By Nathalie Pei Yu Chiam, MD, Daniel Shu Wei Ting, MD, PhD, Lee Shu Yen, FRCS(Ed), and Chong Lye Ang, FRCOphth
Edited By: Sharon Fekrat, MD, and Ingrid U. Scott, MD, MPH
Download PDF
Retinal detachment is a condition in which the neurosensory retina is separated from the retinal pigment epithelium. If untreated, permanent loss of vision may occur. Types of retinal detachment include rhegmatogenous, exudative, tractional, combined tractional-rhegmatogenous, and macular hole–associated detachment. Rhegmatogenous retinal detachment (RRD) is the most common of these. Part 1 of this 2-part article covers RRD risk factors, features, and examination. Next month, part 2 covers management.
 |
|
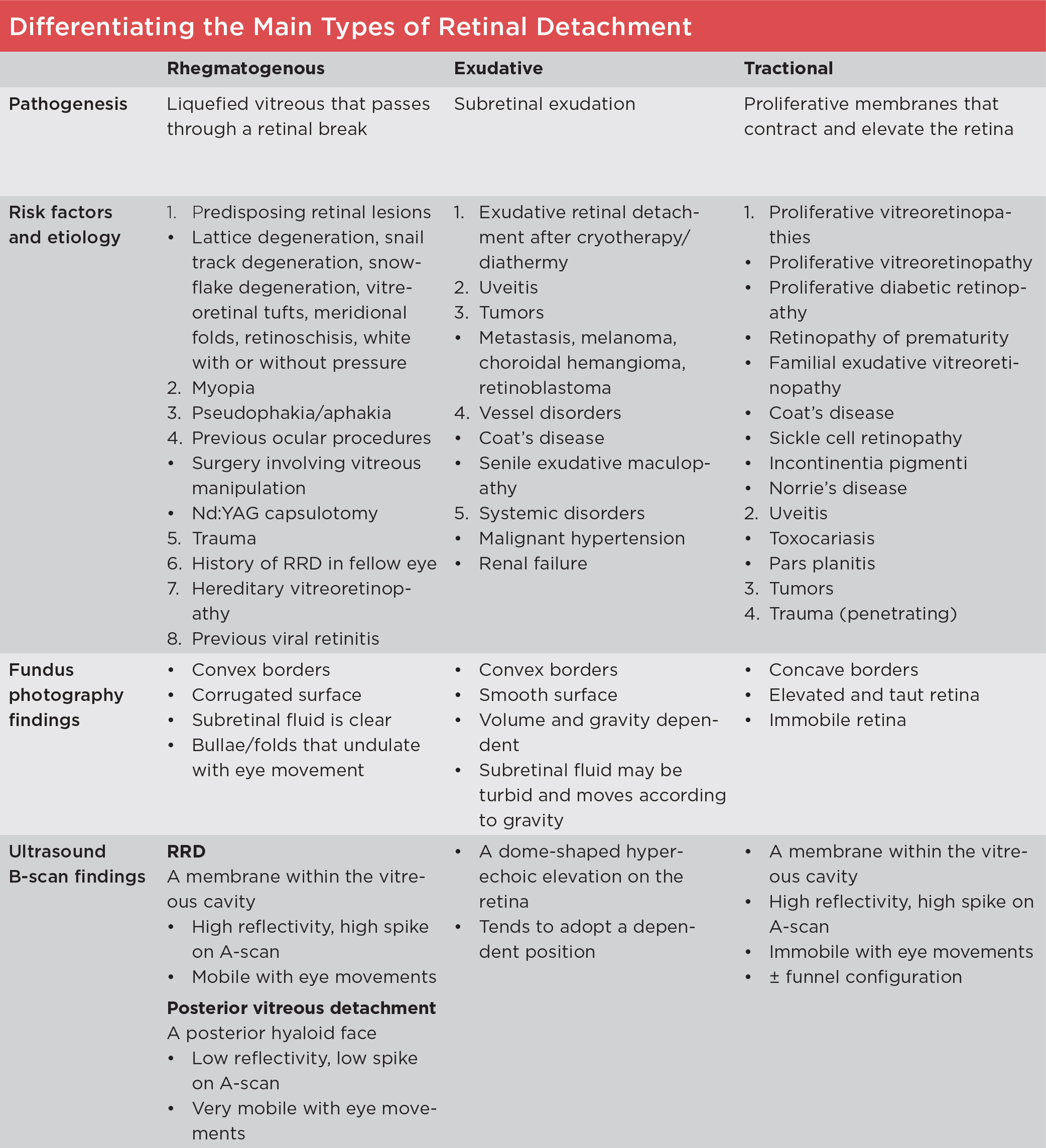
RRD. Macula-off primary rhegmatogenous retinal detachment with multiple breaks located within 1.5 clock hours of the highest border of the detachment (consistent with Lincoff rules).
|
Defining RRD
The word rhegmatogenous is derived from the Greek word rhegma, which means broken. The pathogenesis of RRD involves vitreoretinal tractional forces that result in a full-thickness retinal break. Liquefied vitreous gel then enters the subretinal space through the break, causing separation of the neurosensory retina from the underlying retinal pigment epithelium.1
Total RRD denotes separation of the entire retina; subtotal RRD refers to detachment of most of it. Subclinical retinal detachments are those with subretinal fluid extending more than 1 disc diameter from the break but less than 2 disc diameters posterior to the equator. If subretinal fluid extends less than 1 disc diameter, the condition is defined as a retinal break without detachment.2
 |
|
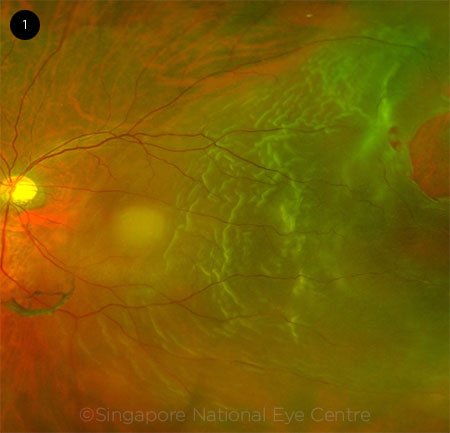
AMSLER-DUBOIS RETINAL CHART. The innermost circle represents the equator, the middle circle represents the ora serrata (scalloped edges), and the outermost circle represents the junction of the pars plana and pars plicata. Lesions commonly associated with RRD are marked: a horseshoe tear (2 o’clock position) with a torn vessel, a resultant retinal detachment (extending through 3 clock hours), lattice degeneration (8 o’clock), and vitreous hemorrhage inferiorly (green area).
|
Risk Factors
Risk factors for RRD include high myopia, trauma to the eye or head, RRD in the fellow eye, underlying hereditary vitreoretinopathy, previous intraocular surgeries, and previous viral retinitis. Other risk factors are intraocular procedures (especially vitreous manipulation), laser capsulotomy, pseudophakia/aphakia,3 and retinal lesions such as lattice degeneration, snail track degeneration, snowflake degeneration, vitreoretinal tufts, meridional folds, retinoschisis, and white lesions (with or without pressure).2
 |
|
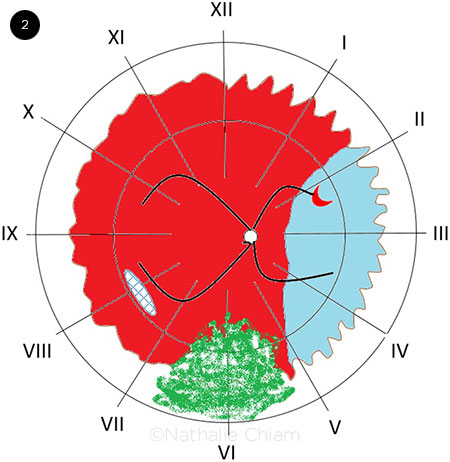
TOTAL RETINAL DETACHMENT. Ultrasound A-scan and B-scan of an eye with total retinal detachment, resulting in a funnel-shaped configuration. A hyperechogenic interface can be seen in the vitreous cavity on the B-scan (black-and-white image). This interface has a high-amplitude A-scan spike (blue line) that reaches the posterior sclera, suggesting that the interface is retinal tissue. If the A-scan spike of a hyperechogenic focus within the vitreous is of low amplitude, it is more likely to indicate posterior vitreous face, vitreous hemorrhage, or vitritis, depending on the clinical picture. In a rhegmatogenous detachment, the hyperechogenic interface of the high-amplitude A-scan spike should undulate with eye movement, whereas cases with PVR or tractional retinal detachment will demonstrate reduced mobility of the retinal interface.
|
Clinical Features
Patients with RRD may present with floaters, photopsia, and/or a “curtain” defect that obscures part of the visual field. Visual acuity (VA) ranges from excellent to poor, depending on whether the macula is still attached. In patients with macula-off RRD, vision usually is decreased. If the area of detachment is large, an afferent pupillary defect may be present.
Intraocular pressure (IOP) can be low or high. Low IOP results from increased outflow of intraocular fluid through the subretinal space and peripapillary connective tissue, particularly if the optic disc border is involved. High IOP may occur with chronic RRD, in which photoreceptor outer segments transgress into the anterior chamber and trabecular meshwork, impeding aqueous outflow. This is also known as Schwartz-Matsuo syndrome. Other features of chronic RRD may include a pigmented demarcation line at the detachment border, intraretinal macrocysts, atrophic thinned retina, subretinal white precipitates, and signs of proliferative vitreoretinopathy (PVR), such as fixed retinal folds.
Assessment of RRD requires a thorough 360-degree fundus examination. When visualization of the fundus is poor, as in patients with dense cataract or vitreous hemorrhage, an ultrasound B-scan may be useful.
Lincoff Rules: Finding the Break in Primary RRD
Rule 1. In superior-temporal or superior-nasal detachments: The primary break is within 1.5 clock hours of the highest border (in 98% of cases).
Rule 2. In total detachments or superior detachments that cross the 12 o’clock meridian (vertically above the disc): The primary break is at 12 o’clock or the break is a triangle with the apex at the ora serrata and the base at the equator, extending from 11 to 1 o’clock (in 93% of cases).
Rule 3. In inferior detachments: The higher side of the detachment indicates the side of the disc where the primary break lies, and the break is found below the horizontal meridian (in 95% of cases).
However, in inferior detachments where right/left borders are equally high, the break is in the inferior retina at 6 o’clock.
Rule 4. In inferior bullous detachments: The primary break is located above the horizontal meridian.
SOURCE: Lincoff H, Gieser R. Arch Ophthalmol. 1971;85(5):565-569.
|
Examination
Binocular indirect ophthalmoscopy (BIO) of the fundus. BIO with a lens of 20 or 28 D allows visualization of the peripheral retina. For some eyes, scleral depression during indirect ophthalmoscopy or contact fundus lens examination using the slit lamp (e.g., Goldmann 3-mirror lens) may help view smaller peripheral retinal breaks.
Examination. This should include the following steps:
- Identify the extent of detachment. The detached area will appear opaque and corrugated, with undulating retinal folds during eye movement. The borders of the detached tissue usually are convex, and the subretinal fluid is clear and nonshifting. (See “Differentiating the Main Types of Retinal Detachment,” for pearls to differentiate tractional and exudative retinal detachment from RRD.) Other features that may accompany RRD include a positive Shafer’s sign (pigment in the anterior vitreous), vitreous hemorrhage, and lower IOP than in the fellow eye.
- Find all retinal breaks, which will help guide the surgical approach. It is important to note the size, number, and location of each break. Lincoff rules are useful for identifying the precise location of the retinal break in cases of primary RRD.3 If there are multiple breaks, the highest retinal hole is considered the primary hole. (See “Lincoff Rules.”)The location of retinal detachment plays a major role in management and prognosis.
- Determine whether the RRD is macula-on or macula-off (Fig. 1). Although visual prognosis is much better for macula-on RRD that spares the fovea, urgent intervention is still needed.
- Check for associated features. Retinal lesions that predispose to retinal breaks, such as lattice degeneration, should be identified. Also look for features that might affect management and prognosis, such as coexisting vitreous hemorrhage and PVR.
- Document the findings on an Amsler-Dubois chart or in the electronic medical record, using color codes and symbols to represent retinal lesions (Fig. 2).
Ultrasonography. If the fundus view is obscured, dynamic B-scan ultrasonography is helpful to confirm RRD and determine the status of macular involvement, presence of posterior vitreous detachment, location of retinal break (occasionally), and chronicity of RRD (mobile or fixed).
Typical ultrasound findings for RRD include high reflectivity, a high spike on the A-scan, a membrane within the vitreous cavity, and mobility during eye movements. Posterior vitreous detachment is characterized by a posterior hyaloid face, low reflectivity, a low spike on the A-scan, and a high degree of mobility during eye movements (Fig. 3, demonstrates the ultrasound appearance of a funnel retinal detachment).
___________________________
Acknowledgment: The authors thank the Ocular Imaging Department, Singapore National Eye Centre, for providing the retinal photographs, including the B-scan ultrasonography image.
___________________________
1 Kuhn F, Aylward B. Ophthalmic Res. 2014;51(1):15-31.
2 Schubert HD et al. Retinal detachment and predisposing lesions. Retina and Vitreous. San Francisco: American Academy of Ophthalmology; 2016-2017.
3 Lincoff H, Gieser R. Arch Ophthalmol. 1971;85(5):565-569.
___________________________
Dr. Chiam is an ophthalmology resident at Singapore National Eye Centre. Dr. Ting is the surgical retinal fellow at the Singapore National Eye Centre and an assistant professor at Duke-National University Singapore (Duke-NUS) Medical School. Dr. Lee is the senior consultant, deputy head of the Surgical Retinal Department of Singapore National Eye Centre, and an adjunct associate professor at Duke-NUS. Dr. Ang is a senior consultant at the Surgical Retinal Department of Singapore National Eye Centre and a clinical professor at Duke-NUS. Financial disclosures: None.