Download PDF
Many early-onset ocular disorders are genetic, yet pediatric ophthalmologists lack a solid understanding of genetic disorders and how to approach them, according to a survey conducted by the American Association for Pediatric Ophthalmology and Strabismus (AAPOS) Genetic Eye Disease Task Force.1
“No one can know everything,” conceded task force chair Arlene V. Drack, MD, at the University of Iowa in Iowa City. “But pediatric ophthalmologists should be able to recognize a disorder as potentially genetic and either pursue a workup or refer to the appropriate subspecialist.”
The 16-question survey, which was emailed to 1,489 AAPOS members, focused on physicians’ ability to understand and use genetic tests and to counsel patients. While most respondents (93%) reported caring for children with genetic eye disorders on a weekly basis, nearly half (48%) reported no understanding of genetic testing modalities. A majority (81%) described themselves as “a little or not at all comfortable” explaining genetic test results to patients. Of those who order testing, 90% work with a genetic counselor.
The good news: Most respondents appeared eager to learn more about testing modalities, citing interest in continuing education.
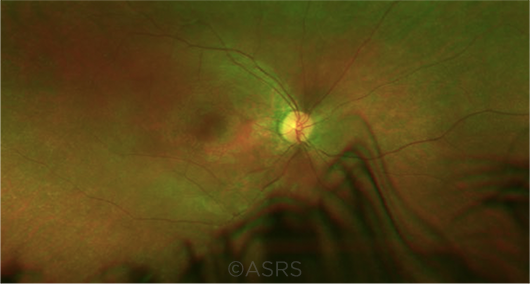 |
EARLY DX. Nystagmus in an infant, along with visual loss, should raise suspicion for Leber congenital amaurosis (shown here). An FDA-approved therapy is now available for selected cases. This image was originally published in the ASRS Retina Image Bank. Audina M. Berrocal, MD. Leber Congenital Amaurosis. Retina Image Bank. 2019; Image Number 28844. © The American Society of Retina Specialists.
|
Missed diagnoses? Despite two email reminders, the survey response rate was only 18%. The researchers suspect that the nonresponders may think they don’t see patients with genetic disorders, so a survey about genetic eye disease does not apply to them. But, said cochair Virginia Miraldi Utz, MD, at the University of Cincinnati, “most likely, they are seeing at least one genetic eye disease patient a week, but they may not realize that the underlying cause of that patient’s problem is genetic.”
What you don’t know … Most pediatric ophthalmologists have not been trained to do genetic testing for congenital/infantile nystagmus, infantile/juvenile cataracts, pediatric glaucoma, and congenital malformations. And this is occurring at a time when novel gene-based diagnostic strategies2 and therapies continue to emerge.
“It is not common for children to have serious eye disorders,” Dr. Drack said. Thus, when such disorders present, she urged ophthalmologists to have a high level of suspicion for a genetic cause. “We won’t find what we don’t look for.”
—Miriam Karmel
___________________________
1 Drack AV et al. J AAPOS. Published online June 21, 2019.
2 Gillespie RL et al. Ophthalmology. 2016;123(1):217-220.
___________________________
Relevant financial disclosures—Drs. Drack and Utz: None.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
Dr. Chen National Institute on Aging: S; Quark Pharmaceuticals: C.
Dr. Drack Chakraborty Foundation: S; Canadian FFB: S; NIH: S; ProQr; S; Retrophin: S; Spark: S; Vision for Tomorrow: S.
Dr. Lass Alcon: S; Eversight Ohio: C; Transcend Medical: S.
Dr. Reiss Aerie: L; Alcon: C,L,S; Allergan: C,S; Bausch Medical: L; Glaukos: L; Haag-Streit: L; Santen/InnFocus: C,S.
Dr. Sauer Novartis: C; Tesseract: C.
Dr. Utz Retrophin: S; Springer Publishing: P.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|
More from this month’s News in Review