Uveitis and Blau Syndrome: Preliminary Findings
By Lynda Seminara
Selected By: Richard K. Parrish II, MD
Journal Highlights
American Journal of Ophthalmology, December 2017
Download PDF
Blau syndrome, a rare autoinflammatory disease that can be debilitating, usually presents as a triad of uveitis, arthritis, and dermatitis. Sarens et al. are studying the course of Blau syndrome in a prospective multicenter interventional case series. Preliminary findings of their international 5-year study showed that many patients experience severe ocular morbidity despite continuous immunomodulatory therapy.
Preliminary findings were reported for 49 patients (75 eyes), each with follow-up for 1-3 years. Ophthalmic data were obtained at baseline and annual visits.
The median age at onset of Blau syndrome was 60 months, and the duration of eye disease at baseline was 145 months. In addition, 38 patients (78%) had uveitis at baseline, with 37 of the 38 experiencing bilateral involvement. Eighteen of 66 eyes (the number for which information was available) had moderate or severe visual impairment at baseline, and panuveitis was found in 38 of the 75 eyes (51%). The most common signs of optic nerve involvement were optic disc pallor (9 eyes; 12%) and peripapillary nodules (9 eyes; 12%). Of the 49 patients, 31 (63%) manifested all 3 classic features of Blau syndrome.
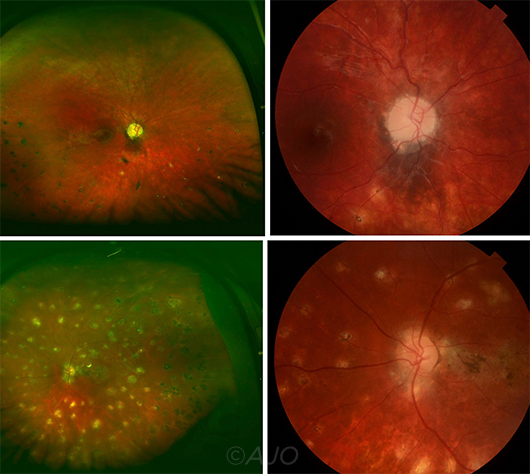 |
BLAU SYNDROME UVEITIS. Widefield fundus (left set) and optic disc fundus (right set) images from 2 patients with Blau syndrome uveitis. In the first patient, chorioretinal scars are evident in the inferior retina (top left photograph), and the pale optic disc has a nodular border (top right). Images from the second patient show multiple chorioretinal scars (bottom left) as well as peripapillary nodules around the border of the optic disc and a macular scar (bottom right).
|
Active anterior chamber inflammation was observed in 30 of the 75 eyes (40%). Panuveitis was associated with longer duration of disease. At baseline, 56 of all eyes (75%) were on topical corticosteroids. Twenty-six patients received a combination of systemic corticosteroids and immunomodulatory therapy. Despite prolonged treatment in all patients, there was no significant decrease of inflammatory activity from baseline to the yearly exams; at year 3, active inflammation was evident in 11 of 18 eyes (61%).
These findings emphasize the need for frequent ophthalmologic surveillance and effective treatments in affected patients, the authors said. Greater understanding of the downstream effects of NOD2 mutations may be instrumental in the development of targeted therapies. (Also see related commentary in the same issue by Gary N. Holland, MD.)
The original article can be found here.