Download PDF
Because therapy for poststroke motor deficits follows the axiom “time is brain,” rehabilitation usually begins within a few days after the stroke. But when a stroke in the visual cortex leaves a patient with hemianopia or quadrantanopia, visual rehab therapy generally begins only after the patient’s vision has stabilized, at around six months.
But as it turns out, “time is vision,” too, according to researchers at the University of Rochester, New York.1
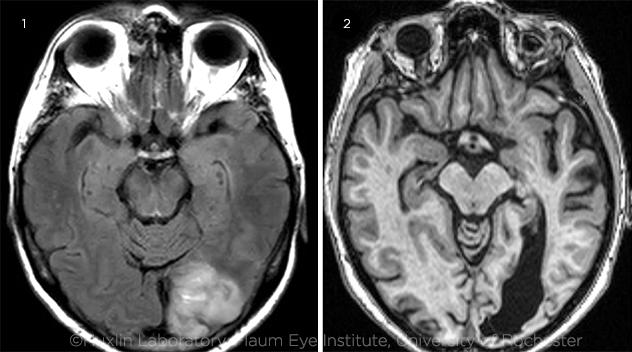 |
COMPARISON. Early visual training after an occipital stroke appears to halt further degradation of visual perception and may allow for greater recovery of vision than if given during the chronic phase. Brain imaging of visual stroke damage in (1) a subacute versus (2) a chronic patient.
|
Surprise finding. Using a special assessment and training system, the scientists discovered for the first time that after an occipital stroke, the brain retains small, residual visual pathways able to process vision in the hemianopic field. Early rehabilitation can strengthen these pathways, the researchers found—but without early intervention, the pathways cease functioning by six months following the stroke, said senior author Krystel R. Huxlin, PhD, at the University of Rochester.
“To our surprise, we discovered that vision loss was not immediate or absolute right after the stroke,” Dr. Huxlin said. “Instead, many visual functions appeared preserved when measured in the subacute period, disappearing by the onset of the chronic period.” For example, a portion of the subacute patients had measurable contrast sensitivity functions in their blind field. “To our knowledge, good luminance contrast sensitivity in perimetrically-defined blind fields has never been described in the literature on this patient population,” the researchers wrote.1
Study specifics. The researchers evaluated two groups of patients: those defined as subacute (evaluated less than three months after an occipital stroke; n = 18) and those defined as chronic (evaluated six months or more poststroke; n = 14). Both groups were tested for their ability to detect and discriminate the direction of motion of random dot patterns and luminance contrast gratings in the hemaniopic field.
After this initial evaluation, the patients were given testing software and a chin/forehead positioning device and instructed to do at-home practice of both tasks on a precise schedule. After about four months of home training, repeat testing in the lab (with controlled fixation) showed that both groups improved at discriminating motion direction, but the subacute patients improved much faster and over a larger area of their blind field than did the chronic patients, Dr. Huxlin said.
Will the benefits persist? Further research must be done to determine whether the training improvements will persist and to assess possible clinical benefits, Dr. Huxlin said.
Anecdotally, after undergoing training, the subacute patients reported that the ability to distinguish the presence and the movement of faint objects in their hemianopic field improved their ability to function as they went about their everyday tasks, Dr. Huxlin said. “They’re more confident about navigating in new environments and at getting around independently. They can actually detect objects, and they can tell when something is coming at them, so they don’t trip over the cat or bump into a pole or traffic sign while walking on a footpath.”
—Linda Roach
___________________________
1 Saionz EL et al. Brain. 2020:143;1857-1872.
___________________________
Relevant financial disclosures—Dr. Huxlin: Coinventor on U.S. Patent No. 7,549,743, which describes the visual retraining approach used in this research.
For full disclosures and the disclosure key, see below.
Full Financial Disclosures
David F. Chang, MD Carl Zeiss: C; Eyenovia: O; iDrops: C,O; Ivantis: C,O; Johnson & Johnson Vision: C; Mynosys: C,O; Perfect Vision: C; PowerVision: C,O; Presbyopia Therapies: O; RxSight: C; Slack: P; Surface: O; Versant Ventures: O; Viewpoint: C,O.
Dr. Cheng None.
Dr. Huxlin Clerio Vision: O. Dr. Huxlin also is coinventor on U.S. Patent No. 7,549,743.
Dr. Medeiros Aerie Pharmaceuticals: C; Allergan: C,S; Annexon: C; Biogen: C; Carl Zeiss: C,S; Galimedix: C; Google: S; Heidelberg: S; IDx: C; nGoggle: P; Novartis: S; Stealth Biotherapeutics: C; Reichert: C,S.
Disclosure Category
|
Code
|
Description
|
| Consultant/Advisor |
C |
Consultant fee, paid advisory boards, or fees for attending a meeting. |
| Employee |
E |
Employed by a commercial company. |
| Speakers bureau |
L |
Lecture fees or honoraria, travel fees or reimbursements when speaking at the invitation of a commercial company. |
| Equity owner |
O |
Equity ownership/stock options in publicly or privately traded firms, excluding mutual funds. |
| Patents/Royalty |
P |
Patents and/or royalties for intellectual property. |
| Grant support |
S |
Grant support or other financial support to the investigator from all sources, including research support from government agencies (e.g., NIH), foundations, device manufacturers, and/or pharmaceutical companies. |
|
More from this month’s News in Review