Download PDF
The recently published PPP guidelines shed a new light on treatment options for keratoconus and other ectasias. One approach—corneal cross-linking—may well reshape the future of therapy.
When members of the Academy’s Cornea Panel for the Preferred Practice Pattern (PPP) guidelines convened last year, they quickly realized the challenge inherent in their assignment. They’d been charged to write a single PPP bringing the comprehensive ophthalmologist up to date on the current management of corneal opacification, edema, and ectasia. But with an expanding number of advanced diagnostic techniques and treatments now available, said cochair Robert S. Feder, MD, it soon became clear that corneal ectasia warranted a PPP of its own.1
Of the various therapeutic options included in the guidelines, corneal collagen cross-linking (CXL) is garnering the greatest interest. Approved in Europe since 2007, the technology has been used on an investigational basis in the United States since 2008. At time of press, an FDA determination was expected on March 15. Thus far, CXL is the only approach that addresses the underlying biomechanical weakness of the ectatic cornea.
 |
|
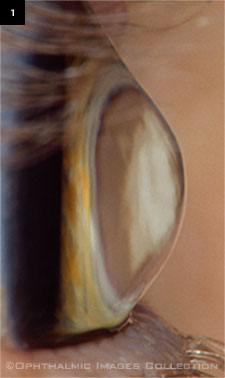
Keratoconus.
|
Ectasia Overview
Corneal ectasia is a thinning and bulging of the cornea, varying in severity and rate of progression, that can cause substantial visual distortion and other visual symptoms. The causes of this condition typically fall into two groups: 1) spontaneously occurring, usually bilateral entities such as keratoconus (Fig. 1), pellucid marginal degeneration, and keratoglobus; and 2) ectasia that manifests after keratorefractive surgery and is often considered at least partially iatrogenic.2
When to suspect ectasia. Although the diagnosis is straightforward in severely affected patients, subtle cases may be difficult to detect. In general, said Dr. Feder, the comprehensive ophthalmologist should pursue further investigation in certain types of patients, such as 1) a young person whose refraction changes frequently, for example, every three months; 2) an apparently healthy individual who cannot achieve sharp vision on refraction; or 3) a patient with a strong family history of keratoconus. “Investigation of ectasia is also recommended following keratorefractive surgery if the refractive error becomes unstable,” he said.
Postsurgical ectasia. Post-keratorefractive corneal ectasia can develop in a previously normal eye if the LASIK flap was thicker than intended or if the laser ablation was too deep for that particular cornea, said Dr. Feder, at the Northwestern University Feinberg School of Medicine in Chicago. In other cases, a preexisting subclinical form of ectasia is present—a condition that could be detected preoperatively by corneal mapping. A third category includes patients with no presurgical sign of ectasia in whom surgery was planned and executed properly.
Stephen Slade, MD, a cornea and refractive specialist in Houston, offered an explanation for these puzzling cases of post-keratorefractive ectasia. “I don’t think refractive surgery causes keratoconus, but it can uncover subclinical dystrophies.” Just as walking on a treadmill may bring on symptoms of angina, he said, thinning the cornea may prompt symptoms of keratoconus. “What we’ve been doing in these cases is ‘stress testing’ already diseased corneas.”
Another possibility is the influence of estrogens. Women whose corneas remained stable for many years after LASIK sometimes develop ectasia during their first pregnancy.3
Other associated conditions. Even the microtrauma associated with eye rubbing or contact lens wear can be a significant risk factor for the development of keratoconus, said Dr. Feder. This may partially explain why keratoconus is often associated with atopic diseases such as hay fever and eczema.
“Another theory is that a genetic predisposition to developing keratoconus may be present,” said Dr. Feder, “but does no harm until a trigger like eye rubbing incites the condition to become manifest.” Keratoconus can be associated with Down syndrome, tapetoretinal degenerations, and even floppy eyelid syndrome, he said. Obstructive sleep apnea is also more prevalent in patients with keratoconus.4
 |
|
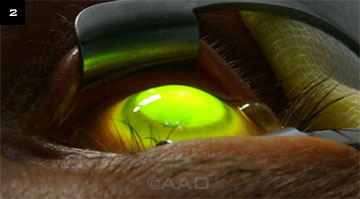
The cornea is thoroughly soaked with photosensitizing riboflavin before exposure to the UVA light beam.
|
The Treatment Stepladder
The new PPP reviews the range of treatment options for corneal ectasia and keratoconus, starting from least invasive step—spectacles—to penetrating keratoplasty.
Glasses and contact lenses. The first step has traditionally been glasses, with the patient moving on to contact lenses as the disease progresses, typically wearing them for many years, said Dr. Feder. “The vast majority of patients with keratoconus can be fitted with a rigid gas-permeable lens. When comfort is an issue, it’s possible to fit toric soft lenses and get good results in many patients, particularly those with mild disease.” But the biggest innovation, he said, has been the scleral lens, which is well tolerated and can result in dramatic improvements in visual function.
Intrastromal corneal ring segments (ICRS). Inserted at two-thirds of the stromal depth through channels created mechanically or with a femtosecond laser, these semicircular rings may help improve contact lens tolerance in mild to moderate cases by recentering the cone, said Dr. Feder. They may also improve vision by reducing contour irregularities.1
Intraocular lens (IOL). When cataract surgery is indicated, a toric lens implant can correct both myopia and regular corneal astigmatism. “In the setting of ectasia, the toric IOL should only be used if the astigmatism is regular, meaning it can be corrected with spectacles, and the corneal condition is stable,” said Dr. Feder. He noted that only corneal astigmatism, not total refractive astigmatism, can be corrected with a toric IOL. The toric IOL is a poor choice, he said, if a contact lens will still be required due to irregular astigmatism or if keratoplasty is likely to be necessary because the degree and axis of astigmatism are likely to change.
In keratoconus patients without cataract, a phakic IOL may be helpful if anisometropia is the predominant refractive problem, crisp acuity can be achieved with manifest refraction, and contact lenses are not tolerated.
Keratoplasty. The indications for considering a keratoplasty include a patient’s inability to achieve adequate visual function, reasonable comfort, or a stable fit with less invasive therapies, said Dr. Feder. Penetrating keratoplasty was traditionally the procedure of choice, but the trend is shifting toward more targeted procedures, such as deep anterior lamellar keratoplasty (DALK). In this approach, the surgeon removes the corneal stroma to Descemet’s membrane but preserves the healthy endothelium. Best visual outcomes are achieved when the stroma is completely dissected off Descemet’s membrane. “DALK is probably the future of keratoplasty for many cases, but it has a fairly steep learning curve and has risks of stromal rejection or corneal perforation,” said Dr. Feder.
Reshaping the Landscape of Ectasia Treatment
Unlike other treatment options, CXL halts the progression of ectasia by improving the structural integrity of the cornea.5 Utilizing ultraviolet-A (UVA) light and a topical photosensitizer (riboflavin; Fig. 2), the procedure create new cross-links between collagen lamellae. According to the new PPP, CXL can be considered in the early stages of the disease.1
CXL systems. Today, there are about a dozen corneal cross-linking systems on the market internationally. Each emits UV light of a specific wavelength but can vary the power, or fluence, as measured in milliwatts (mW), said Dr. Slade.
Dresden protocol. “The original Dresden protocol developed by Prof. Theo Seiler and Prof. Eberhard Spoerl involves exposing eyes to 3 milliwatts of UVA for 30 minutes after saturating the cornea with topical riboflavin,” Dr. Slade said. He added that a variety of innovations, including eye trackers, timers, and video cameras—as well as different ways to apply the light—have since been incorporated into various systems.
 |
|
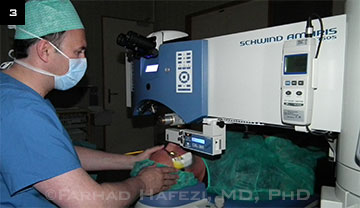
Administering treatment with a system that allows for varying levels of fluence and exposure time.
|
A faster method. “Accelerated cross-linking is simply providing the same amount of treatment with a higher fluence but shorter treatment time,” said J. Bradley Randleman, MD, at Emory University. According to the Bunsen-Roscoe law of reciprocity, you can theoretically treat in one-fifth of the time, for example, by using five times the energy, added Farhad Hafezi, MD, PhD, a CXL pioneer in Zurich (Fig. 3). However, he noted that beyond a certain point, speed offers diminishing returns.
Dr. Hafezi said, “We tested this in the lab, and starting at 18 milliwatts, we see a drastic decrease in biomechanical stiffness because the cornea simply doesn’t have enough oxygen molecules to sufficiently cross-link the collagen tissue.6 If you do cross-linking more slowly, the tissue has time to reoxygenate.”
Results: stability plus. “We have a 96 or 97 percent success rate in stabilizing the cornea with cross-linking,” said Dr. Hafezi, “and a flattening of the cornea occurs in about 40 percent of patients, which translates into better visual acuity. We never promise, but many patients gain one, two, or even three lines of vision.”
A collaborator in one of the earliest Dresden clinical trials, Frederik Raiskup, MD, PhD, FEBO, recently reported that the effect of the procedure can last up to 10 years. “Even in cases where the ectasia progressed again after CXL, such as in very young patients, atopic patients, or women during pregnancy,” he said, “it has been possible to repeat the procedure with few side effects, which all resolved within several weeks.”
Candidates for CXL. In general, a good CXL candidate is a patient with mild to moderate progressive keratoconus or progressive pellucid marginal degeneration. There is controversy about the effectiveness of CXL for post-keratorefractive ectasia, but Dr. Hafezi said that in his experience, it is as effective in this setting as with those other conditions, citing follow-up of 26 eyes from 11 countries up to five years after refractive surgery.7 He added that a dozen small case studies have reported similar results in a total of about 120 eyes.
How thin is safe? “Corneal thickness is one of the initial drivers of whether or not cross-linking can be performed, how to perform it, and how successful it will be,” said Dr. Randleman. Initially, inclusion criteria required a minimum corneal thickness of 400 µm after removal of the epithelium, added Dr. Hafezi. However, in 2006, he and his colleagues introduced hypo-osmolar riboflavin solutions that can swell a thinner cornea above 400 µm and allow treatment with cross-linking.
Still, very thin corneas and high keratometric (K) readings are more likely to develop permanent corneal scars, said Dr. Raiskup, which can result in reduction of visual acuity and failure of the treatment. “For these reasons, we recommend CXL in corneas with a minimal stromal thickness of 320 µm or greater after epithelial removal and prior to swelling with hypo-osmolar riboflavin solution,” he said.
K’s and VAs. Dr. Raiskup said that the higher the K readings, the higher the failure rate, citing a study showing that reducing the Kmax value from less than 65 D to less than 58 D in selecting candidates would reduce the failure rate from 7.6 percent to 2.8 percent.8
Nevertheless, Dr. Raiskup considers visual acuity (VA) to be the deciding factor. If the patient still has best-corrected VA that he or she considers usable, it is worth “fighting for this cornea” to find a way to stop the progression of ectasia with cross-linking, even in marginal cases. Such cases must be considered individually, and the patient carefully counseled, he said.
Pediatric use. The 9th International Congress of Corneal Cross-Linking (held in 2013) reached consensus that CXL is beneficial in children and adolescents, with three papers reporting effectiveness up to two years after the procedure. A study coauthored by Dr. Hafezi, however, did not find a flattening effect after three years.9 “This could mean the effect is less long lasting in children,” he said, “which would make sense since the disease is most aggressive during this time.” Dr. Hafezi recommends continuing to follow these young patients closely and re-treating, if necessary.
Dr. Hafezi’s study also found that keratoconus progressed in 88 percent of children and teens. Given this significant finding, the International Congress of Corneal Cross-Linking recommends immediate treatment after diagnosis in this group, rather than waiting to see if progression occurs.
Prophylactic CXL During Refractive Surgery
Some surgeons are currently performing cross-linking at the time of laser refractive surgery with the goal of preventing subsequent ectasia.
Such combinations can be problematic. Even though the laser component is highly precise, the CXL procedure is good at stabilizing the cornea—but not at producing a predictable refractive outcome, said Dr. Hafezi. He noted that the K readings of the cornea can change several diopters after cross-linking.
Uncharted. With no clinical validation, this is really uncharted territory, warned Dr. Hafezi, who is concerned about surgeons testing the limits of biomechanics under the cover of cross-linking. A false sense of security, he said, may prompt refractive surgery on patients who would never have been considered as candidates in the past, such as young patients with thin corneas and subclinical keratoconus.
A fine line. Prophylactic cross-linking combined with refractive surgery would have to be the “Goldilocks” model, agreed Dr. Randleman. In other words, just right. You would need to create some increase in strength but no change in curvature, he said, which, in theory, could be beneficial. “But that’s a fine line to determine.” |
CXL Controversy: Epi-on vs. Epi-off
Epi-off. The standard protocol for CXL involves epithelial debridement (epi-off)—an easy, relatively safe, and effective procedure temporarily followed by pain of varying intensity and decreased visual acuity, said Dr. Raiskup. However, vision-threatening complications such as corneal infection, melting, and even perforations have been reported in rare cases.10
Epi-on (transepithelial). “To minimize postoperative problems, clinicians have looked for ways to treat without removing the epithelium,” said Dr. Raiskup. “Epion’s noninvasive nature makes it potentially useful in cases where epithelial debridement is ideally avoided, such as with uncooperative patients or those with additional ocular surface pathology, dry eyes, or very thin corneas.”
 |
|
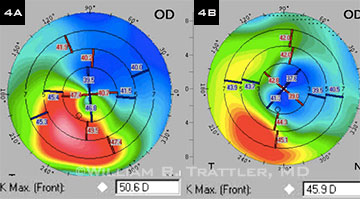
(4A) Preop topography of a 68-year-old man with keratoconus. (4B) Eighteen months after he underwent epi-on CXL, his corneal topography had improved significantly, with a 4.7 D reduction in Kmax and a reduction in irregular astigmatism within the visual axis.
|
Research underway. Epi-on’s safety goes unchallenged, but questions remain about its efficacy. “Everyone would love to go to epi-on,” said Dr. Randleman, “but we don’t have good proof-of-concept evidence that a sufficient amount of riboflavin actually reaches the corneal stroma.” Dr. Raiskup said that clinical observations from his colleagues across Europe regarding epi-on results range from “less effective than standard CXL” to “moderately effective” to “appearing to halt keratoconus progression, with a statistically significant improvement in visual and topographic parameters.”
William B. Trattler, MD, a cornea specialist in Miami, is currently conducting a 15-center clinical epi-on CXL trial for patients aged 8 years old and up. Dr. Trattler said that the procedure has stopped progression in 99 percent of trial patients to date, with improvement in corneal shape (Fig. 4) in a large majority and improvement in vision in over 50 percent of eyes. Patients have been followed for up to three years. Data from this trial, presented at the 2013 Academy annual meeting, are being prepared for publication, he said. The positive clinical results, along with its high level of safety, convinced Dr. Trattler to treat his daughter with this technique when she was diagnosed with keratoconus.
“At age 9, she had mild myopia and normal corneal topography, but she developed keratoconus at age 12, and we performed her epi-on cross-linking in January 2013,” said Dr. Trattler, who hasn’t performed epi-off in about four years. “A year out, she’s had nice improvements in both corneal shape and vision.”
Enhancing penetration. Dr. Trattler said he thinks that previous epi-on trials have been unsuccessful because not enough riboflavin reached the cornea before the initiation of the UV light. “For transepithelial cross-linking, we use corneal sponges designed to help maximize the contact time between the riboflavin and the cornea,” he said. “We also use a proprietary riboflavin formulation that penetrates the cornea in a much more rapid fashion.”
A variety of other methods are being explored to enhance penetration of riboflavin into the stroma while preserving the epithelium, said Dr. Randleman. They include using iontopheresis, disrupting tight epithelial junctions with 20 percent alcohol, and making many channels into the cornea with a femtosecond laser or mechanical device.
CXL in the USA
In the United States, patients have been treated with CXL since 2008, but only in clinical trials. Currently, more than 130 sites offer cross-linking, said Dr. Trattler. He strongly encourages colleagues to refer their ectasia patients to one of these sites, especially given the variability of the disease’s progression.
Seeking FDA approval. Started at Emory University and based on the Dresden CXL protocol, an earlier multicenter study sponsored by the Swiss-based company, Peschke Meditrade GmbH, failed to secure FDA approval. Dr. Randleman and Doyle Stulting, MD, PhD were co-investigators. Avedro later purchased the data, which was granted priority review status in Nov. 2013. At time of press, an FDA determination was expected on March 15.
Other U.S. studies. More than 20 trials are currently underway in the United States. Among the topics of investigation are epi-on versus epi-off approaches, different formulations and protocols for riboflavin administration, and CXL in combination with ICRS.
With more than 800 eyes and close to 100 investigators, a phase 3 randomized controlled CXL trial sponsored by the American College of Ophthalmic Surgery (ACOS) is one of the largest, said Dr. Slade, chairman of the board of ACOS. This trial is testing the safety and efficacy of three accelerated cross-linking treatment regimens of 15, 30, and 45 mW. |
Combination Treatment: Building Future Bridges
“Cross-linking is the cornerstone from which we build,” said Dr. Randleman. That’s because, although it stops progression, it does not reverse ectasia nor provide complete visual rehabilitation by itself.
Still a role for contacts. Some patients will achieve improved shape and vision after cross-linking, said Dr. Trattler, and not feel the need for another refractive procedure. “After cross-linking, many patients will be able to get into a contact lens for the first time,” added Dr. Randleman. “Rigid contact lenses still provide the best acuity for patients with irregular corneas, and I don’t see that changing anytime in the near future.”
Feasibility first. Others—both patients and physicians—may be eager to try something more. “We have 15 years of data on cross-linking alone, but what we know now about combination treatment is that we don’t know enough yet,” said Dr. Randleman. Thus far, data on the combination of CXL with other refractive treatments have come from case reports and small studies, added Dr. Raiskup. “To determine which refractive procedure and which sequence is most suitable to combine with CXL will require controlled studies with larger cohorts and longer follow-up.”
CXL plus ablation. Topography-guided ablation is sometimes combined with CXL to improve the corneal contour. Although these procedures often are performed simultaneously, both Dr. Trattler and Dr. Hafezi prefer to do the cross-linking procedure first.
“You can wait until the cornea finds its plateau and comes to rest; then you have a stable refraction and can remove tissue by surface ablation,” said Dr. Hafezi. He has performed hundreds of ablations on porcine corneas to determine the amount of tissue to ablate in a cross-linked cornea, which he said behaves quite differently from a virgin cornea. He hopes that this research will help establish nomograms for more accurate treatments in the future.
 |
|
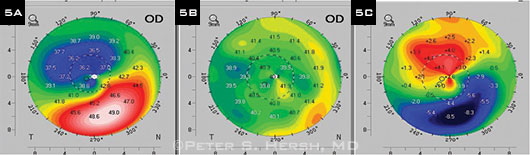
(5A) Preoperative corneal topography of patient who had CXL and Intacs. (5B) One year after CXL followed by placement of one 400-µm Intacs segment three months later, map shows improved symmetry of topography. (5C) Difference map; note flattening of 8.5 D over cone apex.
|
CXL plus ICRS. It’s also possible to improve the contour of an ectatic cornea by implanting intracorneal ring segments in addition to performing CXL (Fig. 5). It’s too soon to know which sequence works better—CXL or ICRS first—although there are several small reports of ICRS implantation followed by CXL resulting in greater improvement in curvature and visual acuity.10
There may be an advantage to performing ICRS first because it is more difficult to create the intrastromal channels after CXL, said Dr. Feder, and you want to lock in the contour after you have recentered the cone with the ICRS procedure, not before.
__________________________
1 American Academy of Ophthalmology, Cornea/External Disease Panel. Preferred Practice Pattern Guidelines. Corneal Ectasia. 2013. Available at: www.aao.org/ppp.
2 Seiler T et al. J Refract Surg. 1998;14(3):312-317.
3 Hafezi F et al. J Refract Surg. 2012;28(4):242-243.
4 Gupta PK et al. Cornea. 2012;1(6):595-599.
5 Raiskup F, Spoerl E. Ocul Surf. 2013;11(2):65-74.
6 Richoz O et al. Transl Vis Sci Technol. 2013;2(7):6.
7 Richoz O et al. Ophthalmology. 2013;120(7):1354-1359.
8 Koller T et al. J Cataract Refract Surg. 2009;35(8):1358-1362.
9 Chatzis N, Hafezi F. J Refract Surg. 2012;28(11):753-758. [Erratum in J Refract Surg. 2013;29(1):72.]
10 Raiskup F, Spoerl E. Ocul Surf. 2013;11(2):93-108.
Meet the Experts
ROBERT S. FEDER, MD Professor of ophthalmology, Chief, Cornea/External Disease Service, Northwestern University Feinberg School of Medicine, Chicago. Financial disclosure: None
FARHAD HAFEZI, MD, PHD Chair and professor of ophthalmology, University Eye Clinic of Geneva, Switzerland; clinical professor of ophthalmology, USC Keck School of Medicine, Los Angeles; cofounder of the International Congress of Corneal Cross-Linking; cofounder of the IROC Institute, the developer of the first UV-X CXL device. Financial disclosure: None.
FREDERIK RAISKUP, MD, PHD, FEBO Senior consultant, Cornea, External Diseases and Refractive Surgery Unit, at Carl Gustav Carus University Hospital, Dresden, Germany; associate professor of ophthalmology, Hradec Králové, Charles University, Prague. Financial disclosure: None.
J. BRADLEY RANDLEMAN, MD Professor of ophthalmology and director of Cornea & Refractive Surgery Section at Emory Eye Center, in Atlanta. Financial disclosure: None.
STEPHEN SLADE, MD Specialist in cataract and refractive surgery, Slade & Baker Vision Center, in Houston; chairman of the board of American College of Ophthalmic Surgeons. Financial disclosure: None.
WILLIAM B. TRATTLER, MD Refractive, corneal, and cataract eye specialist, Center for Excellence in Eye Care, in Miami. Financial disclosure: Has a financial interest in CXLO. |