Update: On Aug. 29, 2018—after the article below went to press—Alcon announced a voluntary, global recall of the CyPass microstent.
Download PDF
In the decade since Iqbal Ike K. Ahmed, MD, coined the term microinvasive glaucoma surgery—better known as MIGS—the field has grown exponentially.
Dr. Ahmed noted several reasons for the growing interest in MIGS: “Compliance is really poor in glaucoma patients, and even if the patient is compliant, quality of life and the high costs of medicine continue to be issues. We must continually look for solutions for these patients. MIGS are moving in that direction,” said Dr. Ahmed, who is a glaucoma specialist with the Prism Eye Institute in Mississauga, Ontario, Canada.
An online survey of glaucoma surgery practice preferences conducted by the American Glaucoma Society found that among patients who had initial surgery for primary open-angle glaucoma (POAG), the percentages were 59% for trabeculectomy with mitomycin C (MMC), 23% for a glaucoma drainage device (GDD), and 14% for MIGS. When the glaucoma procedure was combined with cataract surgery, the percentages were as follows: trabeculectomy with MMC, 24%; MIGS, 22%; and GDD, 9%.1
The survey also found that iStent and Trabectome were the most commonly used MIGS procedures in 2016.
Debating the Role of MIGS
Although multiple studies have associated MIGS with a favorable safety profile and modest efficacy, others cite a lack of evidence in proving the effectiveness of these techniques.
In response to “good, healthy skepticism” from some quarters, Dr. Ahmed said that “MIGS have been very well studied for many years, with a wealth of published data.” He emphasized that MIGS are not designed to replace trabeculectomy in advanced glaucoma. Rather, “Surgeons are using MIGS procedures in their mild to moderate patients who need lower intraocular pressure (IOP) but in whom they are reluctant to operate because of the side effects associated with trabeculectomy.”
A MIGS Primer
(See Web Extra for images)
MIGS procedures share 5 key characteristics1:
- Ab interno microincision through a clear corneal approach, allowing MIGS to be performed easily in conjunction with cataract surgery; providing a direct view of the angle; and avoiding conjunctival scarring, in case later glaucoma surgery is required.
- Minimal trauma, maintaining normal ocular anatomy and function as much as possible.
- At least modest efficacy, making them a reasonable option in selected patients.
- Favorable safety profile, avoiding the serious complications seen with traditional surgeries, including bleb infections, hypotony, and corneal decompensation.
- Rapid recovery, reducing the impact on patients’ quality of life.
Implanted MIGS
Stent devices fall into 3 main categories:
- Increasing trabecular outflow:
- iStent (Glaukos). Implanted in the trabecular meshwork, the stent allows aqueous humor to flow from the anterior chamber into Schlemm’s canal (FDA approved in 2012).
Glaukos recently received approval for a pivotal U.S. trial of the iStent SA system (consisting of 2 stents in a single inserter) as a standalone procedure in pseudophakic patients.
- Hydrus Microstent (Ivantis). Described as an intracanalicular scaffold, this 8-mm-long device is inserted into Schlemm’s canal to establish outflow (approved in Europe but not in the United States or Canada).
- Targeting the suprachoroidal space:
- CyPass Micro-Stent (Alcon). This device, implanted in the supraciliary space, allows suprachoroidal aqueous outflow (FDA approved in 2012).
- Opening a subconjunctival filtration pathway:
- XEN 45 Gel Stent (Allergan). This soft, collagen-derived gel device creates a new pathway for aqueous flow from the anterior chamber into an ab interno bleb in the subconjunctival space (FDA approved in 2016).
Nonimplant MIGS
- Trabectome (NeoMedix). Electrocautery, irrigation, and aspiration are used to selectively ablate the trabecular meshwork and the inner wall of Schlemm’s canal to allow aqueous free access to the canal and its collector channels (FDA approved in 2004).
- Kahook Dual Blade (New World Medical). This relatively inexpensive single-use disposable handpiece employs 2 parallel blades to remove a strip of trabecular meshwork to improve outflow, without need for an expensive electrocautery or irrigation/aspiration system.
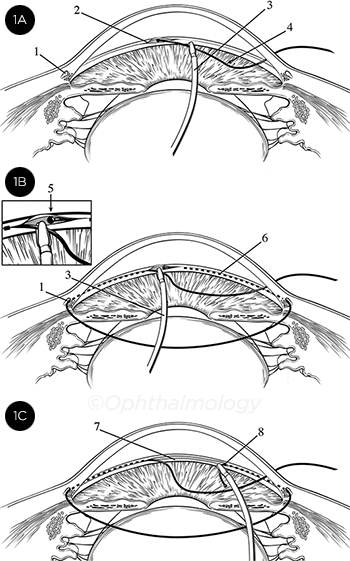
- Gonioscopy-assisted transluminal trabeculotomy. GATT is a minimally invasive ab interno circumferential trabeculotomy (see Fig. 1) that is performed through two 1.0-mm corneal incisions and employs either a microcatheter, 5-0 Prolene suture, or TRAB 360 handpiece (Sight Sciences). After cannulation, the entire trabecular meshwork is unroofed.
- Ab interno canaloplasty (ABiC). The procedure, performed through a single self-sealing clear corneal incision, involves 360-degree viscodilation of the canal using either the iTrack microcatheter (Ellex) or the VISCO360 (Sight Sciences) handpiece and an ophthalmic viscoelastic device inserter.
- Endoscopic cyclophotocoagulation (ECP). An endoscopic probe is inserted via a corneal or pars plana incision to ablate a selected portion of the ciliary epithelium under direct endoscopic visualization. This process decreases aqueous production.
___________________________
1 Saheb H, Ahmed IK. Curr Opin Ophthalmol. 2012;23:96-104.
|
The Case for MIGS
Glaucoma specialist John P. Berdahl, MD, with Vance Thompson Vision in Sioux Falls, South Dakota, said he considers the entire range of MIGS options when tailoring treatment for his glaucoma patients.
Fitting the procedure to the patient. “It is my duty to fit the procedure to the patient,” Dr. Berdahl noted. “That being said, when you are first starting out with MIGS, it is good to get comfortable with one procedure and then expand out to other procedures because there are a lot of similarities.” He added that the learning curve for a MIGS procedure is between 10 and 20 cases.
Dr. Berdahl discussed his 3-year results from patients who had an iStent implanted in combination with cataract surgery.2 “This approach effectively lowered IOP in open-angle glaucoma (OAG) patients from a mean of 19.13 ± 6.34 mm Hg to 15.17 ± 3.53 mm Hg after 2 years,” he said. “Interestingly, we found that the magnitude of IOP reduction was more significant in patients with higher preoperative pressure and also that medication use was significantly reduced.”
An “early adopter’s” experience with iStent. Glaucoma specialist Mark J. Gallardo, MD, of El Paso Eye Surgeons in Texas, is an “early adopter” of several MIGS procedures, an interest fueled by his desire to provide patients with the most advanced technology, especially if it proves safer, with a quicker recovery.
Dr. Gallardo believes that the modest results from the early clinical trials of the iStent do not reflect the full potential of this approach, as the trial investigators were the first in the world to use the device in a clinical setting and had minimal experience in the best placement. He noted that 70% of the stents were implanted by surgeons who had performed 5 or fewer procedures.
“The learning curve, as well as the previous lack of knowledge on how to maximally manipulate the outflow system with targeted stent implantation, adversely impacted the data,” he said.
Placement and patients. Ultimately, surgeons, including Dr. Gallardo, learned that targeting areas adjacent to collector channels could enhance the efficacy of the stent. Intraoperative visual cues, such as increased regurgitation of blood (blotching) within Schlemm’s canal or increased areas of pigmentation on the posterior trabecular meshwork, help to highlight the location of patent collector channels.
“This, coupled with our identification of ideal candidates for the procedure—those already on 1 to 3 glaucoma drugs with IOP targets in the mid-teens range—have led to results superior to those of the pivotal trial, and subsequent research has demonstrated the safety and effectiveness of the trabecular microbypass stent [iStent],” he said.
Case series shows benefits. Dr. Gallardo conducted a retrospective case series in a predominantly Hispanic patient population with moderate to severe glaucoma to assess reduction of IOP and/ or medication burden at 12 months following implantation of 1 trabecular microbypass stent during cataract surgery.3
Results of the entire cohort showed that IOP was reduced from 16.5 mm Hg preoperatively to 12.9 mm Hg, and the mean number of medications decreased from 2.3 to 0.9.
Reducing medication burden. “At 12 months, 94% of all eyes achieved their predefined treatment goal of reduced IOP and/or medications,” Dr. Gallardo noted. Among patients who had medically controlled glaucoma entering cataract surgery plus stent implantation, 69% were able to completely eliminate their need for medications. Of this latter group, 55% had been on 3 or more medications preoperatively, he said.
“The earlier we use MIGS, the less we need to rely on medications,” Dr. Gallardo said.
He noted several benefits from reducing a patient’s medication burden: increased compliance, lower monthly costs (generic drugs are skyrocketing in price), and less exposure to the preservative benzalkonium chloride, which has been associated with ocular surface disease and damage to endothelial cells in the trabecular columns.
 |
|
KEY STEPS IN GATT. (1A) Initial cannulation of Schlemm’s canal. (1B) The microcatheter (or suture) has been passed 360 degrees around the canal. (1C) The distal tip of the catheter/suture has been retrieved and is being externalized, creating the circumferential trabeculotomy. KEY: 1, Schlemm’s canal (SC); 2, initial goniotomy site; 3, microsurgical forceps; 4, either the microcatheter or suture; 5, distal end of the catheter/suture after it has been passed around SC; 6, path of the catheter/ suture within SC; 7, trabecular shelf created by this procedure; 8, trabeculotomy resulting from GATT.
|
ABiC: Impact on Practice
Dr. Gallardo also performs ab interno canaloplasty (ABiC), or transluminal viscodilation, which aims to improve outflow by dilating Schlemm’s canal 360 degrees through a small corneal incision, using a microcatheter and viscoelastic.
Pearls for ABiC. He makes the temporal clear corneal wound directly across from the nasal angle with side-port incisions at a 90-degree angle. He recommends avoiding the limbal vessels as much as possible, as surface bleeding can stain the viscoelastic used as a coupling agent for the gonioscope and obstruct the view of the drainage angle.
Dr. Gallardo uses the iTrack catheter, which, he said, “provides tactile feedback on the patency or health of the canal during circumnavigation, while the illuminated tip allows me to track the catheter’s movement, providing me assurance that I am actually in the canal and not in the suprachoroidal space.”
What about moderate to severe glaucoma? Dr. Gallardo said that the advent of MIGS has had an enormous impact on his practice, not only in his treatment of patients with mild glaucoma but also in those with moderate to severe glaucoma.
“Whether [they are] performed as a standalone procedure or as an adjunct to cataract surgery, I have found these microinvasive procedures very effective at meeting my patients’ needs,” he said. “In patients requiring further reduction in IOP, I try a MIGS procedure or a combination of MIGS procedures before filtering, in most but not all circumstances.” He added, “I was doing 8 to 10 filters a week, and now I perform 1 ab externo filtration every 4 to 6 weeks.”
 |
|
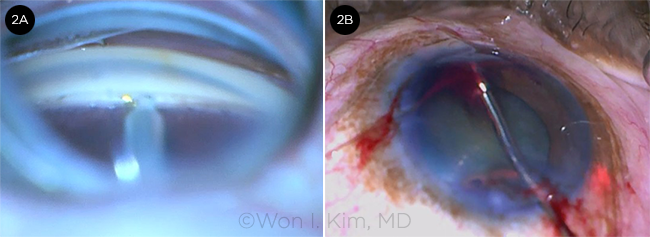
TRABECTOME + ABIC. (2A) Trabectome is used to remove a sector of trabecular meshwork. (2B) ABiC is performed through the Trabectome’s ablation zone.
|
GATT: A New Twist on Trabeculotomy
While ABiC is a minimally invasive approach to canaloplasty, gonioscopy-assisted transluminal trabeculotomy (GATT) is a minimally invasive modification of standard trabeculotomy.
“When I was a medical student, I felt that trabs and tubes seemed really harmful to the eye, and I kept questioning why we were doing what we were doing,” said glaucoma specialist Davinder S. Grover, MD, MPH, of the Glaucoma Associates of Texas in Dallas.
“I started practice right around the time the iStent was being investigated,” Dr. Grover added. “My partners and I were primary investigators on the CyPass microstent, Hydrus, and XEN gel stent. Additionally, Dr. Ronald L. Fellman and I were developing techniques of our own (ab interno bleb revision and ab interno Ex-PRESS shunt removal), and all this research converged to provide a productive environment for our own innovations.”
Development of GATT. Drs. Grover and Fellman (along with their colleagues Drs. David Godfrey and Oluwatosin Smith) developed the GATT procedure, an ab interno circumferential trabeculotomy that is performed through 2 corneal incisions, 1 mm each. A small goniotomy is created, and a microcatheter or 5-0 Prolene suture is used to cannulate 360 degrees of Schlemm’s canal and then unroof the entire trabecular meshwork (Fig. 1).
Dr. Grover said a major advantage of this procedure is that the entire drainage system is accessed, rather than just a small portion. In addition, it spares conjunctival tissues from incision and scarring, allowing better outcomes if traditional glaucoma surgery is required later.
Findings from 2 studies. In the first study, “we looked back at our 2-year data on 10 patients (14 eyes) under 30 years old with a dysgenic anterior segment angle and uncontrolled primary congenital glaucoma or juvenile open-angle glaucoma who underwent GATT,” Dr. Grover said. “They experienced a mean decrease in IOP from 27.3 to 14.8 mm Hg and a mean decrease in meds from 2.6 to 0.86.4
“Moreover, when we evaluated GATT outcomes in 198 patients with POAG and secondary open-angle glaucoma (SOAG), either isolated or combined with cataract surgery, we found very encouraging results that were similar to if not better than previously published data on ab externo circumferential trabeculotomy,” Dr. Grover said.
The patients in this study with POAG had an average IOP decrease of 9.2 mm Hg (a mean reduction of 37.3%) at 24 months, with an average decrease of 1.43 glaucoma medications.
At that same time point, the SOAG patients had an average decrease in IOP of 14.1 mm Hg (a mean reduction of 49.8%) on an average of 2.0 fewer medications.5
Learning from failure. While Dr. Grover was pleased with the results, he gained greater insight from the treatment failures. In the POAG group, there was a correlation between mean deviation (MD) in visual field defect parameters and outcomes: Patients with a worse MD had a higher chance of treatment failure in the first 3 months.
“This is suggestive of the health of the eye’s inherent drainage system,” Dr. Grover said. “Since it is difficult to visualize the collector channels and episcleral vasculature, we searched for other indicators that would serve as a proxy to determine the patency of the outflow system.”
The wave as an indicator. In seeking such a proxy, Drs. Fellman and Grover drew on their experience with an earlier MIGS, Trabectome. During that type of surgery, they had observed a nasal perilimbal and/or episcleral vessel wave of fluid adjacent to the trabeculotomy site. “We believe this fluid wave, which we named an episcleral venous fluid wave (EVFW), signifies intraoperative structural patency of the conventional outflow system and is a sign that the collector system is at least anatomically functional.”6
Dr. Grover said the presence or absence of this EVFW could be a prognostic indicator for success after a Trabectome surgery. In a study of 68 eyes of 49 patients with glaucoma who underwent phaco plus Trabectome or Trabectome alone, the eyes with a poorly defined EVFW had a higher likelihood of further glaucoma surgery.7
He uses the EVFW as a prognostic sign for GATT as well. “When I perform a GATT and I see an EVFW, I am very optimistic about that surgery.”
 |
|
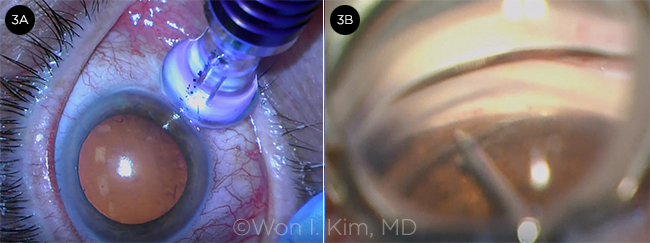
TSCPC + ABIC. (3A) After micropulse TSCPC is completed, (3B) ABiC is performed.
|
Combined MIGS Procedures
One of the hot topics at the American Glaucoma Society’s 2017 annual meeting was combining MIGS procedures. Won I. Kim, MD, a glaucoma specialist with Walter Reed National Military Medical Center in Bethesda, Maryland,* gave a presentation suggesting that multiple MIGS procedures can be successfully combined without significant additional risk.
Dr. Kim said, “Because of their relatively modest efficacy, MIGS procedures have traditionally been limited to mild to moderate disease; but perhaps combined MIGS procedures, with their potentially improved efficacy, can be extended to include those with severe disease.”
Mix-and-match MIGS. Dr. Kim has been mixing and matching MIGS procedures, based on specific patients’ needs. One of these combinations is ab interno trabeculectomy plus ABiC.
“My approach was removing a section of trabecular meshwork with the Trabectome or Kahook Dual Blade and then viscodilating the rest of Schlemm’s canal 360 degrees with the iTrack,” Dr. Kim said (Fig. 2). “This could take advantage of the different mechanisms of both sectoral trabecular meshwork removal and canaloplasty while simultaneously addressing their weaknesses,” such as the limited sectoral aspect of Trabectome and the residual trabecular meshwork resistance after canaloplasty.
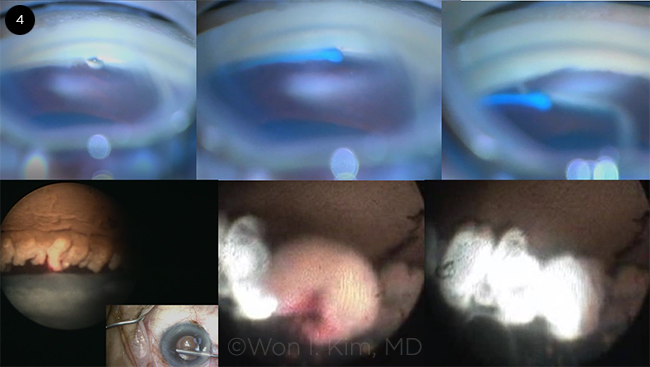
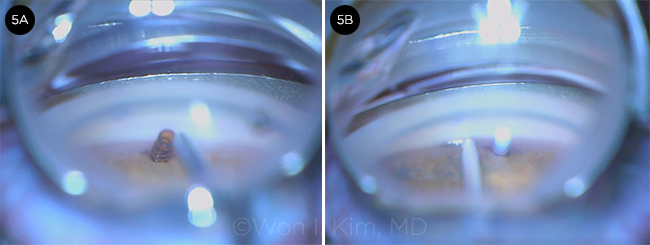
He has also successfully used several other multi-technique approaches. These include ABiC combined with micropulse transscleral cyclophotocoagulation (TSCPC; Fig. 3), trabecular meshwork bypass (using either iStent, GATT, or Trabectome) combined with endoscopic cyclophotocoagulation (ECP; Fig. 4), and CyPass supraciliary stent combined with trabecular meshwork bypass (using iStent or Kahook Dual Blade; Fig. 5).
He said that in his experience, all of these techniques have shown the ability to lower IOP into the low-teens range, reduce medication burden, maintain an excellent safety profile, and allow for rapid visual recovery.
Multi-MIGS plus phaco. Dr. Berdahl is also an advocate of combined MIGS. He compared the outcomes of combined microbypass stent implantation, cataract extraction, and ECP to those obtained with just the microbypass stent and concomitant cataract surgery in patients with OAG.8
He found that patients who underwent the combined approach experienced a mean IOP reduction of 7.14 mm Hg compared with 4.48 mm Hg in the control patients who did not have ECP. He found that the combination procedure was also effective in patients with severe OAG.
“The combined approach makes sense,” Dr. Berdahl said. “We are trying to avoid the morbidity of more aggressive glaucoma surgeries. The question then becomes whether the efficacy is good enough. My approach is safety first and efficacy second. I will try the MIGS first.”
 |
|
GATT + ECP. (4) TRAB 360 handpiece is used to perform GATT, which is followed by ECP.
|
MIGS Caveats
Steven L. Mansberger, MD, MPH, of the Devers Eye Institute in Portland, Oregon, has closely watched the advent of MIGS. He expressed concerns in the areas of efficacy and costs.
“As a glaucoma specialist, I am always interested in finding new ways of lowering pressures safely and effectively, and I applaud the investigators in this space,” Dr. Mansberger noted.
“That being said,” he added, “MIGS may be useful for some patients, but traditional surgeries are required by the vast majority of surgical glaucoma patients, and it is imperative that we continue to learn the ins and outs of trabs and tubes in our glaucoma fellowships.”
Efficacy concerns. Dr. Mansberger pointed out that some MIGS such as the iStent make only a millimeter of difference in IOP, and “we do not understand the characteristics of patients who most benefit from MIGS above and beyond cataract surgery alone.”
Dr. Mansberger recounted a number of earlier implant devices that had failed, including several limbal and suprachoroidal stents in the 1950s and 1960s, and a XEN-like implant in the 1980s.9
“We have new modifications such as mitomycin C [e.g., with XEN implants],” he added. “But time will tell if these new MIGS will be more effective or if history will repeat itself.”
Cost factors. The downside of many of the MIGS surgeries is their cost, Dr. Mansberger pointed out. MIGS such as iStent, XEN, and Trabectome can add up to $4,000 to the cost of cataract surgery alone when factoring in surgeon charges, device costs, anesthesia costs, and surgical center fees. “We need to look at changing the cost-benefit ratio in terms of costs,” Dr. Mansberger said.
On the other hand, he noted that the GATT procedure is one of the most cost-effective MIGS—it can be done using a $5 suture. “And Dr. Grover has shown good results 2 to 3 years out, making this the approach to watch,” Dr. Mansberger said.
Multiple surgeries? He also has concerns about patients who may need subsequent surgeries if a MIGS procedure does not effectively lower IOP.
“In most patients we see who can’t use their drops or who have severe glaucoma, we do a traditional procedure, and we only need to operate one time to treat the problem. That is preferable to multiple surgeries,” he said.
“Down the road, MIGS will be considered based on cost, IOP response, and visual field outcomes,” said Dr. Mansberger. “Through the efforts of these MIGS pioneers, we will learn more about how to better treat glaucoma. We don’t have the perfect MIGS yet, but in the end, we will get there.”
 |
|
CYPASS + ISTENT. (5A) CyPass is placed in the supraciliary space. (5B) iStent-L is placed, followed by an iStent-R facing in the opposite direction (not shown). The 2 iStents and a CyPass allow aqueous outflow through multiple pathways.
|
The Next Phase: Sustainability
Dr. Ahmed expressed a similar view regarding future developments. He said that while it has been greatly satisfying to be involved in the early innovation process and see a large number of MIGS procedures “go mainstream,” he had not fully anticipated the need for research on their cost-effectiveness and appropriate utilization.
Reimbursement challenges. “We are currently recognizing and building the right studies to look at quality-of-life issues, helping payers in the United States and Canada understand why MIGS should be funded a certain amount,” Dr. Ahmed said. “We are looking at recovery, number of postsurgical visits, return to vision, and refractive changes—metrics that are of concern to entities funding these procedures.”
Dr. Berdahl also pointed to reimbursement as one of the biggest challenges to widespread MIGS adoption. “MIGS is one of the only really impressive innovations in glaucoma in the last few decades. It will be tremendously sad if it withers on the vine,” he said, for lack of reimbursement and resources to support innovation.
___________________________
*The views expressed are those of the individual and do not reflect the official policy or position of the Department of the Army, Department of Defense, or the U.S. government. Any mention of products is for informational purposes only and should not be considered an endorsement.
___________________________
Be sure to check out “MIGS Reimbursement,” a Web Extra for February’s Feature.
___________________________
1 Vinod K et al. J Glaucoma. 2017;26(8):687-693.
2 Ferguson TJ et al. Clin Ophthalmol. 2016;10:1767-1773.
3 Gallardo MJ et al. Clin Ophthalmol. 2016;10:1931-1937.
4 Grover DS et al. Br J Ophthalmol. 2015;99(8):1092-1096.
5 Grover DS et al. Gonioscopy-assisted transluminal trabeculotomy: an ab interno circumferential trabeculotomy midterm follow-up. Paper presented at: annual meeting of the American Academy of Ophthalmology; Nov. 17, 2015; Las Vegas.
6 Fellman RL, Grover DS. J Glaucoma. 2014;23(6):347-350.
7 Fellman RL et al. Opthalmology. 2015;122(12):2385-2391.
8 Ferguson TJ et al. J Cataract Refract Surg. 2017;43(3):377-382.
9 Lim KS et al. Br J Ophthalmol. 1998;82(9):1083-1089.
A Comprehensive Ophthalmologist’s Perspective on MIGS
Do MIGS have a place in the comprehensive ophthalmologist’s armamentarium? Absolutely, according to Jeffrey Whitman, MD, a comprehensive ophthalmologist at the Key-Whitman Eye Center in Dallas.
“In our cataract patients with mild to moderate glaucoma, if we can get them off even one of their eyedrops by utilizing a MIGS approach, then we should offer this alternative,” Dr. Whitman said. “One eyedrop may not seem like much, but over the course of a lifetime, that adds up to significant savings and greatly impacts the quality of life.”
iStent, CyPass, and beyond. Dr. Whitman has been inserting iStents for the past 3 years and, within the last year, has begun using the CyPass. “In my early experience, I find the CyPass somewhat easier to insert, and I am obtaining much lower pressures than I could have imagined,” he said.
He believes that the field will continue to advance, perhaps with combinations of MIGS or the addition of medications to stents for more potent treatment. (Glaukos and other companies are investigating these possibilities.)
MIGS myths limiting usage. Dr. Whitman observed that misconceptions about the effectiveness of MIGS procedures, the learning curve, and the time MIGS adds to cataract surgery are preventing many comprehensive ophthalmologists from adopting this approach.
“I encourage comprehensive ophthalmologists not to give up on MIGS but rather to reach out to other ophthalmologists and learn better techniques,” Dr. Whitman said. “In terms of time, it makes an efficient cataract surgery take up to 50% longer, but I believe the benefits to the patient are well worth it.”
Getting comfortable with MIGS. He added that getting accustomed to using a gonioscopic lens to view the angle and keeping the magnification at 9× (higher magnification results in less depth of field) will help comprehensive ophthalmologists become more comfortable with learning to do MIGS.
“My real take-home message to my colleagues is to get on the bandwagon with MIGS,” Dr. Whitman said. “It doesn’t add much time to cataract surgery, and it provides great benefit to your patient.”
|
One Patient’s ABiC Experience
Mark J. Gallardo, MD, recounted the story of a prominent ophthalmologist from Australia who came to El Paso for bilateral ABiC. Although his glaucoma was controlled with medications, he experienced painful adverse effects from the drugs. He had constant eye irritation that was so debilitating that he was planning to retire.
“Following treatment with the iTrack microcatheter, he was able to stop his medications, while maintaining control of his pressure,” Dr. Gallardo said. “He has had a renewed vigor for life and has continued to practice medicine. Without MIGS, he would have retired.”
|
Meet the Experts
Iqbal Ike K. Ahmed, MD Glaucoma specialist at the Prism Eye Institute in Mississauga, Ontario, Canada, assistant professor at the University of Toronto, and clinical professor at the University of Utah. Financial disclosures: Abbott Medical Optics: C,L,S; Aerie Pharmaceuticals: C; Alcon: C,L,S; Allergan: C,L,S; ArcScan: C; Bausch + Lomb: C; Carl Zeiss Meditec: C,L,S; Centervue: C; Clarity: C,S; Envisia: C; Equinox: C; Eyelight: C; ForSight Labs: C; Glaukos: C,O,S; Gore: C; Iantech: C; InjectSense: C; InnFocus: C; Iridex: C; iStar: C; Ivantis: C,L,S; LayerBio: C; Leica: C; New World Medical: C,S; Oculus: C; Omega Ophthalmics: C; PolyActiva: C; Sanoculis: C; ScienceBased Health: C; SOLX: C; Stroma: C; Transcend Medical: C; TrueVision: C.
John P. Berdahl, MD Glaucoma specialist at Vance Thompson Vision in Sioux Falls, S.D. Financial disclosures: Abbott: C; Alcon: C,L; Allergan: C,L; Avedro: C; Bausch + Lomb: C; Calhoun Vision: C; Clarvista: C; Digisight: C,O; Envisia: C; Equinox: C,O; Glaukos: C,O; Imprimis: C,P; Ocular Surgical Data: C,O; Ocular Theraputix: C; Omega Ophthalmic: C,O; Vittamed: C.
Mark J. Gallardo, MD Glaucoma specialist at El Paso Eye Surgeons in Texas. Relevant financial disclosures: Alcon: L; Ellex: C,L; Glaukos: C,L; New World Medical: S.
Davinder S. Grover, MD, MPH Glaucoma specialist at Glaucoma Associates of Texas in Dallas. Relevant financial disclosures: Allergan: C,L; New World Medical: C,L; Reichert: C,L.
Won I. Kim, MD Glaucoma specialist with Walter Reed National Military Medical Center and professor of surgery at Uniformed Services University of the Health Sciences (USUHS), both in Bethesda, Md.* Relevant financial disclosures: None.
Steven L. Mansberger, MD, MPH Senior scientist, vice-chair, and director of Glaucoma Services at the Devers Eye Institute at Legacy Health in Portland, Ore. Adjunct professor of ophthalmology and clinical assistant professor of Public Health and Preventive Medicine at Oregon Health & Science University (OHSU). Relevant financial disclosures: Santen: C; New World Medical: C.
Jeffrey Whitman, MD President and chief surgeon of the Key-Whitman Eye Center in Dallas. Relevant financial disclosures: Glaukos: C,L; Ivantis: C.

|
Web Extras