By Albert Cheung, Ingrid U. Scott, MD, MPH, Timothy G. Murray, MD, and Carol L. Shields, MD
Edited by Ingrid U. Scott, MD, MPH, and Sharon Fekrat, MD
This article is from February 2012 and may contain outdated material.
Download PDF
Choroidal nevi are benign melanocytic lesions of the posterior uvea. In the United States, their prevalence ranges from 4.6 percent to 7.9 percent in Caucasians.1 By comparison, choroidal melanoma is rare, manifesting in approximately six in 1 million Caucasian individuals.
Metastasis of and death from choroidal melanoma have been shown to correlate with increasing basal diameter and increasing thickness of the lesion. Thus, early detection is important. In addition, making the correct diagnosis of choroidal nevi in a timely fashion protects patients against the visually damaging effects of unnecessary treatment.
Symptoms
Choroidal nevi, which typically are found on routine dilated fundus examination, usually are asymptomatic. However, they can be associated with symptoms such as central and peripheral visual loss secondary to subretinal fluid, cystoid retinal edema or, rarely, choroidal neovascularization.
Choroidal melanoma also tends to be asymptomatic, although it is more likely to be symptomatic than a benign nevus. Symptoms of a choroidal melanoma may include decreased vision, flashes or floaters.
Melanoma Risk Factors
Host risk factors. Significant risk factors for choroidal melanoma include being of Caucasian ethnicity and having light-colored eyes (blue or gray), fair skin and a propensity to burn when exposed to ultraviolet light.2 This susceptibility may be secondary to reduced number of melanocytes (melanin) in the choroid and reduced melanin in the retinal pigment epithelium, which provides this population with less protection from UV light. Lighter skin color also could be indicative of reduced or different melanin.
Other risk factors for choroidal melanoma include nevi and freckles, which are viewed as markers of a predisposing phenotype and environmental UV light exposure. The number of common cutaneous nevi, atypical cutaneous nevi, cutaneous freckles and iris nevi have all been found to be associated with an increased risk of choroidal melanoma.3
Environmental risk factors. These include chronic sunlight exposure and arc welding.4 Studies examining the association between choroidal melanoma and UV light exposure using surrogate markers (such as birth latitude, outdoor leisure activities or occupational sunlight exposure) have been inconsistent. 4
Differentiation and Documentation
Differentiating between benign choroidal nevi and small malignant melanomas can be challenging.
Shared features. Choroidal nevi and choroidal melanoma can show several overlapping features, including tumor size; color, which may be either pigmented or nonpigmented; location; associated dormant features, such as overlying retinal pigment epithelial alterations and drusen; and suspicious features, including subretinal fluid and orange pigment.
Distinguishing features. Choroidal nevi tend to have clearly defined margins and to be flat or slightly elevated, and they remain stable in size. Over time, choroidal nevi display features such as overlying drusen as well as retinal pigment epithelial atrophy, hyperplasia or fibrous metaplasia.
In contrast, choroidal melanomas are more likely to show signs of activity such as relatively indiscrete margins, irregular or oblong configuration, overlying subretinal fluid and orange pigment, and abruptly elevated edges.
Documentation of growth. Most authorities agree that documentation of growth over a relatively short period of time, such as one to two years, is a convincing characteristic of a mitotically active melanoma. However, it is ideal to detect choroidal melanoma before the recognition of growth, as documented growth imparts an almost eightfold greater risk for metastasis.5 On the other hand, slow growth of 0.5 mm over many years or decades may simply reflect the natural progression of a benign choroidal nevus.6
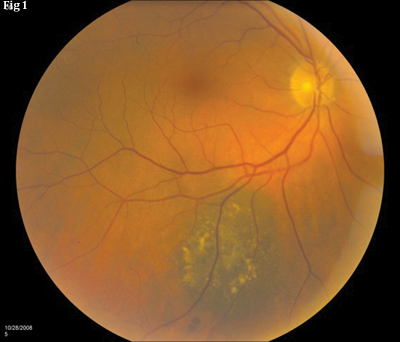
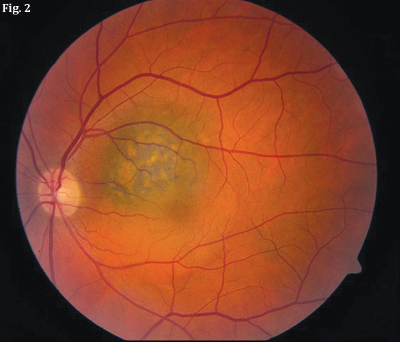 |
| DIFFERENTIATION. (Fig. 1) Choroidal nevus with drusen. (Fig. 2) Choroidal melanoma with orange pigment and subretinal fluid. |
Risk of Malignant Transformation
Choroidal nevi rarely evolve into malignant melanoma; the annual rate of malignant transformation is estimated to be 1 in 8,845.1 The rate of transformation increases with age; it has been estimated that by age 80, the risk for malignant transformation of a choroidal nevus is 0.78 percent.7
Although nevus thickness has been reported to be the most important risk factor for malignant transformation, other factors are also predictive. The mnemonic “To Find Small Ocular Melanoma Using Helpful Hints Daily” (TFSOM-UHHD) has been proposed.6 This stands for thickness greater than 2 mm, subretinal fluid, symptoms, orange pigment present, margin within 3 mm of the optic disc, ultrasonographic hollowness (versus solid/flat), absence of halo and absence of drusen.6 (A halo refers to a pigmented choroidal nevus surrounded by a circular band of depigmentation.)
Low to medium internal reflectivity, often compatible with acoustic hollowness on B-scan echography, has been correlated with choroidal melanoma. Echographically measured thickness of at least 2 mm and a largest basal diameter of at least 7 mm can be helpful in identifying small melanomas. Other echographic features that are suspicious for melanoma include excavation of choroidal tissue, sound attenuation, orbital shadowing and spontaneous vascular pulsations.8 Echographic follow-up for at least 1.5 years may be necessary to differentiate between suspicious nevi and melanomas.
Management of Choroidal Nevi
Management of a choroidal nevus is determined by its risk of transforming into a choroidal melanoma.
By risk factors. From studies using the TFSOM-UHHD risk factors, choroidal melanocytic tumors that displayed none of the risk factors had a 3 percent chance for growth at five years and most likely represented nevi.9 For lesions that display one factor, imparting a 38 percent chance for growth, observation is a reasonable option, especially if the lesion is in a visually important location. Lesions with three or more factors will show growth in more than 50 percent of cases. Such lesions likely represent small choroidal melanomas; and early intervention may be warranted, as they occasionally lead to metastasis.
By lesion size. Some clinicians suggest observation for lesions smaller than 2 mm; lesions larger than 2 mm but smaller than 2.5 mm may be managed based on clinical risk factors, with either close observation or immediate treatment.
Recommended follow-up. Patients with choroidal nevi who show no suspicious features require no treatment. During the first year, they should be monitored twice; subsequently, they should be evaluated annually as long as the nevi remain stable. Although the link between UV light exposure and choroidal melanoma has not been proved, sunglasses could possibly reduce ocular melanoma risk.
Patients who have one or two risk factors for malignant transformation should be monitored every four to six months. Patients with nevi and three or more suspicious features should be evaluated at an experienced center for management alternatives and possible treatment due to their increased risk of developing melanoma.
Conclusion
Differentiating between choroidal nevi and choroidal melanomas can be challenging, but knowing which risk factors are associated with an increased risk of developing melanoma will aid the clinician in making the correct diagnosis in a timely fashion and managing patients appropriately.
___________________________
Mr. Cheung is a medical student and Dr. Scott is professor of ophthalmology and public health sciences; both are at Penn State College of Medicine in Hershey, Pa. Dr. Murray is professor of ophthalmology at Bascom Palmer Eye Institute in Miami. Dr. Shields is professor of ophthalmology and associate director of the ocular oncology service at Wills Eye Institute in Philadelphia.
___________________________
1 Singh AD et al. Ophthalmology. 2005;112(10):1784-1789.
2 Weis E et al. Arch Ophthalmol. 2006;124(1):54-60.
3 Weis E et al. Ophthalmology. 2009;116(3):536-543, e2.
4 Shah CP et al. Ophthalmology. 2005;112(9):1599-1607.
5 Shields CL et al. Ophthalmology. 1995;102(9):1351-1361.
6 Shields CL et al. Arch Ophthalmol. 2009;127(8):981-987.
7 Kivelä T, Eskelin S. Ophthalmology. 2006;113(5):887-888, e1.
8 Pavlin CJ, Foster FS. “Ultrasound Biomicroscopy of the Eye,” in Ultrasound of the Eye and Orbit, 2nd ed. (St. Louis: Mosby; 2002.)
9 Shields CL et al. Arch Ophthalmol. 2000;118(3):360-364.