The glued IOL technique is a relatively new method for fixing a posterior chamber IOL in an eye without a capsule. It was first described by me and my colleagues in December 2007. The current article provides details about how the procedure is performed. Video demonstrating this procedure is also available on the ONE Network.
The technique
The glued IOL technique consists of making two partial-thickness scleral flaps exactly 180 degrees apart (Figure 1A) followed by a sclerotomy with a 20/22-gauge needle 1 mm from the limbus (Figure 1B). The anterior chamber can be maintained with a 23-gauge sutureless trocar and cannula or an anterior chamber maintainer. Viscoelastics can be used if one prefers.
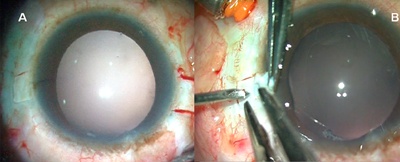
Figure 1A. Two partial scleral flaps 180 degrees apart. 1B. Sclerotomy with a 20/22-gauge needle.
An anterior vitrectomy or mid-vitrectomy is performed to clear the vitreous. A clear corneal incision is made for implanting an IOL. A foldable three-piece lens from Abbott Medical Optics Inc. (Santa Ana, Calif.), Alcon Laboratories, Inc. (Fort Worth, Texas) or Bausch & Lomb (Rochester, N.Y.) with slightly firm haptics or a nonfoldable IOL is appropriate.
Next, a 23/25 gauge MicroSurgical Technology (Redmond, Wash.) forceps or any end-opening micro rhexis forceps is passed through the sclerotomy to grasp the tip of the IOL haptic, which is then brought out through the sclerotomy (Figures 2 A, B and C).
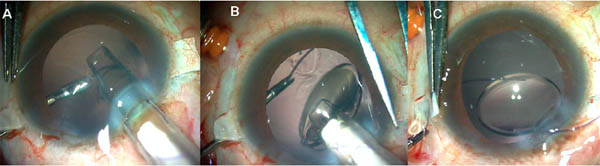
Figure 2A. The injector is passed through the corneal incision. 2B. The IOL haptic is held with the forceps while the lens is injected. 2C. The IOL haptic is externalized under the scleral flap.
The trailing haptic is flexed into the anterior chamber and the tip grabbed once again with the end-opening forceps and externalized (Figures 3A, B and C).
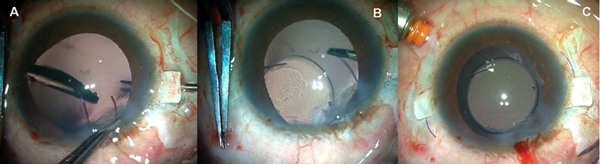
Figure 3A. The IOL haptic is flexed into the anterior chamber. 3B. The haptic end is grasped with another forceps. 3C. Both haptic ends are externalized under the flaps.
With a 26-gauge needle, tunnels are made at the edge of the flap into which the haptics are then tucked for additional stability (Figures 4A and B).
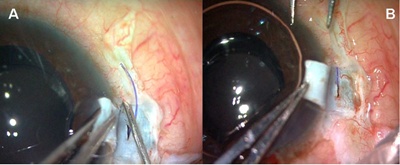
Figure 4A. A scleral tunnel is made with a 26-gauge needle. 4B. The haptic end is tucked into the tunnel.
The scleral flaps are then glued back into place using biological glue (Figures 5A and B).
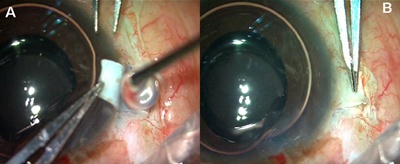
Figure 5A. Fibrin glue is placed below the flap. 5B. The flap apposed with the scleral bed.
While the glued IOL technique is being performed, an assistant must hold the haptics of the IOL once they have been externalized through the sclerotomies. If an assistant is not available, the surgeon can use a silicone tire from a Mackool iris retractor set to prevent the haptic from being pulled into the eye (Figure 6).
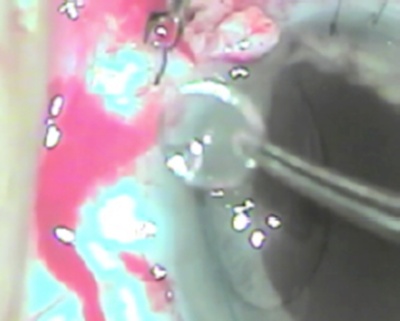
Figure 6. One forceps holds a silicone tire, while another forceps engages the haptic and threads it into the central hole of the tire.
Use of fibrin glue
I use Tisseel fibrin glue (Baxter Healthcare Corp., Deerfield, Ill.), which is a quick-acting surgical glue sealant derived from human plasma. It has both hemostatic and adhesive properties. It is composed of two separate solutions: fibrinogen or factor 1 and thrombin. Fibrinogen is activated by thrombin to form fibrin monomers. Activated factor XIII polymerizes fibrin monomers to form a stable fibrin clot, thus mimicking the last stages of the clotting cascade. Aprotinin delays fibrinolysis, the process that leads to the breakdown of blood clots. Tisseel is virus-inactivated and is checked for viral antigen and antibodies with polymerase chain reaction. This makes the chances of infection with its use very low.
IOL haptic stability
Since the flaps are manually created, the rough apposing surfaces of the flap and bed heal rapidly and firmly around the haptic, helped early on by the fibrin glue. The major uncertainty is the stability of the fibrin matrix in vivo. Animal studies have shown that the fibrin glue is still present at four to six weeks. Because postoperative fibrosis starts early, the flaps become stuck secondary to fibrosis prior to full degradation of the glue. The ensuing fibrosis acts like a firm scaffold around the haptic, preventing movement along the long axis.
In order to make the IOL more stable, my colleagues and I have started tucking the haptic tip into the scleral wall through a tunnel. This prevents all movement of the haptic along the transverse axis as well. The tissue glue gives it extra stability and also seals the flap down. Externalization of the greater part of the haptic along its curvature stabilizes the axial positioning of the IOL, preventing any IOL tilt.
IOL memory and tilt
Another concern is the change in the properties of the biomaterial that can occur when the haptic is placed in a stretched position in vivo. Loop rigidity (the resistance of the haptic to external forces that act to bend the loops centrally) and loop memory (the ability of the loops to re-expand laterally to their original size and configuration) contribute to the ability of IOL loops to maintain their original symmetrical configuration. Loop rigidity is seen as the centrifugal force vector of the haptic, which resists compression by the capsular bag in an in-the-bag IOL. Loop memory is responsible for the gently stretched haptic following the globe curvature and for creating a centripetal force which, along with the intralamellar scleral tuck, stabilizes the IOL. These two concepts can be understood by compressing or stretching the haptic in vitro.
Another issue to be addressed is IOL centration and tilt. We measured IOL centration with a dilated pupil on retroillumination slit-lamp photographs processed with Matlab version 7.1. IOL tilt was assessed using anterior segment OCT and by noting the change between the third and fourth Purkinje images. The topographic (using Orbscan) and manifest refraction were also found to be constant in all eyes during the entire postoperative period, which was suggestive of minimal new IOL-induced astigmatism.
In the study we conducted of our own patients, centration of the IOL was good, with no detection of IOL tilt. Postoperative follow-up anterior segment OCT also showed perfect scleral flap adhesion as early as day one and continuing through the first week and first month.
We conducted a two-week-long in vitro study in which the IOL with haptics on stretch was continuously immersed in a water bath with balanced salt solution that was maintained at body temperature. There were no differences between pre- and post-immersion measurements or after two weeks of drying.
Pediatric glued IOLs
Sutured scleral-fixated IOLs in pediatric eyes have been known to be associated with problems. However, we have had very good results with pediatric glued IOLs in multiple complicated cases, including homocystinuria with subluxation, aniridia with a cataractous subluxated lens and Weill-Marchesani syndrome with microspherophakia and glaucoma.
References
- Agarwal A, Kumar DA, Jacob S, Baid C, Agarwal A, Srinivasan S. Fibrin glue-assisted sutureless posterior chamber intraocular lens implantation in eyes with deficient posterior capsules. J Cataract Refract Surg. 2008;34(9):1433-1438.
- Kumar DA, Agarwal A, Prakash G, Jacob S, Saravanan Y,B Agarwal A. Glued posterior chamber IOL in eyes with deficient capsular support: a retrospective analysis of 1-year post-operative outcomes. Eye. 2010;24(7):1143-1148.
- Agarwal A, Kumar DA, Jacob S, Prakash G, Agarwal A. Fibrin glue-assisted sutureless scleral fixation [author reply to letter by Falavarjani KG, Modarres M, Foroutan A, Bakhtiari P]. J Cataract Refract Surg. 2009;35(5):795-796.
- Kumar DA, Agarwal A, Jacob S, Prakash G, Agarwal A, Sivagnanam S. Repositioning of the dislocated intraocular lens with sutureless 20-gauge vitrectomy. Retina. 2010;30(4):682-687.
- Kumar DA, Agarwal A, Jacob S, Prakash G, Agarwal A. Use of 23-gauge or 25-gauge trocar cannula for globe maintenance in glued intraocular lens surgery. J Cataract Refract Surg. 2010;36(4):690-691.