Download PDF
Although anti-VEGF therapies can improve visual acuity in many patients with neovascular age-related macular degeneration (AMD), predicting response to treatment remains a clinical challenge. Researchers from Johns Hopkins School of Medicine in Baltimore have found that aqueous biomarkers may help identify patients who might not require or benefit from lifelong anti-VEGF therapy.1
The study findings suggest that up to one-third of patients receiving anti-VEGF injections for AMD can be safely taken off treatment, thus sparing them any potential risks of repeated intraocular injections and the burden of frequent clinic visits, said coauthor Akrit Sodhi, MD, PhD.
Moreover, the results also suggest that apolipoprotein-B100 and other aqueous proteins that have been implicated in macular degeneration may, in fact, play a protective role in AMD, Dr. Sodhi added.
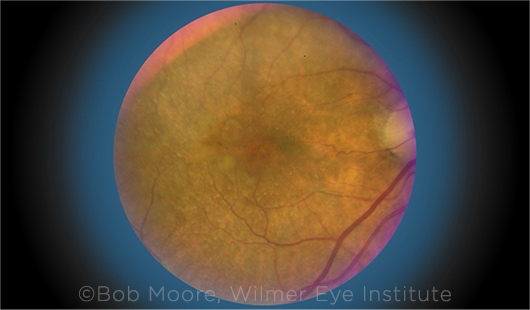 |
PARADOX. Drusen, pigmentary changes, pigment epithelial detachments, edema, and scant hemorrhage in a patient with untreated neovascular AMD. Apolipoprotein-B100 may play a paradoxical protective role in some patients with the disease.
|
Study design. The researchers used an approach that they termed “treat-and-extend-pause/monitor,” Dr. Sodhi said. Eyes with neovascular AMD had three consecutive monthly anti-VEGF injections, followed by a treat-and-extend protocol. If eyes remained quiescent, they were switched to treatment on a pro re nata basis. Proteomic assays were used to compare the levels of aqueous proteins between patients who responded well to therapy and those who did not.
Results. At the end of 12 and 24 months, 31% and 38% of eyes, respectively, were successfully weaned off of treatment.1
With regard to whether the aqueous fluid proteins could be used as a surrogate marker, approximately 170 proteins were found to correlate with patients’ response to anti-VEGF therapy, Dr. Sodhi noted. He added that many of these proteins had been previously implicated in AMD pathogenesis.
An unexpected finding. “Perhaps the most surprising and exciting finding of the study is that apolipoprotein-B100 was found in higher levels in patients who required less frequent injections,” Dr. Sodhi said. In contrast, prior data have suggested that apolipoprotein-B100 may contribute to AMD pathogenesis by driving dry macular degeneration.
Investigating further. To validate their finding, the researchers measured aqueous apolipoprotein-B100 levels in an independent cohort. “We found that aqueous apolipoprotein-B100 levels were higher in AMD eyes than in controls and lower in AMD eyes with choroidal neovascularization than in patients with dry AMD,” Dr. Sodhi said. The researchers also studied mice that overexpress apolipoprotein-B100 in the part of the retina that plays a role in macular degeneration—and found that apolipoprotein-B100 overexpression protected the mice from laser-induced choroidal vascular lesions.
Looking ahead. Dr. Sodhi noted that further research is needed to elucidate the role of apolipoprotein-B100 in wet AMD. “We also want to conduct a prospective clinical trial to validate these potentially protective proteins as well as aqueous biomarkers that could predict the response to anti-VEGF therapy.”
—Christos Evangelou, PhD
___________________________
1 Cao X et al. J Clin Invest. 2022;132(2):e133369.
___________________________
Relevant financial disclosures—Dr. Sodhi: NEI: S.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Chen UCB: C; Roche: C.
Dr. Pfau Apellis: C; Novartis: L.
Dr. Redd NEI: S; Research to Prevent Blindness: S.
Dr. Sodhi Arrowhead Pharmaceuticals: C; HIF Therapeutics: O; XCaliber Biotechnology: C.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|
More from this month’s News in Review