By Chris McMillan, Kay T. Khine, MD, and Nathan W. Blessing, MD
Edited by Bennie H. Jeng, MD
Download PDF
Floppy eyelid syndrome (FES) is characterized by an easily everted upper eyelid due to underlying tarsal plate laxity and an associated chronic, reactive papillary conjunctivitis. Although FES is being recognized more frequently as a result of heightened awareness among physicians, it often remains undetected until multiple conservative treatment regimens have failed. In recent years, FES has reemerged as a topic of interest in ocular surface disease because of its systemic and ocular associations with obstructive sleep apnea (OSA) and keratoconus, respectively.
The prevalence of FES in the adult population has been reported to range from 3.8% to 15.8%, although this is likely an underestimate.1 FES was originally described in a population of obese men between 45 and 55 years of age. More recent studies, however, have described FES in nonobese populations of women and men of all ages.2
Associations of Interest
Obstructive sleep apnea. Several studies have reported a nearly 100% prevalence of OSA among FES patients, while other authors have presented more conservative estimates.1 Regardless, all of the literature has found a statistically significant association between FES and OSA.3 This association—with an odds ratio of 12.5—persists even when controlled for obesity and other confounding factors.4 Although the prevalence of OSA is high among FES patients, only an estimated 4% to 16% of OSA patients met the criteria for FES; an additional 61% presented with at least eyelid hyperlaxity.5
Keratoconus. The relationship between FES and keratoconus has been well documented. It was first reported in a longitudinal study, which revealed that over a 10-year period, 18% of FES patients developed clinical or subclinical keratoconus in the eye on the side they slept on most frequently.6
Subsequent case-control studies also revealed a significantly increased prevalence of keratoconus among FES patients, yielding an odds ratio of 19.3.4 Interestingly, the corneas of keratoconus patients were found to have increased levels of an elastin variant, oxytalan. Increased levels of this same variant are also found in eyelid tissue samples from FES patients.1
Pathophysiology
Although the mechanisms underlying FES are poorly understood, several plausible theories have been proposed regarding the development of this condition.
Histopathologic findings. Numerous studies have shown decreased levels of mature elastic fibers within the tarsal plate and eyelid skin in patients with FES.7 This is believed to be the result of increased elastolytic proteases in these regions. Rather than mature elastic fibers, FES patients paradoxically have increased levels of a stiff elastin variant, oxytalan, in their floppy lid and tarsal plate. This stiff variant is characteristically found in tissues subjected to recurrent mechanical trauma, which supports the initial and most commonly cited theory of FES development.
Mechanical theory. The mechanical theory hypothesizes that easily everted eyelids allow exposure and mechanicalirritation of the tarsal conjunctiva and ocular surface.1 This ultimately results in chronic inflammation of the conjunctiva and other characteristic features of FES. This model is supported by the association of FES with keratoconus and OSA. Keratoconus is similarly associated with corneal trauma such as excessive eye rubbing. Thus, it would be expected to be more common in a syndrome caused by recurrent mechanical stress.
Proponents of the mechanical theory also point to the association between FES and OSA. Patients with OSA characteristically have depressed central nervous system reflexes to noxious stimuli while asleep and do not reflexively reposition themselves.4 Thus, FES patients with easily everted eyelids are more likely exposed to recurrent trauma while sleeping on either their side or prone. This has been further supported by studies examining sleep behavior of patients with FES and OSA, which found the affected eye is usually on the preferred sleeping side.6
Critics of the mechanical theory raise objections to the model’s explanation for corneal and conjunctival changes. The skepticism often arises from studies demonstrating bilateral corneal and conjunctival involvement even in instances of unilateral lid eversion.1 These corneal findings, specifically, led to the theory of local ischemia and reperfusion.
Local ischemia and reperfusion. The theory of local ischemia and reperfusion postulates that these factors are the root cause of connective tissue changes in the lids of FES patients. The ischemia results from a prone or side sleeping position, as well as intermittent hypoxia due to OSA.6 Reperfusion then occurs upon awakening, which produces oxidative stress and free radical damage. This damage is believed to cause degenerative changes in the eyelid as well as in the cornea, which would then explain the association with keratoconus.
While this theory may be convincing to many, critics express skepticism about the occurrence of ischemia in a remarkably well-vascularized region such as the face. Additionally, the tarsal plate has low oxygen demands. The model does, however, fit well with cases of FES associated with OSA.
 |
|
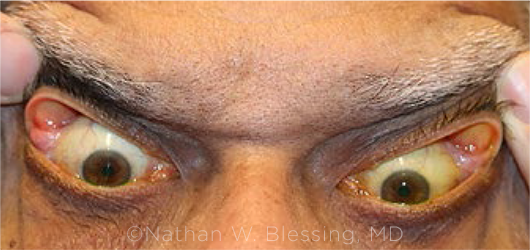
CLINICAL PRESENTATION. Patient’s lids demonstrate characteristic eversion following gentle traction as seen in floppy eyelid syndrome.
|
Clinical Features
FES is characterized by horizontally lax eyelids that can be easily everted with minimal lateral traction and an absence of tarsal rigidity (see “Clinical Presentation”). The frequent eversion results in a chronic papillary conjunctival reaction in response to exposure and irritation. Additionally, patients may present with lateral lid imbrication and lid ptosis.2
Classic presentation. Patients often present with nonspecific findings of chronic ocular surface irritation and inflammation producing redness, photophobia, and a foreign body sensation. Mucoid discharge is also frequently noticed.1 Symptoms often have been present for years and are most severe in the morning upon awakening. The affected side is most commonly the side on which the patient sleeps.
Several common ocular associations include blepharitis, ectropion, entropion, and lid ptosis. Lash ptosis is strongly associated with FES and should immediately raise clinical suspicion in the appropriate setting. A substantial proportion of FES patients also present with corneal involvement, including exposure keratopathy and keratoconus.6 Finally, FES has historically been associated with a common clinical phenotype of a patient with high body mass index along with OSA.
Diagnosis
The diagnosis of FES is typically based on a clinical exam demonstrating easy eversion of the eyelids with lateral traction, along with papillary conjunctivitis. Clinical suspicion should be raised if symptoms have been chronic, recurrent, or resistant to conservative management. Suspicion is additionally heightened in patients with comorbid conditions such as OSA.3
Because FES is a clinical diagnosis, it is recommended that patients with chronic irritation of the ocular surface receive lid laxity screening in their comprehensive ophthalmic evaluations. In patients diagnosed with FES, further investigation for associated conditions should be performed. This includes a slit-lamp examination to assess for keratoconus as well as papillary conjunctivitis. Although additional findings are not necessary for diagnosis, associated findings such as lid malposition or lid ptosis can be strongly supportive.
Differential diagnosis. Other conditions featuring eyelid hyperlaxity can present similarly to FES, including blepharoptosis, blepharochalasis, and dermatochalasis.8 There are also conditions associated with FES, such as entropion or ectropion, that can present with ocular surface inflammation and lid hyperlaxity but without other characteristic features of FES. Finally, the chronic conjunctivitis of FES must be distinguished from other forms of chronic ocular surface irritation such as allergic conjunctivitis, atopic conjunctivitis, giant papillary conjunctivitis, blepharitis, ocular rosacea, and superior limbic keratoconjunctivitis.
Management
Conservative measures. Management of FES should initially focus on reestablishing proper eyelid position and preventing further corneal exposure. Measures include topical lubricants in addition to use of eye shields or humidity goggles while sleeping. Lid taping has also been used as a means of preventing eversion.
However, such localized conservative therapies are frequently inadequate.1 For patients with concomitant obstructive sleep apnea, continuous positive air pressure (CPAP) devices and weight loss can lead to significant improvement of both OSA and ocular symptoms.8 Patients without a formal OSA diagnosis should be referred to an internist or sleep specialist for further evaluation.
Surgical measures. If symptoms remain refractory, surgical interventions are quite effective.9 Surgical solutions focus on tightening the upper lid to prevent repeated eversion. Techniques include horizontal tightening via a lateral tarsal strip procedure, medial canthal and/or lateral canthal plication, and full-thickness wedge excision.10 All surgical solutions have been shown to provide symptomatic improvement as well as decreased conjunctival inflammation.
Conclusion
Floppy eyelid syndrome is an underrecognized cause of chronic ocular discomfort and can manifest in a variety of clinical phenotypes. FES should be considered in patients with symptoms of inflammation, lid malposition, and lid hyperlaxity that are refractory to conservative measures. The pathophysiology of FES may involve a combination of repetitive mechanical trauma along with local ischemia and reperfusion. FES is a clinical diagnosis, and suspicion should be raised in patients with OSA, upper lid and lash ptosis, and other associated ocular conditions. Conservative management should be attempted, but surgical correction is often required for definitive treatment. Among surgical interventions, a lateral tarsal strip procedure or a wedge excision is a viable treatment option.
___________________________
1 Salinas R et al. Ocul Surf. 2020;18(1):31-39.
2 Miyamoto C et al. Arq Bras Oftalmol. 2011;74(1):64-66.
3 Wang P et al. J Ophthalmol. Published online June 5, 2016. doi:10.1155/2016/6980281.
4 Ezra DG et al. Ophthalmology. 2010;117(4):831-838.
5 Muniesa MJ et al. Br J Ophthalmol. 2013;97(11):1387-1390.
6 Culbertson WW, Tseng SC. Cornea. 1994;13(1):33-42.
7 Schlötzer-Schrehardt U et al. Ophthalmology. 2005;112(4):694-704.
8 McNab AA. Ophthalmic Plast Reconstr Surg. 1997;13(2):98-114.
9 Burkat CN, Lemke BN. Ophthalmic Plast Reconstr Surg. 2005;21(1):52-58.
10 Ezra DG et al. Ophthalmology. 2010;117(4):839-846.
___________________________
Mr. McMillan is a fourth-year medical student at the Brody School of Medicine at East Carolina University in Greenville, N.C. Dr. Khine is a voluntary assistant professor of ophthalmology at University of Miami Bascom Palmer Eye Institute in Miami, and a glaucoma specialist at Focus Eye Center in Pasadena, Calif. Dr. Blessing is an oculoplastic specialist and assistant professor of clinical ophthalmology at University of Miami Bascom Palmer Eye Institute in Palm Beach Gardens, Fla. Financial disclosures: None.
Got Pearls?
Interested in sharing with your colleagues the latest evidence-based information about the diagnosis and medical and/or surgical management of a specific disease entity? Write a Pearls article for EyeNet!
Be sure to read the writers guidelines, available at aao.org/eyenet/write-for-us. Then submit your Ophthalmic Pearls topic proposal to eyenet@aao.org.
|