Download PDF
Intravitreal bevacizumab can be an effective treatment for retinopathy of prematurity (ROP) even at very low doses, a team of British clinical researchers has reported.
In a retrospective case series,1 the group reported that ROP regressed completely in nearly 80% of the eyes after a single injection of bevacizumab (Avastin). The dosage—0.16 mg in 0.025 mL—was approximately one-fourth the amount that has been used in these infants for several years (0.625 mg in 0.025 mL), said Roxane J. Hillier, FRCOpth, at the Royal Victoria Infirmary in Newcastle upon Tyne, U.K.
Addressing systemic concerns. “The introduction of anti-VEGF agents to treat ROP has completely transformed our management of these babies with severe retinopathy,” Dr. Hillier said. “But it’s imperative that we make sure that the dose we’re using is as low as possible, because there are concerns about the impact these drugs might have on the babies’ systemic development,” particularly their neurological and respiratory systems, she said.
In an Academy Ophthalmic Technology Assessment published earlier this year,2 the authors concluded that there is level II and III evidence that intravitreal anti-VEGF therapy is at least as effective as laser photocoagulation for resolving ROP. But they noted lingering concerns about potential effects on organ development. “After intravitreal injection, bevacizumab can be detected in serum within 1 day, and serum VEGF levels are suppressed for at least 8 to 12 weeks,” the OTA report said.
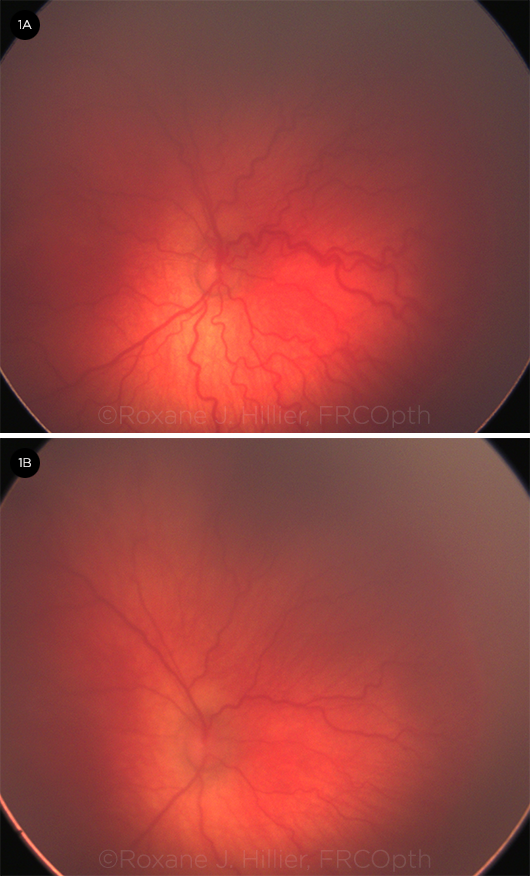 |
BEFORE AND AFTER. This male twin was born at 23 weeks’ gestational age. These images are of his right eye at baseline (1A) and 2 days after he received an intravitreal injection of low-dose bevacizumab (1B).
|
Study results. In Dr. Hillier’s case series, there were 15 infants (29 eyes) with severe and posterior ROP. Primary success with intravitreal bevacizumab alone was seen in 23 of 29 eyes (79.3%). The other 6 eyes required retreatment, either with another injection or with laser photocoagulation.
Improvement in retinopathy and in plus disease began in all cases within 48 hours after the injection, Dr. Hillier and her colleagues reported. “This response was notably less brisk when compared with higher dose intravitreal bevacizumab. Nonetheless, in our experience, early signs of resolution may be expected within 48 hours, even at this very low dose,” they wrote.
“What dose should we be using? We still don’t know the answer,” Dr. Hillier said. “But I think this paper brings us a step closer to understanding that the traditional dose that we’ve been using for the last few years is probably vastly in excess of what is needed.”
Additional investigations. To clarify the issues of dosing and potential systemic risks, the Pediatric Eye Disease Investigator Group currently is conducting a prospective phase 1 multicenter trial to determine the lowest effective bevacizumab dose for treatment of type 1 ROP. This study also will collect serum levels of VEGF and bevacizumab after the injection.
Other studies in Germany and the United States are looking at the possibility that ranibizumab (Lucentis) might reduce the potential systemic risks because it has a half-life of a few hours in the circulation, compared to weeks for bevacizumab.2
“I think that actually there is a very good rationale potentially for using Lucentis rather than Avastin,” Dr. Hillier said. “But thus far, the most robust scientific evidence for using anti-VEGF drugs in this group of babies is with Avastin.”
—Linda Roach
___________________________
1 Hillier RJ et al. Br J Ophthalmol. Published online June 27, 2017.
2 VanderVeen DK et al. Ophthalmology. 2017;124(5):619-633.
___________________________
Relevant financial disclosures—Dr. Hillier: None.
For full disclosures and disclosure key, see below.

More from this month’s News in Review