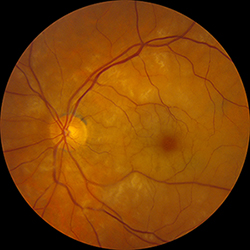
Acute central retinal artery occlusion (CRAO) presenting in the absence of a retinal embolus on exam. This patient was ultimately diagnosed with vasculitic CRAO secondary to biopsy-confirmed giant cell arteritis (GCA).
As ophthalmologists, we sometimes focus too intently on the eye and miss opportunities to use our expertise to prevent systemic disease. A prime example is the diagnosis and management of retinal transient ischemic attacks and retinal artery occlusions.
Most retinal arterial occlusions are thromboembolic in etiology, but many clinicians falsely consider retinal ischemia to be different and less serious than brain ischemia. However, a growing preponderance of research shows that retinal stroke and TIAs carry serious neurologic and cardiovascular implications spanning far beyond vision loss.
Here are seven clinical pearls to help you better manage retinal ischemia.
1. Know the Definition of Retinal TIA
The American Heart Association defines TIA as a “transient episode of neurologic dysfunction caused by focal brain, spinal cord or retinal ischemia without acute infarction.”
Current research shows that retinal TIAs carry the same risk for future stroke and cardiovascular disease as central nervous system TIAs. Importantly, patients who suffer from retinal TIAs do not show evidence of ischemia and will have normal fundus exams.
To diagnose retinal TIA, you must therefore listen carefully to the history of vision loss provided by the patient.
2. Know the Symptoms of Retinal TIAs
A retinal TIA is typically described as a monocular “blacking-out” episode that normally lasts between three and 20 minutes. Many cases that last longer also exhibit evidence of infarction (retinal thickening and pallor) and should be termed a “retinal artery occlusion” or “retinal stroke.”
Retinal TIAs less commonly include positive visual phenomena (e.g., flashing lights, kaleidoscope/prism effect, etc., which suggest migraine visual aura. Visual auras may precede, follow, occur simultaneously with or occur in the absence of migraine headache. Migraine visual auras usually last between five and 60 minutes and afflict patients with a history of chronic migraine disorder. It also normally presents in both eyes (in the same homonymous hemifield) and evolves during the episode (e.g., expands over the field with time).
Retinal migraines are perceived as monocular auras and often exhibit positive visual phenomena. They may be stereotypically recurrent and can be more difficult to distinguish from retinal ischemia based on history alone. Transient visual obscurations are often position- or Valsalva-related visual blurring or “graying-out” episodes that last a few seconds. They are associated with papilledema.
3. Evaluate Patients with Retinal Ischemia Promptly
The risk of clinical CNS stroke after any TIA (retinal or CNS) is 3 percent to 10 percent within two days and 9 percent to 17 percent within 90 days. Retinal TIAs carry the same risk for subsequent stroke as brain TIAs. In fact, one recent study found that 18 percent of patients with a retinal TIA and 33 percent of patients with a retinal artery occlusion exhibited acute brain ischemia on MRI at the time.
Bearing this in mind, an appropriate provider should evaluate patients diagnosed with acute retinal TIA or retinal stroke for CNS ischemia. Regional resources vary. If you can’t do an expedited outpatient work-up within 24 hours, the best alternative may be to send patients to the emergency department or stroke- evaluation unit, if available.
4. Work Up Retinal Ischemia Like Brain Ischemia
Include the following minimum workup in your evaluation:
- MRI brain: stroke protocol with diffusion weighted imaging;
- Noninvasive neck imaging (magnetic resonance angiography, computed tomography angiography or carotid ultrasound);
- Electrocardiography.
5. Note the Reason and Urgency in Any Referrals
Not all providers are attuned to the latest research regarding retinal ischemia and CNS stroke. If you need to refer the patient to an emergency department, you should consider sending a brief note stating that the patient requires rapid evaluation by a neurologist and an urgent stroke workup. Include the aforementioned studies in the note.
6. Treat the Eye and the Brain
Although many treatments for acute retinal stroke have been attempted, evidence-based medicine has failed to prove a highly effective and safe way to restore perfusion and vision to the eye. Relatively safe first-line treatments include intermittent ocular massage and IOP-lowering agents, both of which you should implement quickly when a patient with acute vision loss presents. Other treatments carry risks and can be logistically challenging. In addition, limited evidence supports their efficacy. These treatments include anterior chamber paracentesis, hyperbaric oxygen, ophthalmic artery catheterization with tPA infusion and intravenous tPA administration.
Start treatment immediately for any acute retinal TIA or retinal stroke patient without a contraindication to antiplatelet therapy (e.g., aspirin, 325 mg daily). If you are concerned about starting antiplatelet therapy, consider calling the patient’s primary care physician for guidance. Following acute work-up and management, the patient’s primary care physician and neurologist should also address additional modifications to reduce stroke risk (e.g., statin therapy, blood pressure control, etc.).
7. Don’t Miss a Giant Diagnosis
Not all retinal strokes are thromboembolic in etiology. Giant cell arteritis causes approximately 1 percent to 2 percent of central retinal artery occlusions. Furthermore, GCA can present with retinal TIA or retinal stroke (CRAO, branched retinal artery occlusion or cilioretinal artery occlusion).
You should screen for GCA with any patient over the age of 50 who has signs or symptoms of retinal ischemia and does not show an embolic etiology on examination. Make sure to ask the appropriate GCA screening questions. And depending on your degree of clinical concern, consider checking erythrocyte sedimentation rate, C-reactive protein and complete blood count with platelets prior to temporal artery biopsy.
References
1 Easton JD et al. Stroke. 2009;40(6):2276-2293.
2 Helenius J et al. Ann Neurol. 2012;72(2):286-293.
3 Go AS et al. and American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2013;127(1):143-152.
* * *
 About the author: Collin McClelland, MD, is a neuro-ophthalmologist and assistant professor of ophthalmology and visual neurosciences at the University of Minnesota. Dr. McClelland is a member of the Young Neuro-Ophthalmology Committee of the North American Neuro-Ophthalmology Society. He has won several teaching awards and is a prolific contributor to the OphthoQuestions.com board review website.
About the author: Collin McClelland, MD, is a neuro-ophthalmologist and assistant professor of ophthalmology and visual neurosciences at the University of Minnesota. Dr. McClelland is a member of the Young Neuro-Ophthalmology Committee of the North American Neuro-Ophthalmology Society. He has won several teaching awards and is a prolific contributor to the OphthoQuestions.com board review website.