By Arthur Stone, Contributing Writer, interviewing Nicole R. Fram, MD, Soosan Jacob, MS, FRCS, DNB, Gregory S.H. Ogawa, MD, and Joanne F. Shen, MD
Download PDF
When zonular support is lacking, which surgical approach is best for IOL implantation? The Academy has updated its Ophthalmic Technology Assessment (OTA) on the topic, to address evolving surgical practices and assess their impact on outcomes.1
The Need for Guidance
“Understanding how to address issues with intraoperative zonulopathy or IOL fixation in the absence of capsular support is essential,” said Nicole R. Fram, MD, who practices in Los Angeles. “These techniques require surgical planning and comfort with specialized surgical equipment and anterior vitrectomy techniques.”
The first version of this OTA was published in 2003. Since then, surgeons have developed new techniques, including intrascleral haptic fixation (ISHF) of three-piece posterior chamber IOLs (PCIOLs). They’ve also experimented with different suturing materials. Of note: At present, only anterior chamber IOLs (ACIOLs) are FDA approved for implantation in the setting of aphakia without zonular support.
 |
|
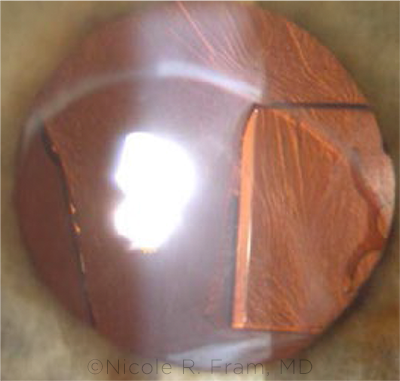
MULTIPLE CHALLENGES. This patient’s problems included amputated IOL fragments from a previous surgery. Dr. Fram’s solution included using the instrascleral haptic fixation technique.
|
OTA Highlights
For the OTA, Joanne F. Shen, MD, and her colleagues assessed 734 studies published in English. Of these, 45 were determined to be relevant.
Fixation techniques. The committee evaluated eight different types of IOL fixation techniques with at least six months of follow-up: 1) ACIOLs; 2,3) iris-claw and retropupillary iris-claw IOLs; 4,5) 10-0 polypropylene (Prolene) iris- and scleral-sutured PCIOLs; 6) 8-0 Prolene scleral-sutured PCIOLs; 7) scleral-sutured PCIOLs using CV-8 polytetrafluoroethylene (Gore-Tex); and 8) the ISHF method.
If the iris is intact. In this instance, surgeons can use any one of the eight different fixation techniques. However, as the committee noted, iris-claw IOLs are not available in the United States.
If the iris is not intact. For these cases, surgeons may choose from among the scleral fixation approaches. The OTA noted that there may be advantages to using 8-0 Prolene, 9-0 Prolene, or Gore-Tex sutures instead of 10-0 Prolene suture material, given ongoing concerns regarding the potential degradation of 10-0 Prolene.
Visual outcomes. According to the studies reviewed, all fixation approaches appear to produce improvements in postoperative mean best-corrected visual acuity (BCVA), ranging from 20/35 to 20/76. The best BCVA outcomes were noted in the ISHF group; the worst were experienced by those in the Gore-Tex-sutured group.
Complications. Overall, complications of cystoid macular edema (CME) were greater with 10-0 Prolene iris-sutured and 8-0 Prolene scleral-sutured PCIOLs. Chronic glaucoma was more prevalent among those who received 8-0 Prolene scleral-sutured PCIOLs. Although retinal detachment was infrequent overall, it was twice as common in both iris- and scleral-sutured PCIOLs using Prolene as in nonsutured approaches.
“Although this OTA relied heavily on retrospective studies, there are both expected and surprising trends,” said Dr. Shen, at the Mayo Clinic in Phoenix. “ACIOLs had less associated CME, but these anterior segments would also be expected to be more intact. Iris-sutured PCIOLs had significant CME trends.”
Need for further study. “I look forward to FDA trials of posterior iris-clipped IOLs, as the retrospective studies conducted outside of the United States had favorable complication profiles,” Dr. Shen said. The OTA highlighted other areas in need of research, including:
- longer follow-up of 8-0 and 9-0 Prolene sutures and formal evaluation of Gore-Tex suture performance;
- long-term integrity of haptics placed within the sclera; and
- reporting on the duration of surgery and timing of pars plana vitrectomy and the sequence of previous surgeries.
Which Approach Is Best?
In its conclusion, the OTA states, “The evidence reviewed shows no superiority of any single IOL implantation technique in the absence of zonular support.” As each approach carries its own risk of post-op complications, “Surgeons should consider matching the best techniques for their skill sets, OR setup, and patient characteristics,” Dr. Shen said. In addition, she noted, patients should be fully briefed on all potential long-term risks.
“Whichever surgical technique one chooses, it is important to do it well following safe practices,” said Soosan Jacob, MS, FRCS, DNB, at Dr. Agarwal’s Refractive and Cornea Foundation in Chennai, India. “Knowledge of complications and when to avoid specific techniques is important.”
Dr. Jacob added that it’s also critical to be cognizant of the fact that “many of these eyes have had prior complicated surgery or will have more complex maneuvers performed during the planned surgery than a routine phacoemulsification. Extra care is therefore a must, and pre- and postoperative evaluation of the retina, together with proper intraoperative handling of the vitreous, is crucial. Appropriate vitreous management plays an important role in the outcome of any case of IOL implantation in the absence of zonular support.”
Sample Cases
The following cases offer an overview of the challenges patients can present:
Fifteen years after trauma. Dr. Shen offered the case of an 80-year-old man who had been struck by a tennis ball in his right eye 15 years before she saw him.
A year before he consulted her, the patient had undergone complicated cataract surgery by another surgeon, which led to retained lens fragments and placement of an ACIOL, she said. “His ACIOL was noted to move with blinking and eye movements, and he later developed pseudophakic bullous keratopathy and 20/400 vision. He was also noted to have scleral thinning diffusely and superior sectoral iris loss.”
Dr. Shen performed an IOL exchange, placing a CZ70BD IOL (Alcon) with Gore-Tex sutures at 1:30 and 7:30 clock hours. She also performed a superior pupilloplasty with 10-0 Prolene sutures. “After one month, I performed a Descemet stripping endothelial keratoplasty (DSEK) in the same eye.”
One year later, the patient’s corrected vision was 20/40, limited by a mild epiretinal membrane.
Twenty years after PRK. A 66-year-old woman consulted Gregory S.H. Ogawa, MD, complaining of two weeks of blurred vision in her right eye.
“She had undergone PRK in both eyes more than 20 years previously,” said Dr. Ogawa, at Eye Associates of New Mexico in Albuquerque. Eleven years after her PRK surgeries, she underwent uncomplicated cataract surgery with astigmatic keratotomies and placement of silicone PCIOLs in both eyes. Then, 16 years after her PRK (roughly four years before Dr. Ogawa saw her), she underwent macular hole surgery in her right eye.
“Her BCVA was 20/200 in the right eye. The slit-lamp exam showed pseudoexfoliative deposits on the pupil and an inferiorly subluxated PCIOL–bag complex in that eye,” Dr. Ogawa said. He performed an IOL exchange, using a retrobulbar block and suturing the PCIOL to the sclera with Gore-Tex sutures. “One week postoperatively, her uncorrected VA was 20/40. One year later, her BCVA was 20/25,” Dr. Ogawa said (see also www.youtube.com/watch?v=feNAsol6MGE&t=19s).
One complication after another. One of Dr. Fram’s patients, a 72-year-old woman, had initially undergone uncomplicated cataract surgery with a Crystalens PCIOL (Bausch + Lomb) placed in the capsular bag. “Unfortunately, she complained of significant glare and was unhappy with the IOL aim,” Dr. Fram said.
Another surgeon had attempted to remove the Crystalens two years before Dr. Fram saw the patient and had encountered fibrosed haptics and zonulopathy. “The Crystalens is the most challenging IOL to remove from the capsular bag,” Dr. Fram noted. “It is typical that the haptics become fibrosed, and they are often amputated and left in the bag.”
The other surgeon removed the optic and left the amputated haptics in the capsule. An acrylic three-piece IOL was placed in the posterior chamber. “Unfortunately, this IOL was tilted, with half of the IOL left in the capsular bag and the remainder in the sulcus.”
The patient developed CME, uncontrolled intraocular pressure, uveitis-glaucoma-hyphema (UGH) syndrome, and some endothelial failure. “The amputated Crystalens haptics were bisecting the visual axis, causing severe glare and starbursts. The patient was in need of visual rehabilitation; leaving the IOL fragments and malpositioned secondary IOL would result in progressive visual damage,” Dr. Fram noted.
Given the multiple challenges posed by this case, “I decided to remove the sulcus IOL and all IOL remnants, perform a pars plana–assisted anterior vitrectomy, and place a secondary IOL using intrascleral haptic fixation—e.g., the Yamane technique,” Dr. Fram said.
A large subluxation. Dr. Jacob treated a patient with a large subluxation (more than two quadrants) of progressive etiology.
“Since it was large and progressive, I considered three options, two of which are techniques I have described—the first being two-sided scleral fixation of the capsular bag using the glued capsular/paper clip capsule stabilizer (device designed by me, manufactured by Morcher) and second, a supracapsular glued IOL technique that combines the advantages of retaining an intact posterior capsule while providing the stable long-term fixation of glued (ISHF) IOL. The third option was a conventionally glued IOL,” Dr. Jacob said.
“Because the subluxation was significant and I did not have the paper clip capsule stabilizers available at the time, I chose to perform the conventional glued IOL procedure,” she said. “I extracted the lens, did a cortical cleanup, and then proceeded with a glued IOL, using the handshake technique. The patient did well postoperatively and had a stable, well-centered IOL without tilt. There were no significant postoperative complications.”
Dr. Jacob said that she personally likes the ISHF technique and finds it “to be easily adaptable to a variety of situations while still having a favorable safety profile.” Even so, she pointed out, “prospective randomized clinical trial data are still needed to reach definitive conclusions” with regard to its use.
__________________________
1 Shen JF et al. Ophthalmology. 2020;127(9):1234-1258.
__________________________
Dr. Fram is at Advanced Vision Care in Los Angeles. Relevant financial disclosures: None.
Dr. Jacob is director and chief of Dr. Agarwal’s Refractive and Cornea Foundation and senior consultant for the Cataract and Glaucoma Services at Dr. Agarwal’s Group of Eye Hospitals, both in Chennai, India. Relevant financial disclosures: Patent pending for glued capsular hooks and variants.
Dr. Ogawa is at Eye Associates of New Mexico in Albuquerque. Relevant financial disclosures: None.
Dr. Shen is at the Mayo Clinic in Phoenix. Relevant financial disclosures: Ocugen: S.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Fram Alcon: C,L; Allergan: C; Bausch + Lomb: L; CorneaGen: C,O; Dompé: C,L; Johnson & Johnson: C,L; Novartis: L; Ocular Science: C,O; Sun Pharmaceutical: L.
Dr. Jacob Patent pending for glued capsular hooks and variants.
Dr. Ogawa None.
Dr. Shen NEI: S; Ocugen: S.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|