steady and gentle injection of solution in the midvitreous cavity.
|
Limiting Oral Contamination
The spread of aerosolized droplets from the mouth of the patient or medical staff wasn’t mentioned in the 2004 guidelines, but there is now evidence implicating oral contaminants in aerosolized droplets as a potential source of injection-related endophthalmitis. “Both the patient and medical providers should minimize speaking or wear surgical masks during the injection preparation and procedure,” said Dr. Flynn. This is a major addition to the 2014 guidelines.
|
Web Extra: Clinical Update
|
 |
Proper Povidone-Iodine Prep
The panel emphasized the continued importance of applying povidone-iodine to, and avoiding eyelid contact with, the intended injection site and needle. Povidone-iodine (5-10 percent) should be the last agent applied to the injection site before injection. In the case of gel anesthetic, however, povidone-iodine should be applied both before and after the gel. If the gel is applied before the povidone-iodine, it may block the povidone-iodine from contacting the conjunctival surface, according to Dr. Cunningham. This could compromise its effectiveness.
Changes in Contraindications
Anticoagulants. Before the last decade’s worth of experience, retina specialists were worried that IVT injections would raise the risk of intraocular bleeding in patients on anticoagulation therapy. However, no such increase in risk has been observed, according to Dr. Flynn. “It’s simply too dangerous to take people off their anticoagulant therapy for monthly injections,” said Dr. Haller. “Patients don’t bleed in a way that’s medically dangerous. They may have a red eye and dislike how it looks, but that’s just cosmetic.”
Allergies. “True allergy to povidone-iodine is rare,” according to Dr. Flynn. There has never been a reported case of anaphylaxis after the use of topical ophthalmic povidone-iodine, despite many patients reporting an allergy to shellfish or an iodine dye. Dr. Flynn noted that patients occasionally experience localized swelling after the use of povidone-iodine, but he considers this “nuisance” swelling a small price to pay for povidone-iodine’s bacteria-killing properties.
A skin patch test may be considered, but the panel did not recommend routine testing. “In the thousands of IVT injections I’ve done, I’ve never had a patient unable to receive povidone-iodine,” said Dr. Flynn. Of course, if a patient has had a prior anaphylactic reaction to iodine, referral to an allergist is recommended.
Summary of Discussions
Areas of General Agreement by Committee Members
Povidone-iodine (5-10%) should be the last agent applied to the intended injection site before injection. If a gel anesthetic is used, povidone-iodine should be applied both before and after application of gel, as retained gel may prevent povidone-iodine from contacting the conjunctival surface of the injection site.
Pre-, peri-, or postinjection topical antibiotics are unnecessary.
There is no evidence to support the routine use of a sterile drape.
Avoid contamination of the needle and injection site by the eyelashes or the eyelid margins.
Avoid extensive massage of the eyelids either pre- or postinjection (to avoid meibomian gland expression).
Use adequate anesthetic for a given patient (topical drops, gel, and/or subconjunctival injection).
Use of sterile or nonsterile gloves as consistent with modern office practice, combined with strong agreement regarding the need for handwashing before and after patient contact.
Either surgical masks should be used or both the patient and providers should minimize speaking during the injection preparation and procedure to limit aerosolized droplets containing oral contaminants from the patient and/or provider.
Monitor IOP both pre- and postinjection.
Routine anterior chamber paracentesis is not recommended.
___________________________
Areas With No Clear Consensus by Committee Members
Need for povidone-iodine application to the eyelids, including the eyelashes and eyelid margins. All agreed that when povidone-iodine is applied to the eyelashes and eyelid margins, eyelid scrubbing or eyelid pressure adequate to express material from the meibomian glands should be avoided.
Use of a speculum (some prevent contact between the needle/injection site and the eyelashes and eyelids with manual lid retraction).
Need for pupillary dilation and postinjection dilated examination of the posterior segment (although some viewed the return of formed vision as sufficient, others routinely dilate the pupil and examine the posterior segment after injection).
Use of povidone-iodine flush (most preferred drops only and saw no benefit to allowing povidone-iodine to dry before injection).
___________________________
Adapted with permission from Retina: Avery RL et al. Retina. 2014;34(12):S1-S18.
|
Fine Points
Fortunately, the issues lacking consensus were relatively minor and often come down to personal preference (see “Areas With No Clear Consensus by Committee Members”). Dr. Flynn, for example, puts povidone-iodine over the eyelid and eyelash margins as well as the conjunctiva, but some people put it only on the conjunctival surface of eye. Currently, there is not enough evidence to say which approach is better.
Some of the finer points allow for individual tailoring of treatment. “We tried to take a holistic approach to these guidelines, looking at the whole patient and his/her individual characteristics,” said Dr. Haller. An example is the use of a lid speculum versus manual lid retraction. The goal is good exposure with absolutely no contact between the lids and any portion of the needle or injection site prior to injection. Both the individual characteristics of the eye and the preferences of the physician and patient may determine whether the speculum is helpful, explained Dr. Haller.
“A very deep-set eye with blepharospasm might have a safer injection with a speculum, as opposed to a prominent eye with a weaker squeeze reflex that might do equally well with manual retraction. Some patients experience the speculum as the most uncomfortable part of the procedure, and if they strongly prefer manual lid retraction, that preference should be taken into consideration,” she said. Several of Dr. Haller’s patients have gone into spasm because of the speculum, and they have told her how much more they like it when she uses manual lid retraction. Conversely, some of her patients feel safer with the speculum. Dr. Haller reports that being on the panel taught her to include the lid speculum as part of her discussion with patients, giving them a chance to express their preference about its use.
Take-Away
It can take a long time for evidence-based information to percolate out to the ophthalmic community, according to Dr. Cunningham. In fact, Dr. Haller said, “Just the other day I talked to a recently retired retina specialist who did an anterior chamber tap with every single injection!” The guidelines specifically state that routine anterior chamber paracentesis is not recommended.2
“I tell my patients that IVT injections are among the safest procedures in all of medicine, but this safety requires adherence to an evidence-based protocol in which every step plays an important role,” said Dr. Williams. The protocol has evolved and will continue to do so, and practitioners need to stay current.
___________________________
1 Aiello LP et al. Retina. 2004;24(5 suppl):S3-S19.
2 Avery RL et al. Retina. 2014;34(12):S1-S18. http://journals.lww.com/retinajournal/ Abstract/2014/12001/intravitreal_injection_technique_and_monitoring_.1.aspx.
___________________________
Emmett T. Cunningham Jr., MD, PhD, MPH, is director of the uveitis service at California Pacific Medical Center in San Francisco; adjunct clinical professor of ophthalmology at Stanford University School of Medicine in Stanford, Calif.; research associate, the Francis I. Proctor Foundation, UCSF School of Medicine, San Francisco; and a partner at West Coast Retina Medical Group, San Francisco. Financial disclosure: No related interests.
Harry W. Flynn Jr., MD, is J. Donald M. Gass Chair of Ophthalmology and professor of ophthalmology at Bascom Palmer Eye Institute at the University of Miami Miller School of Medicine in Miami. Financial disclosure: No related interests.
Julia A. Haller, MD, is Ophthalmologist-in-Chief of the Wills Eye Hospital and professor and chair of ophthalmology at Thomas Jefferson University in Philadelphia. Financial disclosure: Consults for Allergan, Genentech, and Regeneron.
George A. Williams, MD, is chair of ophthalmology, director of the Beaumont Eye Institute and vice chief of surgical services for Academic Affairs at William Beaumont Hospital in Royal Oak, Mich. He is professor and chair of ophthalmology at Oakland University William Beaumont School of Medicine. Financial disclosure: No related interests.
Retina Coding
The Academy’s new 2015 Complete Guide to Retina Coding (#0120374) provides the information a retina practice needs to maximize reimbursements. Chapters include: Fundamentals of ICD-9-CM and ICD-10-CM Codes, Retinal Testing Services, Surgery Coding Overview, Costly Coding Errors, Retina Specific Audits and more.
To order, visit www.aao.org/store.
|