By Evan H. Black, MD, FACS, Lawrence Halperin, MD, Christopher J. Rapuano, MD, and Kathryn P. Winkler, MD
Download PDF
A look into the future of cornea, oculoplastics, and retina.
As 2017 wraps up, EyeNet approached several of its editorial board members, asking them to identify a news item or trend from this year that could significantly shape their subspecialty or all of ophthalmology over the near term, say, the next 5 years. These experts—Christopher J. Rapuano, MD, cornea; Evan H. Black, MD, oculoplastics, and his colleague Kathryn P. Winkler, MD; and Lawrence S. Halperin, MD, retina—approached the project with their own style, perspective, and thought process. The write-ups are as personal and unique as each of the authors and will help readers move into 2018 and beyond.
Dr. Rapuano on Cornea
Corneal Endothelial Cell Regeneration Therapy: The Way of the Future?
Endothelial cell dysfunction is a leading cause of corneal transplantation around the world. In fact, corneal edema due to endothelial cell failure has been the No. 1 indication for transplantation in the United States for many years.1 Penetrating keratoplasty (PK) was the procedure of choice for these eyes until the early 2000s, when endothelial keratoplasty (EK) was introduced. EK yields faster visual recovery, less change in refractive error, less irregular astigmatism, and a much smaller wound, resulting in lower risk of a wound dehiscence. EK overtook PK as the primary corneal transplant procedure performed in the United States for all indications in 2012.1
However, EK isn’t perfect. It requires highly trained surgeons, an operating room infrastructure, and follow-up by appropriately trained physicians to manage complications such as graft rejection and glaucoma. It also requires adequate donor material and a functioning eye banking system. All of these components are rather costly, limiting the use of EK worldwide.
These limitations to corneal transplant surgery have led to investigations into alternative treatments for endothelial cell dysfunction, including simply removing the central unhealthy endothelial cells, thus allowing peripheral endothelial cells to migrate and cover the central cornea; using medications to stimulate healthy endothelial cell proliferation; and transplanting donor endothelial cells or, even better, the patient’s own endothelial cells (potentially derived from their own stem cells). While none of these therapies is “ready for prime time” yet, I believe over the next 5 years, we will be treating corneal endothelial cell dysfunction differently from today.
Primary Descemetorhexis
The first time I heard of the concept of simply removing the central unhealthy endothelium and Descemet membrane was when Brad Randleman gave a case presentation regarding a young woman with Fuchs dystrophy/posterior polymorphous corneal dystrophy who underwent Descemet stripping EK (DSEK). The DSEK graft completely detached, but the cornea still cleared, so the detached graft was removed. He later published this case.2 In the article, he included follow-up: The first eye remained clear, and he subsequently performed primary descemetorhexis surgery in the fellow eye, which also cleared, with central endothelial cell repopulation in both eyes. At the time, the hypothesis was that the peripheral endothelial cells were simply migrating centrally to cover the denuded area, as corneal endothelial cells were thought not to proliferate.
There have been several publications on this technique with mixed results.
Poor results. Arbelaez et al.3 had poor results in 3 eyes undergoing 6.0-6.5 mm diameter descemetorhexis for Fuchs dystrophy. Additionally, they found poor adherence of subsequent Descemet membrane EK (DMEK) in the eye that underwent that procedure. Koenig4 had poor results with a 6-mm diameter descemetorhexis in 2 eyes with Fuchs dystrophy at the time of cataract surgery.
Good results. On the other hand, Borkar et al.5 performed a 4-mm diameter descemetorhexis in 13 eyes of 11 patients with Fuchs dystrophy at the time of cataract surgery. Four corneas cleared by 1 month (termed “fast responder”), 4 additional corneas cleared by 3 months (“responder”), and 2 more corneas cleared by 6 months (“slow responder”). Three corneas did not clear, and all 3 underwent successful DMEK with no need to rebubble.6
Mixed results. Iovieno et al.7 found unpredictable results using a 4-mm central descemetorhexis in 5 patients.
The downside to this approach is that most surgeons believe that the actual number of functioning endothelial cells isn’t increased and, therefore, the total endothelial cell density decreases.
Enhancing Corneal Endothelial Cell Proliferation and/or Function
While human corneal endothelial cells are not thought to multiply naturally after birth, one approach is to use “growth factors” to induce the healthy endothelial cells to proliferate (increasing their absolute number). The most studied of these growth factors are the rho kinase inhibitors. In animal models and small human studies, rho kinase inhibitors have been demonstrated to slow progression of endothelial cell degeneration and also restore normal endothelial cell counts after endothelial cell injury.8-13 Needless to say, uninhibited growth of cells in the anterior chamber has potential side effects, including covering the trabecular meshwork and inducing glaucoma.
Using a combination of surgical removal of unhealthy central corneal endothelial cells and a topical rho kinase inhibitor, ripasudil, to improve corneal endothelial cell function, Moloney et al.14 performed a 4-mm diameter descemetorhexis in 12 eyes of 11 patients with Fuchs dystrophy. Nine of the 12 corneas cleared between 2 and 5 months postoperatively. In the other 3 eyes, topical ripasudil was applied to aid in repopulation, and 2 of these 3 corneas cleared.
Corneal Endothelial Cell Replacement Therapies
While corneal transplantation replaces endothelial cells, there is great interest in both simplifying the technique of transferring these cells and potentially using cells from the patients themselves, thereby avoiding the need for donors. There are several potential sources for endothelial cells, including embryonic stem cells; the patient’s own stem cells, including adult stem cells (progenitor cells) and induced pleuripotent stem cells; and donor corneal endothelial cells expanded in vitro.
Embryonic stem cells have issues with immune rejection and tumorigenicity as well as ethical concerns regarding the use of human embryos. Induced pleuripotent stem cells have great potential, but they may not be as “pleuripotent” as originally hoped. Adult stem cells/progenitor cells for the corneal endothelium have been found in the corneal limbus. While not as pleuripotent as embryonic stem cells or induced pleuripotent stem cells, they may be suitable for endothelial cell transplantation. Adult human skin cells have also been successfully induced to produce “corneal endothelial-like cells.”15
Expanding donor endothelial cells in vitro eliminates the problems of getting stem cells to differentiate into corneal endothelial cells, but it obviously involves getting these cells to proliferate, which has been problematic. One important advantage to this technique is the potential ability to treat multiple patients with a single donor cornea.
No matter which cells are being used to repopulate the corneal endothelium, the optimal delivery method is still up for debate. Cells can be injected and the patient placed in a prone position allowing gravity to pull the cells toward the posterior cornea. Tiny magnetic particles and nanoparticles have also been used to position endothelial cells.16,17 Additionally, a variety of artificial corneal scaffolds have been used, but those techniques involve potentially complicated corneal surgical procedures.
Conclusion
Current corneal transplant techniques, while quite successful, are labor and cost intensive. A variety of evolving therapies have the potential to change the manner in which we treat corneal endothelial abnormalities over the next 5 years.
___________________________
1 Park CY et al. Ophthalmology. 2015;122(12):2432-2442.
2 Shah RD et al. Ophthalmology. 2012;119(2):256-260.
3 Arbelaez JG et al. Cornea. 2014;33(12):1295-1299.
4 Koenig SB. Cornea. 2015;34(9):1149-1151.
5 Borkar DS et al. Cornea. 2016;35(10):1267-1273.
6 Rao R et al. Cornea. 2017;36(7):763-766.
7 Iovieno A et al. Cornea. 2017;36(6):637-641.
8 Bartakova A et al. Curr Ophthalmol Rep. 2014;2(3):81-90.
9 Koizumi N et al. Cornea. 2014;33 Suppl 11:S25-31.
10. Meekins LC et al. Invest Ophthalmol Vis Sci. 2016;57(15):6731-6738.
11 Okumura N et al. Sci Rep. 2016;18(6):26113.
12 Okumura N et al. Invest Ophthalmol Vis Sci. 2016;57(3):1284-1292.
13 Okumura N et al. Curr Pharm Des. 2017;23(4):660-666.
14 Moloney G et al. Cornea. 2017;36(6):642-648.
15 Inagaki E et al. Stem Cells Transl Med. 2017;6(3):788-798.
16 Moysidis SN et al. Nanomedicine. 2015;11(3):499-509.
17 Hsu CC et al. Cell Transplant. 2015;24(10):1915-1930.
Dr. Black and Dr. Winkler on Oculoplastics
Beyond Blepharoplasty: Lifting the Forehead
With the wealth of information available to patients, who have become more educated about aesthetic surgery, the demand for minimally invasive and highly efficacious forehead lifting procedures continues to grow. Newer endoscopic forehead lifting techniques allow for faster recovery, less pain, and fewer risks and complications while providing better, longer-lasting results. Over the coming years, surgeons who perform blepharoplasty will need to be equipped with an understanding of the eyebrow and forehead anatomy and the role these have in the appearance of the aging face.
Often when a patient presents with complaints of drooping upper eyelids or excess upper eyelid skin, he or she has a combination of blepharoptosis and/or dermatochalasis and brow ptosis. Descent of the brow, subbrow fat pad, and forehead contributes to an appearance of fatigue or anger, which interferes with the patient’s nonverbal communication. Patients are mostly unaware of changes in the brow position and attribute their tired appearance to excess upper eyelid skin alone. It is therefore imperative to accurately evaluate and communicate the clinical findings with the patient.
Exam Basics
A patient who presents with complaints of upper eyelid crowding should have a thorough clinical evaluation, with particular consideration of the margin to reflex distance 1 (MRD1), presence of dermatochalasis, and presence and severity of brow ptosis. Upper eyelid skin descending over the lateral lashes (Connell’s sign) is classic for brow ptosis. The presence of deep forehead rhytids can be an indication of prolonged frontalis engagement. The true degree of brow ptosis is best measured by manually fixating the brow while the patient closes his or her eyes, then asking the patient to gently open the eyes.
The positioning of the eyebrow is affected by such factors as brow elevator and depressor muscles, genetics, gravity, skin laxity, surgery, trauma, and the patient’s expressivity. In general, the female eyebrow should lie approximately at or above the superior orbital rim. It should have a curve with the tail of the brow higher than the head of the brow. The male eyebrow should be at the level of the superior orbital rim with a less arched configuration. Presence of supraplaced brow tattoo or makeup is important to note as well.
A patient with complaints consisting mostly of visual disturbance in the superior half of the visual field may only be interested in pursuing a blepharoptosis repair or functional blepharoplasty. Those patients interested in achieving facial rejuvenation with a more aesthetic emphasis should be considered for a forehead lifting procedure alone or with a blepharoptosis repair or blepharoplasty. Providing the patient with a handheld mirror during the examination can help with demonstrating the patient’s anatomy and goals of each surgery. Preoperative photos should be obtained, including flashed and unflashed photos, side views, and three-quarter views.
Surgical Options
There are a number of surgical options for lifting the brow, each with varying indications and advantages.
A direct brow lift is achieved by excising an ellipse of suprabrow tissue. This surgical procedure may be considered when the patient has a visual function disturbance that cannot be corrected through upper eyelid surgery alone due to significant and severe brow ptosis. Given the inevitable scarring caused by this intervention, this procedure should not be considered for patients who want a cosmetic outcome. This procedure may be covered by insurance if there is interference with activities of daily living due to the visual obstruction.
A midforehead lift, similarly, is performed by removing an ellipse of tissue across the entire forehead, placing the incision in a deep forehead rhytid. Again, this procedure should not be considered for a cosmetically motivated patient.
A transblepharoplasty brow lift is performed at the time of upper eyelid surgery. A dissection is carried up through the lid crease incision, and the brow is fixated to the superior lateral orbital rim. This achieves a mild temporal lift. For reimbursement purposes, this procedure may be considered “bundled” with functional upper eyelid surgery.
Cosmetic procedures. The pretrichial, coronal, and endoscopic foreheadplasty procedures are all options for the cosmetic brow lift patient.
A pretrichial lift is best performed in a patient with a high forehead as it will shorten the forehead, thus lowering the hairline. It is performed through an incision just anterior to the hairline with a dissection in the subgaleal plane.
A coronal forehead lift requires an incision at the coronal suture, extending from ear to ear. Similar to a pretrichilal lift, the forehead lift is performed in a subgaleal plane.
The endoscopic forehead lift is a minimally invasive procedure to lift the brow, performed in the avascular subperiosteal plane. In general, this procedure results in minimal to no scarring with lasting results.
Performing the Endoscopic Lift
The endoscopic forehead lift will allow for elevation of the brow and subbrow fat pad, as well as reduction of forehead and glabellar rhytids, and improvement of lateral canthal hooding.
Our endoscopic forehead lift technique places all incisions behind the hairline, without the need to shave the hair. Surgical sites are marked with 2 temporal elliptical incisions in all cases, 2 paracentral incisions for men, and 1 central plus 2 paracentral incisions for women. The supraorbital notch should be identified, and a “safety zone” 2 cm around the notch should be drawn.
After thorough local anesthetic is administered, the temporal ellipses are excised, and a dissection is carried out to the deep temporalis fascia. In this plane the facial nerve is superior to the dissection, which prevents injury. The endoscope is used to visualize the temporal dissection down to the lateral canthal angle. Once the zygomaticoemtemporal (or sentinel) vein is encountered the dissection is complete.
Next, the central and paracentral incisions are created down to the periosteum. A blind subperiosteal dissection is carried out, avoiding the areas of the supraorbital “safety zone” markings. Once complete, the endoscope is again used to visualize the supraorbital bundle to allow for complete dissection around these important structures. The temporal and central dissection pockets are then connected by releasing the conjoint tendon.
Once released, 2 holes are drilled into the outer calvarium just anterior to the hairline in the area of maximal desired arch of the brow. Endotine (MicroAire) anchors are placed in these drill holes and the forehead is fixed to the Endotines. The superficial temporalis fascia is sutured and the skin incisions are closed using staples.
Conclusion
Being familiar with the various brow lifting procedures is important for any surgeon who evaluates and operates on patients who present with upper eyelid crowding or drooping complaints. This familiarity allows the patient and surgeon to discuss and determine the most appropriate surgical intervention for any patient depending on the patient’s goals of surgery. Utilizing modern endoscopic techniques, surgeons and their patients can expect to have excellent, long-lasting results along with faster recovery and fewer complications.
Dr. Halperin on Retina
The Future is Now???
Retina has come a long way in the past 15-25 years. Surgical success rates are improving, complications are diminishing, surgical time is dropping. Gone are the days of watching macular disease steal vision from the ever-increasing number of aging or diabetic patients. The era of anti–vascular endothelial growth factor (VEGF) drugs has created nothing short of a sea change in our treatment. Yet, there is still much to do. Here is a list of what I hope is achievable in the next 5-10 years:
Exudative age-related macular degeneration (AMD). Anti-VEGF treatment is outstanding at preventing vision loss but mediocre at improving lost vision. New treatments for vision improvement have been elusive, but I am still hopeful.
Anti-VEGF drugs reliably reduce or eliminate leakage. However, atrophy and/or fibrosis eventually develop in a high percentage of patients. Future treatments to reduce or eliminate these damaging developments should improve vision outcomes. We thought Fovista would be the first-in-class antifibrotic, but the clinical trial did not deliver.
Dry AMD. A treatment for geographic atrophy (GA) is in development at Genentech and other locations. This treatment, too, may prevent further visual and functional loss, but it will not restore lost vision. Now, marginal data is threatening to derail this first-in-class treatment. Whether restoration of some sight is achievable with stem cell therapy, genetic manipulation, or some other method is still unknown, but researchers are actively investigating. Preventing vision loss in GA would be a major step forward, as this condition seems to be increasingly prevalent.
Injections. As the indications for intravitreal drugs expand, the number of these procedures performed by retina surgeons will grow, even if the long-acting delivery device currently under investigation by Genentech is successfully developed. Preloaded syringes from Genentech have already received positive reviews, and I suspect the concept will spread among manufacturers.
Retinal degenerations and genetics. Teams of molecular biologists spent a decade and $1B sequencing the genome of a single human being not that long ago. Now, this can be done in a short time for a few thousand dollars. Clearly this trend will continue, and as it applies to retinal disease, we will be able to identify all gene-related retinal disease more quickly, accurately, and inexpensively than ever before.
As we find disease-causing genes, the ability to make the leap from gene identification to treatment remains mostly elusive. The biggest development in molecular genetics therapy is CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats), the most efficient and approachable gene-editing system ever invented. Our ability to control this environment will become the central issue in the future of medical discovery and treatment.
Surgical retina. Improvements in vitrectomy, from instrumentation to comfort of surgery to results, have been impressive in the past 15 years. However, there is more to accomplish. Specifically, proliferative vitreoretinopathy (PVR) remains an infrequent but important cause of failure of retinal detachment surgery. Medications such as methotrexate have been suggested but not proven to prevent PVR. This could be a perfect target for CRISPR.
Small-gauge vitrectomy has changed the way we approach retinal diseases as well as the patient experience. Gone are the days of postop pain, eye swollen shut, high intraocular pressure, and very slow vision recovery. A vitrectomy/sclera buckle for retinal detachment can be achieved in under an hour. Almost gone are the days of extensive facedown positioning, as many surgeons realize that it does not contribute to improved outcomes.
Diabetic retinopathy (DR). Laser and anti-VEGF therapy are the mainstays of treatment and have clearly reduced the incidence of severe vision loss. As anti-VEGF injections continue to demonstrate a benefit in prevention and treatment of diabetic macular edema and proliferative DR, retina specialists have reached a very high level of effectiveness.
Early identification remains an important problem. For myriad reasons, many diabetic patients do not come in for eye exams at the early stages of disease. Despite advances ranging from nonmydriatic cameras to artificial intelligence–driven photographic interpretation, diabetics are still required to sit in front of a camera. Until drones can fly around and take photos of retinas without permission, I’m not sure how we are going to move the needle on this common and preventable cause of vision loss.
And, despite incredible advances in anti-VEGF treatment and surgical approaches for DR, a treatment for retinal ischemia remains elusive. I hope the next 10 years brings us a treatment to prevent, or even reverse, capillary closure and retinal ischemia in our patients with diabetes.
Conclusion. Retina is at the forefront of advances in medicine. Technologic improvements in the operating room, scientific improvements in biologics, and genetic understanding of disease have contributed to our ability to help patients maintain, improve, and preserve their sight.
This has all come at a high financial cost, and as the health care system bends under the weight, we as physicians will be challenged to provide cutting-edge care in the most efficient and affordable way. We hope to continue to provide the very best care to every patient.
Meet the Experts
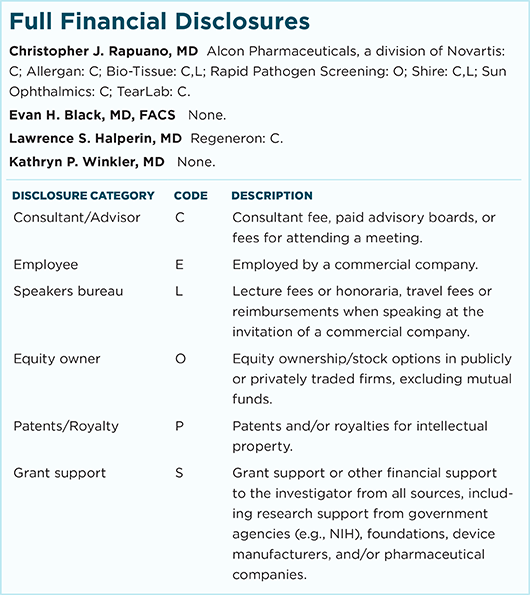
Christopher J. Rapuano, MD Director of the cornea service and co-director of the refractive surgery department at Wills Eye Hospital and professor of ophthalmology at Jefferson Medical College, both in Philadelphia. Relevant financial disclosures: None.
Evan H. Black, MD, FACS Oculoplastic surgeon at Consultants in Ophthalmic & Facial Plastic Surgery in Southfield, Mich., professor of ophthalmology at Oakland University/William Beaumont Hospital School of Medicine in Royal Oak, Mich., and associate professor of ophthalmology at Wayne State University School of Medicine in Detroit. Relevant financial disclosures: None.
Lawrence S. Halperin, MD Retina specialist at the Retina Group of Florida in Boca Raton. Relevant financial disclosures: Regeneron: C.
Kathryn P. Winkler, MD Oculoplastic surgeon at Consultants in Ophthalmic & Facial Plastic Surgery in Southfield, Mich. Relevant financial disclosures: None.
|